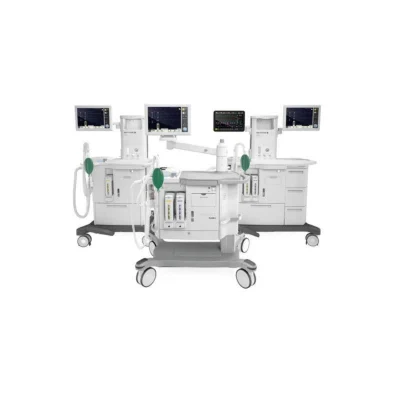
The Flow family is Getinge’s line up of anesthesia machines conceived to cover the needs of all types of surgeries and patients. Developed together with clinicians, these machines are designed to facilitate personalization with patient safety in mind. With an intuitive interface, emphasis on ease of use, smart design, and a high level of automation, the Flow Family brings personalized anesthesia delivery and smooth workflow to the clinician.
“Every detail of this machine has been designed in collaboration with clinicians to ensure optimal care with high efficiency. Flow-e has a smart workspace that makes work easier in the busy OR environment,” explains Lena Evander, Director Product Management Anesthesia at Getinge. “The tools are right where you would want them to be, which makes it possible to work in an ergonomic and comfortable position.”
“FDA 510(k) clearance of Getinge’s Flow-e and Flow-c allows us to complete the portfolio of Flow anesthesia machines and offers innovative solutions with personalized anesthesia for high aquity patients in the large and expansive US marketplace.” said Eric Honroth, President of North America.
The Flow Family core technology includes gas modules that are common with Servo ICU ventilators, allowing lung-protective ventilation and the optional Lung Recruitment Maneuvers tool, which is available on Flow-e and Flow-i models. Lung Recruitment Maneuvers have become the tool of choice to counter atelectasis, improve oxygenation, and help prevent postoperative complications. What was once considered complex and time consuming is now seamlessly integrated so that anesthesiologists can focus on high-acuity patients while monitoring their vital signs.1
The Flow-e and Flow-c will be launched with upgraded software, version 4.7, which includes the MAC Brain indicator, a unique tool that visualizes the difference in agent concentration between the lungs and target organ, the brain.2 The reliability of the data places anesthesiologists in control, which allows planning and delivery of more efficient agent dosing. The other members of the Flow family will have access to the software upgrade in the coming weeks.
All models in Getinge’s Flow Family include O2Guard, the world’s only system for active hypoxia prevention.3,4 O2Guard automatically overrules the settings and increases the flow of oxygen if the inspired oxygen level drops below 21%, minimizing the risk of hypoxia in challenging patient types.5
Getinge creates innovative solutions with the anesthesiologist in mind. The intuitive touchscreen gives the anesthesiologist one point of control for all functions, and the system’s simplicity of operation and innovatate features save time while contributing to more personalized anesthesia delivery. The Flow Family machines are designed to deliver personalized anesthesia with an ease of use and a streamlined workflow — when every measure matters.
Read more about Getinge’s Flow-e and Flow-c anesthesia machines.
References:
Lindberg P, Gunnarsson L, Tokics L, et al. Atelectasis and lung function in the postoperative period, Acta Anaesthesiol Scand.1992; 36:546-53.
Above 1 year of age – see indications for use
Hendrickx JFA, De Wolf AM, De Hert S. O2, anybody? Eur J Anaesthesiol.2015 Jun;32(6):371-3.
De Cooman, S, Schollaert C, Hendrickx JFA, et al. Hypoxic guard systems do not prevent rapid hypoxic inspired mixture formation.J Clin Monit Comput. 2015 Aug;29(4):491-7.