Noninvasive MRI digital physical exam provides a Whole Body approach for personalised health.
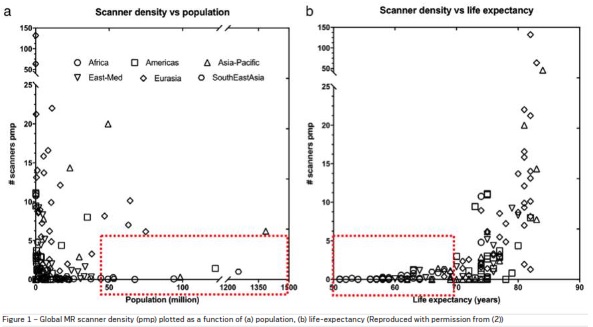
The scarcity of radiologic expertise and resources (hardware, personnel, quality, and other radiology services) impacts up to 4.7 billion people worldwide. Unfortunately, two-thirds of the world's population has no access to basic radiologic services. When it is available, both the quality and safety of the procedures may be questionable or even dangerous, both to the patient, the health care worker and the public. The gap in the access between high-income and low-middle-income countries is also significant (who.int). In the case of Computer Tomography (CT) for example, 14% of low-income countries have at least one CT equipment per million population (pmp) as compared to 100% in high-income countries. Nearly all African countries have reported less than 1 MRI scanner relative to Denmark’s 14 per million inhabitants. A similar divide exists for the availability of appropriate medical personnel.
Although the role of radiology is profound in patient screening, diagnosis, treatment guidance, monitoring and follow-up, due to the challenges with access, the application has remained primarily in diagnosis. The tremendous potential to use imaging as a preventative tool in healthcare remains largely untapped. Amongst
the various imaging modalities, Ultrasound (US) and Magnetic Resonance Imaging (MRI) do not use any ionising radiation for acquiring data from the body. While US is a portable versatile modality, it suffers from limited image resolution. The image quality also varies significantly based on technologist expertise.
MRI provides excellent soft tissue contrast with high
quality images. Access to MRI is limited due to its large
footprint, high cost and complexity in operation. The
global MR scanner density (pmp) plotted as a function
of (a) population and (b) life-expectancy is shown in
Figure 1. These demographics and countries represent the unmet need for accessible MRI (Geethanath,
Vaughan 2019).
Digital healthcare solutions can revolutionise
patient-to-provider encounters. Based on a study
(Kapral et al. 2013), after the implementation of a
digital care delivery system for stroke patients, the
proportion of patients who received appropriate care
increased from 40.0% to 46.5%, the 30-day mortality
after stroke decreased from 38.3% to 34.4%. Also, the
median wait time for treatment procedures decreased
from 50 days to 22 days.
Digitisation allows for utilisation of automated processing and analysis techniques including Artificial Intelligence (AI) to transform the data into meaningful results. For imaging data, AI-based processing and analysis yields simplified workflows, definitive diagnosis as well as predictive disease biomarkers. Figure 2 shows the addition of a quantitative imaging biomarker workflow to traditional radiological workflow.
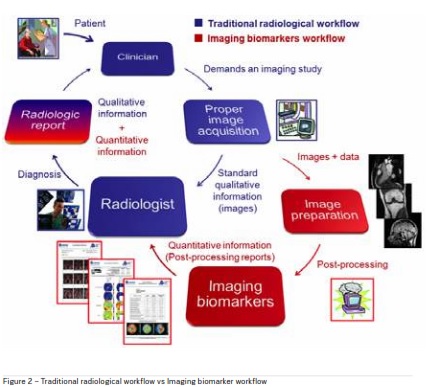
Factors impacting imaging (particularly MRI) access
The W.H.O. definition of access in healthcare includes
availability (availability of imaging solutions in the
market), affordability (cost effectiveness for patients
and healthcare providers), geographical accessibility
(reachable distance for patients), appropriateness
(scientifically validated, addresses local needs), acceptability (culturally acceptable to patients) and quality
(meets national standards for safe and effective use).
