Engineered bone marrow grown in a novel microfluidic chip device responds to damaging radiation exposure followed by treatment with compounds that aid in blood cell recovery in a way that mimics living bone marrow. This new bone marrow-on-a-chip microdevice holds promise for testing and developing improved radiation countermeasures, as described in Tissue Engineering, Part C, Methods, a peer-reviewed journal from Mary Ann Liebert, Inc., publishers.
Yu-suke Torisawa and coauthors from Harvard University (Boston and Cambridge, MA), Children’s Hospital Boston and Harvard Medical School show that the microdevice provides a way to keep the engineered bone marrow alive and to monitor the formation of different blood cell populations long enough after radiation damage to be able to evaluate the effects of experimental drugs being developed as protective agents.
In the article “Modeling Hematopoiesis and Responses to Radiation Countermeasures in a Bone Marrow-on-a-Chip,” the researchers report that unlike the microdevice, conventional static bone marrow culture methods do not mimic the recovery response of bone marrow in the body to these types of drugs.
“The development of relevant high-throughput systems is a field that will have huge impact in the near future for personalized medicine,” said Methods Co-Editor-in-Chief John A. Jansen, DDS, PhD, Professor and Head Dentistry, Radboud University Medical Centre, The Netherlands.
The hematopoietic system is highly sensitive to radiation injury, which results in bone marrow failure with neutropenia, thrombocytopenia, anaemia, and the associated risk of lethal haemorrhage or infection.
Despite a decade-long active search for medical countermeasures to radiation toxicity, to date no effective drug has been approved by the U.S. Food and Drug Administration (FDA) that could help protect or reconstitute the hematopoietic system. Current drug development studies rely on the use of animal studies for efficacy and safety testing; however, these animal models are expensive and rarely reflect human responses precisely.
“Thus, there is a critical need for predictive in vitro models to test effects of radiation countermeasures. Development of in vitro systems that accurately recapitulate bone marrow structure and function also could lead to new platforms for studying the mechanism of hematopoiesis, carrying out marrow toxicity screens, and testing for new drugs that more efficiently regulate blood cell formation,” the researchers said.
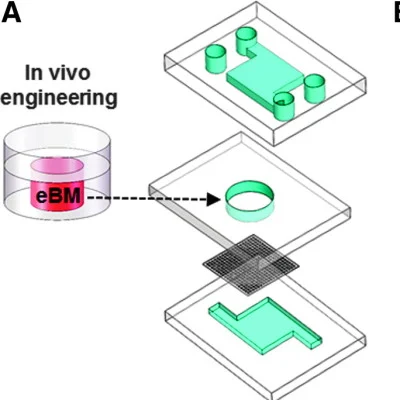
“We recently described a bone marrow-on-a-chip microfluidic device that enables the culture of living hematopoietic bone marrow and mimics radiation toxicity in vitro. In the present study, we used this microdevice to demonstrate continuous blood cell production in vitro and model bone marrow responses to potential radiation countermeasure drugs.
“The device maintained mouse hematopoietic stem and progenitor cells in normal proportions for at least two weeks in culture. Increases in the number of leukocytes and red blood cells into the microfluidic circulation also could be detected over time, and addition of erythropoietin induced a significant increase in erythrocyte production. Exposure of the bone marrow chip to gamma radiation resulted in reduction of leukocyte production, and treatment of the chips with two potential therapeutics, granulocyte-colony stimulating factor or bactericidal/permeability-increasing protein (BPI), induced significant increases in the number of hematopoietic stem cells and myeloid cells in the fluidic outflow.
“In contrast, BPI was not found to have any effect when analysed using static marrow cultures, even though it has been previously shown to accelerate recovery from radiation-induced toxicity in vivo,” the researchers concluded.