ICU Management & Practice, Volume 21 - Issue 3, 2021
and a discussion on optimal oxygen target values.
Oxygen is the most commonly used substance in hospitals today. After its discovery around 1771 by the German-Swedish pharmacist Carl Wilhelm Scheele and the English chemist Joseph Priestley, oxygen underwent rapid adoption as a drug in medicine and was already used in 1783 for the treatment of tuberculosis patients and in newborn asphyxia. However, oxygen synthesis remained problematic and sufficient amounts of oxygen only became available after the development of an air separation process by C. von Linde in 1902 that is still in use today. The Lubeca valve patented in 1889 by J. H. Draeger and his son B. Draeger was originally intended for the dosage of carbon dioxide in draught beer dispensers but served equally well as a pressure regulator for the dosage of medically indicated oxygen from compressed gas cylinders (Koehler et al. 2011). But soon, the first reports of oxygen toxicity surfaced. Laboratory mammals were exposed to different oxygen atmospheres over varying periods of time and e.g. suffered from seizures, pulmonary oedema or perished. Along with the spirit in these times, researchers verified the results on themselves, although these experiments were usually aborted before serious harm was done (Bornstein 1912).
Today, our understanding of oxygen has grown significantly and is not limited to the observation of its toxic effects. We know that in the blood, oxygen is predominantly transported by haemoglobin and that due to the sigmoidal binding curve, only little of oxygen is transported above a partial pressure of approximately 80 mmHg while the overall physically dissolved oxygen remains negligible under normobaric conditions. In the respiratory chain, oxygen is reduced by cytochrome c oxidase, contributing to the formation of a mitochondrial transmembrane gradient for protons, which is required for the synthesis of adenosine triphosphate. If sufficient oxygen is not available, the respiratory chain is interrupted, and energy production is limited to anaerobic glycolysis. Clinically, hypoxia results in lactic acidosis and necrosis of hypoxia-sensitive tissues such as central nervous system cells. Therefore, oxygen is vital for primates and its application life-saving in emergency situations when hypoxaemia is present (Grensemann et al. 2021).
However, sometimes we seem to have forgotten the results of early medical research that has pointed out adverse effects in terms of oxygen toxicity. Often enough, we see blood gas analyses in critically ill patients with partial pressures of oxygen far above the physiological range, despite our understanding that hyperoxia leads to an increase of free oxygen radicals (reactive oxygen species, ROS) that leads to cell damage resulting in apoptosis or necrosis. Additional factors such as an infection, in which pathogen associated molecular patterns (PAMPs) are released, further promote cell damage. Both apoptosis and necrosis lead to the release of further mediators and danger associated molecular patterns (DAMPs), which in turn, together with oxygen free radicals, cause further cell damage and thus maintain a vicious circle (Helmerhorst et al. 2015). The formation of ROS is favoured by a high oxygen partial pressure.
But is this clinically relevant as well? Looking at diseases in which a vascular occlusion is causative as in myocardial infarction or ischaemic stroke, at first sight it seems logical to increase oxygen tension in an attempt to improve oxygenation of the ischaemic tissue. However, this conversely might lead to the demise of tissue at risk by the vicious cycle explained above. With respect to the currently available data, this second mechanism seems to be in the lead.
In the prospective randomised AVOID trial in patients with ST-segment elevation myocardial infarction, one group received oxygen when oxygen saturation as measured peripherally by pulse oximetry was < 94%, whereas the other group received a fixed dose of 8 L/min (Stub et al. 2015). After six months, the fixed-dose oxygen group had a higher serum creatine kinase, a higher re-infarction rate as well as arrhythmias, and larger infarct size on magnetic resonance imaging.
In the DETO2X-AMI study, the oxygen group received a fixed dose of 6 L/min of oxygen, and the other group received oxygen only when peripherally measured oxygen saturation was < 90% (Hofmann et al. 2017). Patients with non-ST-segment elevation myocardial infarction (NSTEMI) were also included. Mortality did not differ, nor did the rate of reinfarction, atrial fibrillation, or cardiogenic shock.
In summary of these two studies, it must be postulated that hyperoxia may be harmful in myocardial infarction, but at least it has no benefit. Therefore, supplemental oxygen should not be given as long as peripheral oxygen saturation is ≥ 90%, as is now recommended by the European and American guidelines.
Furthermore, oxygen has a vaso-constrictive effect not only on the coronary system, presumably by antagonising nitric oxide (NO), which acts as a vasodilator. It could be shown that in patients without coronary artery disease, an acetylcholine provocation test induces coronary spasm in conjunction with breathing pure oxygen but not with breathing ambient air (McNulty et al. 2005).
Concerning ischaemic stroke, several prospective randomised trials with a combined total of over 8,300 patients investigated conventional oxygen administration in non-hypoxaemic patients with ischaemic stroke. The largest study of these was the SO2S trial with over 8,000 patients alone, in which one group received oxygen only at night, one group received oxygen continuously, and one group received no oxygen. However, a minimum oxygen saturation of 94% was maintained in all groups. There was no difference in outcome measured according to the modified Rankin scale (Roffe et al. 2017). This approach has also found its way into the guidelines, which recommend maintaining a target oxygen saturation between 94% and 98% in ischaemic stroke.
