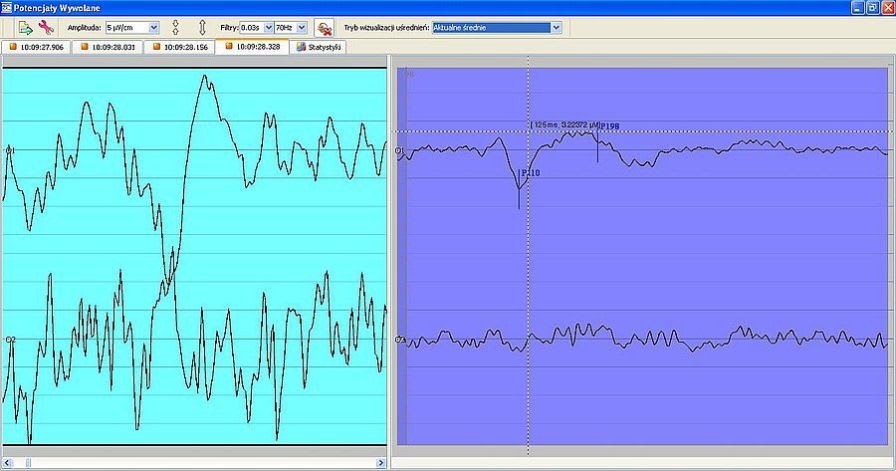
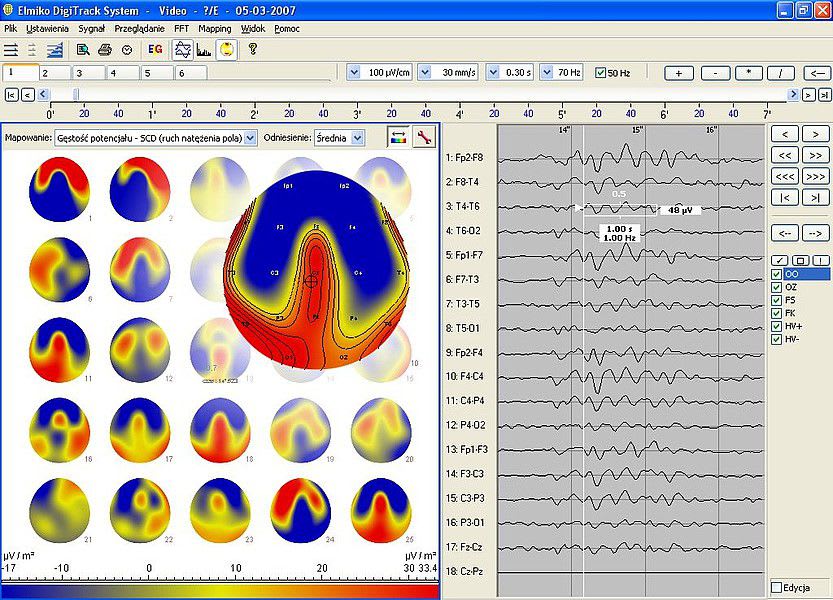
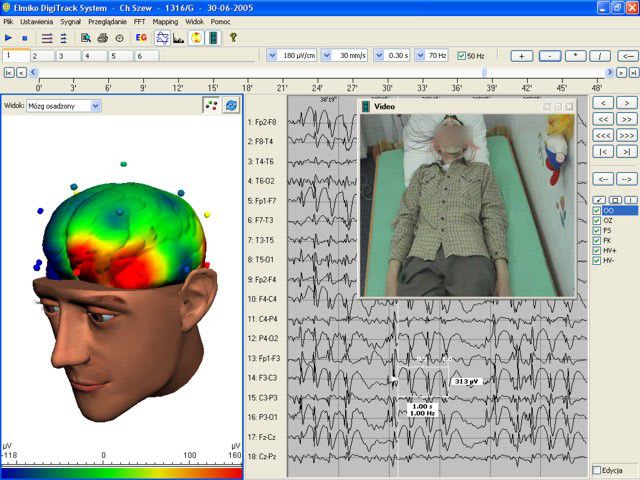
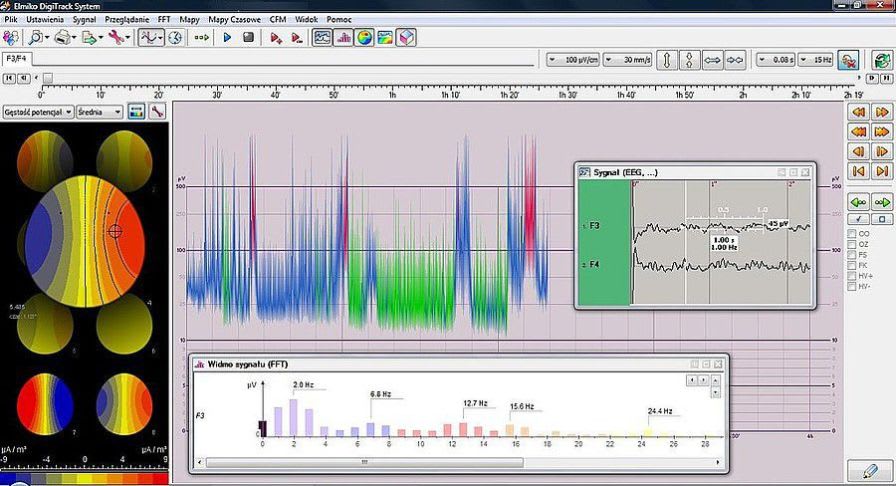
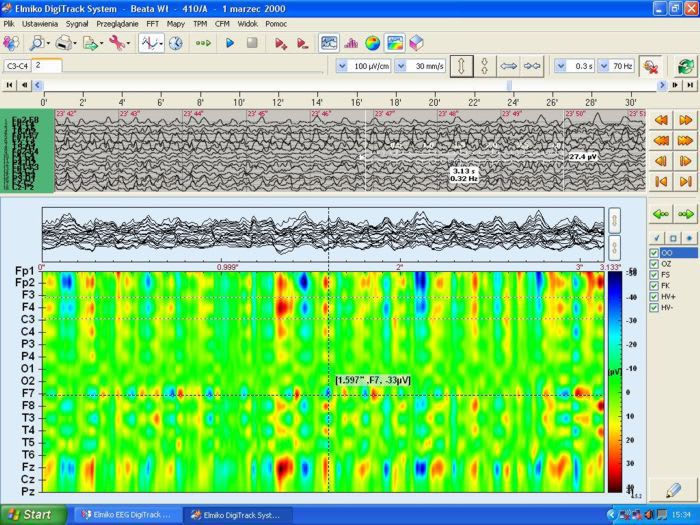
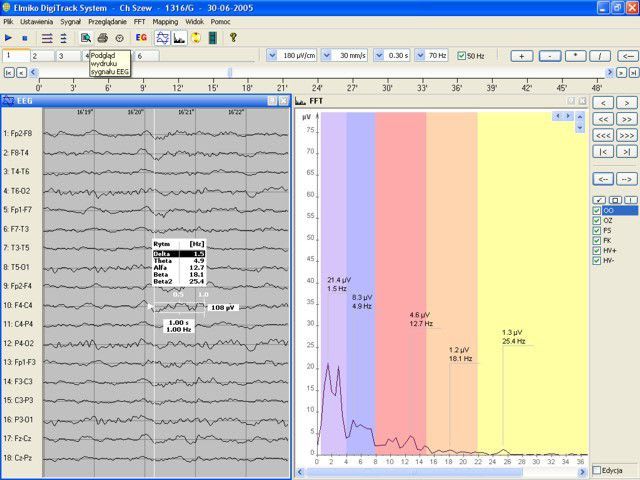
Company Profile ELMIKO MEDICAL - we are developing the future technology ELMIKO company was created in 1978. For over 35 years we have specialized in designing and developing medical electronics and IT solutions. Regular cooperation with important academic centres resulted in creation of a high-end medical device named EEGDigiTrack. Our highly qualified team of engineers and IT specialists constantly works to develop new solutions and improve the quality and functionality of our devices. Thanks to their effort every few months new versions of devices and software are introduced and the name ELMIKO connotates with the most advanced solutions for medicine.
Our equipment is used by scientific institutes, medical universities, hospitals and private clinics across the world. Moreover, ELMIKO also carries out innovative scientific projects involving biomedical engineering and biological signals digital processing. ELMIKO also supports education and publishing, popularizing psychiatric and neurological knowledge. and we are proud to have published numerous works of famous scientists. Our most recent publication is the first in Poland official manual for licence trainings for EEG Biofeedback specialists and therapists, published in co-operation with Polish Society of Clinical Neurophysiology. Since 2003 our Medical Studies Centre AKSON has been hosting trainings and workshops for doctors, offering information about EGG recordings analysis and interpretation, workshops for EEG technicians, EEG Biofeedback EMG and evoked potential workshops. Participants can develop their skills and acquire the PTNK licence. Our instructors are renowned neurologists and neurophysiologists.