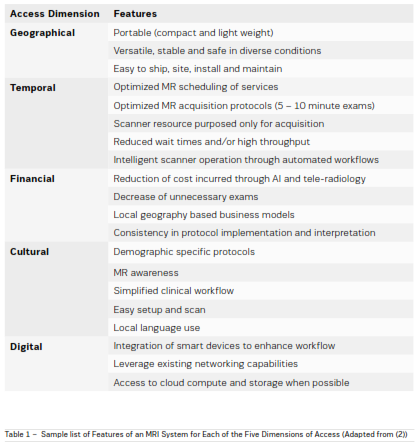
Access to care solutions can be perceived based on
five dimensions namely: geographical, temporal, financial, cultural and digital (Fortney, et al. 2011). A representative set of features for each of the dimensions of
access for an MRI system is shown in Table 1.
Based
on current state-of-art technology, Ultra Low Field (<10 mT) and Very Low Field (10 – 100 mT) MRI systems
have been primarily directed towards geographical and
financial access, while the higher-field systems have
relatively focused more on temporal, cultural and digital
accesses (Geethanath, Vaughan 2019).
The inherent digital nature of the data from most
imaging modalities (if not all in the near future), allows
for the application of automated analysis to obtain
valuable information beyond raw images. Medical
image analysis till the 1990s involved low-level pixel
processing and mathematical modeling to construct
compound rule-based systems that solved particular
tasks.
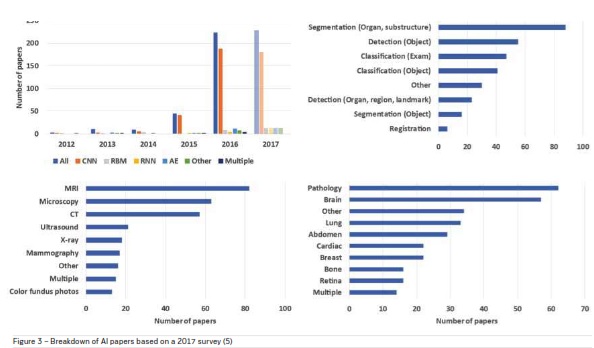
Today AI-based solutions, particularly deep learning
techniques have permeated the entire field of medical
image analysis. A breakdown of papers based on a
survey conducted in 2017 (Litjens et al. 2017) classified according to deep learning architecture, area of
application, imaging modality and anatomical region
is shown in Figure 3. The application of deep learning
methods for MRI data analysis was the highest.
Phenotyping the human body for precision
medicine
Normally people associate precision medicine with
genotyping – identifying an individual’s DNA sequence
(Ritzer-Ross 2015). But that is not the complete
picture. Just looking at an individual’s genome will not
necessarily create cohorts, mainly because of epigenetics, or the cellular trait variations that are caused
outside of the changes to the DNA sequence and other sources of variation in gene expression. While genotyping can create a baseline view of an individual’s
physical makeup, imaging observes or phenotypes the
physical manifestations of disease (Bonner 2015).
An accessible MRI solution with deep data analytic
capabilities creates the ability to peer into an individual’s body to evaluate health status, understand behavioral characteristics and predict disease risks. PhenoMx
Inc., is a digital health company, that has created a
digital physical examination, using noninvasive MRI,
to measure the major vital organs and tissues from a
Whole Body approach, for personalised health, wellness, and longevity. This enables the use of MRI as a
comprehensive screening tool (Ladd 2009), paving
way for affordable and actionable annual health exams.
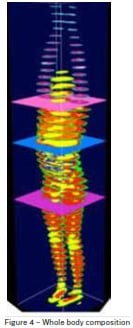
With a mere 20 – 30 minute scan, a detailed fingerprint of the body’s structure, composition and function
in health and disease can be determined. Figure 4,
shows a whole body composition map highlighting the
distribution of different tissues (bones, organs, muscle,
fat etc.) across the body.
Tracking, monitoring and evaluation of the tissue distribution with respect to an
individuals’ own baseline or a demographic population
can provide insights into metabolic anomalies, which
can serve as a biomarker for diseases such as obesity
(Shah, Braverman 2012), diabetes, cachexia, etc.
