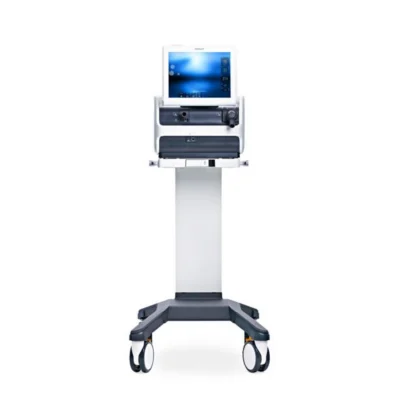
Getinge has received 510(k) clearance from the US Food & Drug Administration (FDA) for the company’s Servo-air® mechanical ventilator, which is part of the Servo family that supports intensive care ventilation ever since the first model was introduced in 1971.
"We are happy to be able to help clinicians in their endeavor to save lives by adding an additional ventilator to our product offering in the US market,” says Elin Frostehav, Vice President Critical Care at Getinge. “This is also an important step for our ventilation business in other parts of the world where 510(k) approval is a prerequisite for governmental tenders.”
Servo-air is a critical care ventilator. Intended for pediatric and adult patients, it includes both Invasive and Non Invasive (NIV) ventilation modes. Servo-air also features options for High Flow therapy and Servo Compass, which allows lung protective ventilation and makes it easier to follow the ARDSnet protocol.
“The turbine driven Servo-air is independent from wall gas. This, in combination with its high battery capacity, makes it suitable for use in intensive care units as well as for intermediate care and intra-hospital transportation,” says Malin Graufelds, Global Product Manager Servo-air.
She adds: “Servo-air has the same proven easy to use user interface with context based guidance as our high-end ventilator Servo-u, which facilitates both learning and managing a mixed fleet of Servo ventilators.”
Mechanical ventilation is a key component in the ongoing fight against the new coronavirus COVID-19. Although the majority of people with COVID-19 experience an uncomplicated or mild illness, some will develop critical illness and require mechanical ventilation.
“For patients with severe impairment of oxygenation, such as ARDS, it is important to use a ventilation strategy with the potential to improve patient outcomes and optimize the benefit-to-cost ratio for the lungs, heart and the respiratory muscles,” Elin Frostehav concludes.
More information about Getinge’s Servo-air can be found on Getinge’s website*:
*Depending on national regulations, the product offering and indications can differ from market to market.