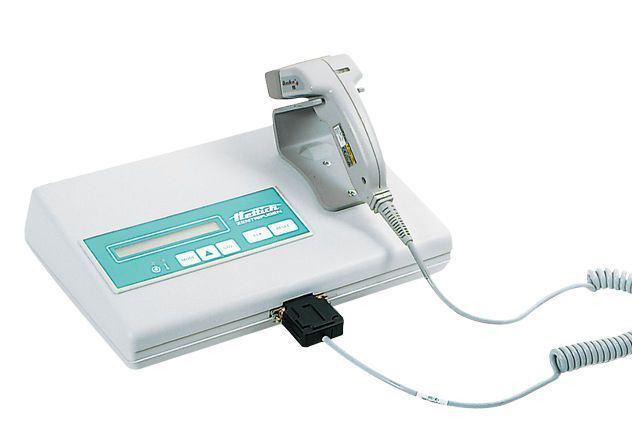
There is an increasing need to document procedures done in the laboratory. All information relating to manufacturing processes has to be properly documented. Quality Assurance Guidelines and accurate documentation of what goes on during production are important components of Good Manufacturing Practice. The paperwork that has to be accomplished by labs continues to increase in volume. HettInfo supports data documentation as well as the utilization of quality management system that has met all the requirement of GMP. For instance, producing blood bags based on GMP guidelines is one procedure that should meet strict requirements. Also, the results should be easy to reproduce again. Staff should also be there to monitor the manufacturing process and process data should be logged. HettInfo enables you to satisfy these requirements. The data documentation system is reliable in recording centrifugation data. Thus, it has an essential contribution to Quality Assurance.