ELMIKO is the only company in Poland with an accreditation of the Polish Society of Clinical Neurophysiology to conduct trainings for licences for adult and children EEG, EEG for technicians, EMG and EEG BIOFEEDBACK. Moreover, participating in our workshops awards education points and is approved by Supreme Medical Chamber. ELMIKO exports its products to many countries across the globe. In addition to producing our own equipment we are also the sole Polish distributor of many world-class medical equipment producers. In order to ensure the highest standard of services, we have implemented ISO 13485:2003 Quality Management System, certified byb12et Norske Veritas (DNV) and successfully passed CE certification for our products. In recent years ELMIKO has been dynamically developing, today we are a company with a clear vision and a leader on the Polish market for EEG devices.
Our experience and highly qualified and professional team guarantee the highest quality of our products. Today, ELMIKO is synonymous with modern technological solutions. We guarantee the highest standards of remote technical support and on-line trainings for our clients working in places across the globe. Today ELMIKO focuses only on production and research and development. To continuously improve the quality of our services and further widen the range of countries and sectors of activity in 2010 we created the ELMIKO MEDICAL LLC, the sole distributor of all ELMIKO products, which distributes EEGDigiTrack devices across the world. ELMIKO, ELMIKO MEDICAL and Medical Studies Centre AKSON form the complementary structure of ELMIKO MEDICAL