Body mass index (BMI) is commonly used to determine adiposity, which serves as an indicator of obesity
for practical reasons. However, BMI presents as an
inaccurate obesity classification method that underestimates the epidemic and contributes to failed treatment. For example, older adults have a particular body size, shape, and composition, and they tend
to lose fat-free mass and increase fat mass. Furthermore, Asians have a higher proportion of body fat for a
given BMI than Caucasians. The BMI does not consider
composition (lean mass vs fat mass). Athletes / wellmuscled individuals will thus fail a BMI test due to the
density of muscle. Alternatively BMI does not identify under muscled individuals (skinny fat), a growing
segment of the population whereby, scale weight may
fall within the approved BMI healthy ranges yet compositionally the individuals have excessive body fat levels
with dangerously low amounts of lean tissue to support
healthy aging. For these reasons, the Lean Mass Index
(LMI), calculated from the MRI-based body composition information has been proposed (© Grant Roberts,
Granite Gym). This purports to promote a positive spin
to assist in revolutionising mindsets.
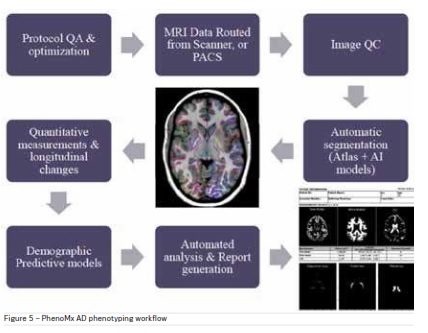
Anatomy specific image acquisition followed by
AI-based automated processing can be useful in
phenotyping various diseases. Multi-parametric MRI
for eg can provide a macroscopic picture of the Alzheimer’s Disease (AD) phenotype and its environment that
is ideally suited to quantifying the onset of the disease
phenotype before, during, and after treatment (Petrella
2013). For Alzheimer’s automated tools to segment
brain sub-regions and track volumetric changes in
these regions have been created. Population cohort
analysis and demographic clustering models developed using deep learning techniques help in databased understanding of cognitive impairment and
decline. An AD solution workflow (© PhenoMx Inc.) is
described in Figure 5.
Conclusion
Successful adoption of this solution at scale would
provide an opportunity to test multiple hypotheses on
biomarkers of health and disease, to facilitate paradigms of affordable and accessible, preventive and
interceptive healthcare. This would lead to critical
discoveries in healthcare and the globe, decisions in
policy and significant impact on lifestyle.
References:
Bonner L (2015) Radiology's Role in Precision Medicine. Diagnostic Imaging.com. Available from diagnosticimaging.com/practice-management/radiologys-role-precision-medicine
Fortney JC, et al. (2011) A re-conceptualization of access for 21st century healthcare. Journal of General Internal Medicine. 26,( Suppl 2): 639 - 647. Available from ncbi.nlm.nih.gov/pmc/ articles/PMC3191218/
Geethanath S, Vaughan JT (2019) Accessible Magnetic Resonance Imaging: A Review. Journal of Magnetic Resonance Imaging. Available from onlinelibrary.wiley.com/ doi/abs/10.1002/jmri.26638
Kapral MK, et al. (2013) Effect of
Provincial system of stroke care delivery on stroke care and outcomes. Canadian
Medical Association Journal. 185(10): E483-E491. Available from cmaj.ca/content/185/10/E483
Ladd SC (2009) Whole-Body MRI as a Screning Tool? European Journal of Radiology. 70(3): 452-462. Available from ejradiology.com/article/S0720-048X(09)00108-9/fulltext
Litjens, G, et al. (2017) A Survey on Deep Learning in Medical Image Analysis. 42(2): 60-88. Available from arxiv.org/abs/1702.05747
Petrella JR (2013) Neuroimaging and the Search for a cure for Alzheimer's Disease. Radiology. 269(3): 671-91. Available from ncbi.nlm.nih.gov/pubmed/24261497
Provincial system of stroke care delivery on stroke care and outcomes.
Canadian Medical Association
Journal. 185(10): E483-E491. Available
from cmaj.ca/content/185/10/E483
Ritzer-Ross J (2015) The Key to Precision
Medicine: Imaging. Radiology Business. Available from
radiologybusiness.com/topics/leadership/key-precision-medicine-imaging
Shah NR, Braverman ER (2012) Measuring Adiposity in Patients: The Utility of Body Mass Index (BMI), Percent Body Fat, and Leptin. PLoS ONE..7(4): e33308. Available from ncbi.nlm.nih.gov/pubmed/22485140