Incorporating Volumetric Breast Density and Imaging Performance Analytics in Quality Assurance Studies
Volpara Solutions is proud to announce that the Cancer Registry of Norway will expand the study of Volpara’s innovative suite of quantitative breast imaging software tools to now include seven of the 16 screening sites in the Norwegian Breast Cancer Screening Programme.
The Norwegian Breast Cancer Screening Programme will utilise the VolparaAnalytics software as part of its quality assurance protocols, monitoring radiation dose, breast compression and other factors to ensure safe and consistent imaging and patient experience.
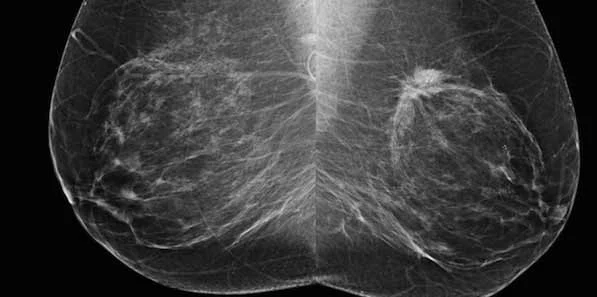
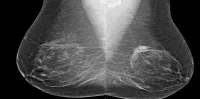
VolparaDensity has previously been installed to generate objective volumetric breast density measurements as part of a quality assurance protocol at a facility in Akershus University Hospital, and is now expanding the installation to six additional sites. The breast density measurements will be used to look at reader performance in the context of the quality of the images they are seeing and the density of the population. Furthermore, research will be conducted looking at how in the future screening might be adjusted according to risk and breast type.
A study from the Cancer Registry of Norway, published in Cancer, (Cancer 29 May 2013 DOI: 10.1002/cncr.28174) showed that the nationwide Norwegian mammographic screening programme reduced breast cancer mortality by 43 percent among attendees compared to non-attendees. Researchers estimate that that the 43 percent reduction in breast cancer mortality among attendees in the program corresponds to a reduction of more than 100 breast cancer deaths per year in a country with about 2.5 million female inhabitants. These are the first results from the screening programme in Norway using individual level data to study the effects of the mammographic screening programme on breast cancer mortality. The researchers analysed breast cancer mortality among almost 700,000 women aged 50 – 69 who were invited to the programme during the period 1996 – 2009.
“The Norwegian Breast Cancer Screening Programme, as evidenced by the reduction in breast cancer mortality reported in Cancer, is one of the leading mammographic screening programs in the world,” said Ralph Highnam, Ph.D., Volpara Solutions CEO and chief scientist. “We are honored to work with them to incorporate Volpara’s quantitative software tools into several centers to evaluate the use of volumetric density and imaging performance data in improving the program.”
Cleared by the FDA, HealthCanada, the TGA and CE-marked, VolparaDensity is in use at breast centers worldwide to help radiologists objectively assess density from both digital mammography and tomosynthesis images to more systematically evaluate who might benefit from additional screening. Highly correlated to breast MR assessments, VolparaDensity is a reliable tool that automatically generates an objective measurement of volumetric breast density correlated to the ACR (American College of Radiology) breast density categories. To date, nearly 4 million women have had their breast density analyzed using VolparaDensity. VolparaDensity is part of a suite of quantitative breast imaging tools built on the Volpara Solutions algorithm that allows for personalised measurements of density, patient dose, breast compression and other factors designed to help maintain accuracy and consistent quality in breast screening.
Source: Volpara Solutions
Image credit: Google Images
Volpara Solutions is proud to announce that the Cancer Registry of Norway will expand the study of Volpara’s innovative suite of quantitative breast imaging software tools to now include seven of the 16 screening sites in the Norwegian Breast Cancer Screening Programme.
The Norwegian Breast Cancer Screening Programme will utilise the VolparaAnalytics software as part of its quality assurance protocols, monitoring radiation dose, breast compression and other factors to ensure safe and consistent imaging and patient experience.
VolparaDensity has previously been installed to generate objective volumetric breast density measurements as part of a quality assurance protocol at a facility in Akershus University Hospital, and is now expanding the installation to six additional sites. The breast density measurements will be used to look at reader performance in the context of the quality of the images they are seeing and the density of the population. Furthermore, research will be conducted looking at how in the future screening might be adjusted according to risk and breast type.
A study from the Cancer Registry of Norway, published in Cancer, (Cancer 29 May 2013 DOI: 10.1002/cncr.28174) showed that the nationwide Norwegian mammographic screening programme reduced breast cancer mortality by 43 percent among attendees compared to non-attendees. Researchers estimate that that the 43 percent reduction in breast cancer mortality among attendees in the program corresponds to a reduction of more than 100 breast cancer deaths per year in a country with about 2.5 million female inhabitants. These are the first results from the screening programme in Norway using individual level data to study the effects of the mammographic screening programme on breast cancer mortality. The researchers analysed breast cancer mortality among almost 700,000 women aged 50 – 69 who were invited to the programme during the period 1996 – 2009.
“The Norwegian Breast Cancer Screening Programme, as evidenced by the reduction in breast cancer mortality reported in Cancer, is one of the leading mammographic screening programs in the world,” said Ralph Highnam, Ph.D., Volpara Solutions CEO and chief scientist. “We are honored to work with them to incorporate Volpara’s quantitative software tools into several centers to evaluate the use of volumetric density and imaging performance data in improving the program.”
Cleared by the FDA, HealthCanada, the TGA and CE-marked, VolparaDensity is in use at breast centers worldwide to help radiologists objectively assess density from both digital mammography and tomosynthesis images to more systematically evaluate who might benefit from additional screening. Highly correlated to breast MR assessments, VolparaDensity is a reliable tool that automatically generates an objective measurement of volumetric breast density correlated to the ACR (American College of Radiology) breast density categories. To date, nearly 4 million women have had their breast density analyzed using VolparaDensity. VolparaDensity is part of a suite of quantitative breast imaging tools built on the Volpara Solutions algorithm that allows for personalised measurements of density, patient dose, breast compression and other factors designed to help maintain accuracy and consistent quality in breast screening.
Source: Volpara Solutions
Image credit: Google Images
Latest Articles
Imaging, Tomosynthesis, breast cancer, Breast density, breast cancer screening, Volpara Solutions, mammogramme
Incorporating Volumetric Breast Density and Imaging Performance Analytics in Quality Assurance Studies Volpara Solutions is proud to announce that the Can...