What are the actual data when focusing on patients without underlying ischaemia? For mechanically ventilated intensive care patients, a retrospective analysis over ten years ago identified a U-shaped relation between oxygen partial pressure and mortality with the highest survival between partial pressures of approximately 65 and 80 mmHg (de Jonge et al. 2008). Interestingly, these limits are close to the values one would expect from a strictly theoretical point of view: below 60 mmHg, oxygen saturation drops below 90% with an increasing risk for hypoxia and above 80 mmHg only little additional oxygen may be transported while the formation of ROS increases. Several randomised controlled trials have been conducted in this patient population since then trying to identify the optimal range for the oxygen partial pressure and to prove superiority for this target range.
The OXYGEN-ICU study with 434 patients demonstrated that ICU and in-hospital mortality were lower when treated with a median partial pressure of oxygen of 87 mmHg vs. 102 mmHg [ICU mortality 11.6% vs. 20.2%, relative risk 0.57, p=0.01] (Girardis et al. 2016). It must be noted though that this study was terminated prematurely due to an earthquake, as no further patients could be included due to the degradation of the necessary infrastructure. Furthermore, although randomised, patients differed in baseline characteristics with a slightly higher predicted mortality in the liberal oxygen group.
The HYPER2S trial in ICU patients with sepsis was stopped for safety reasons after 442 patients due to a significantly higher rate of severe adverse events in the hyperoxia group [85% vs. 76%, p=0.02, including muscle weakness, atelectasis] (Asfar et al. 2017).
In the ICU-ROX study, no difference in mortality was found between the groups in 965 patients. However, one might argue that this study compared like with like as virtually no difference existed between the two groups: one group aimed at an oxygen saturation of 90-97% which resulted in a mean partial pressure of approximately 80 mmHg, while the other group targeting an oxygen saturation of > 90% reached a partial pressure of approximately 90 mmHg (Mackle et al. 2020).
Very recently, the LOCO2 study comparing groups with an arterial oxygen partial pressure of 55-70 and 90-105 mmHg was terminated after the enrollment of 205 of 850 originally planned patients, because it did not appear likely that a difference in the primary outcome measure, the mortality at day 28, could be reached (Barrot et al. 2020). Furthermore, safety reasons were brought up. A slight difference in one of the secondary endpoints, the mortality at day 90 was higher in the lower oxygen group (44.4% vs. 30.4, difference: 14% with 95% confidence interval 0.7 to 27.2). For the interpretation of the results, it is noteworthy that this study included ARDS (acute respiratory distress syndrome) patients with a p/F-ratio of around 120 ± 50 mmHg and an inspiratory fraction of oxygen of 0.8 ± 0.2 in both groups. No other study had included patients with such severe disturbances in pulmonary gas exchange, so far. For interventions as e. g. suctioning, no preoxygenation was performed and this presumably induced hypoxia on several occasions that should be avoided.
The O2-ICU study from The Netherlands (clinicaltrials.gov NCT02321072), comparing 60-90 with 105-135 mmHg is currently still recruiting patients.
The question of the optimal oxygen partial pressure for ventilated intensive care patients currently remains unanswered, but it is undisputed that hyperoxia certainly offers no benefit for this group of patients.
For acute exacerbated chronic obstructive pulmonary disease (COPD), the main focus of treatment undoubtedly lies on the relief of the fatigued respiratory musculature, preferably by non-invasive ventilation. However, special attention should be given to therapy with oxygen as a too liberal application has been shown to have adverse consequences. Oxygen application can result in a further increase in arterial partial pressure of carbon dioxide with subsequent CO2 narcosis. For a long time, the mechanism was thought to be due to desensitisation of the peripheral chemoreceptors in the glomus caroticum involved in respiratory control, caused by the chronic hypercapnia that occurs in COPD. In this case, much of the respiratory drive would be triggered by hypoxaemia, which is reduced by oxygen administration; this would result in a decrease in respiratory minute volume with consecutive hypercapnia. According to experimental studies, this mechanism may only be responsible for a fraction of the observed hypercapnia, with a reduction in respiratory minute volume of approximately 15% and may for example, mathematically account for only 5 mmHg of an increase in carbon dioxide partial pressure of 23 mmHg. Most of the hypercapnia is due to changes of the ventilation/perfusion ratio in the lungs. Oxygen application suppresses hypoxic vasoconstriction, which is important for maintaining an optimal ventilation/perfusion ratio, and results in perfusion of sub-optimally ventilated alveoli and an increase in alveolar dead space. Furthermore, the Haldane effect is involved, which describes that deoxygenated haemoglobin is able binding carbon dioxide in form of carbamino compounds via amino groups and release it after oxygenation in the lungs.
In hypercapnia as present in acute exacerbated COPD, the ability of haemoglobin to bind further carbon dioxide is reduced. Normally, the carbon dioxide would be released in the lungs and exhaled, but this is no longer possible in these patients due to the already maximal respiratory effort. Oxygen administration results in a reduction in the binding capacity of haemoglobin for carbon dioxide with additional release of already bound carbon dioxide, which then also increases hypercapnia (Abdo and Heunks 2012).
That this is clinically relevant could be shown in a randomised trial: 405 patients were treated prehospitally with titration of oxygen to an oxygen saturation between 88-92% or with a fixed oxygen therapy of 8-10 L/min (Austin et al. 2010). In the group treated with titrated oxygen, mortality was significantly lower (2% vs. 9%), likewise acidosis was higher for liberal oxygen administration, and mean pH was 0.12 lower than in the group receiving titrated oxygen. This trial resulted in recommendations to titrate oxygen in acute exacerbated COPD to achieve a target range for the oxygen saturation between 88%-92% [UK recommendation as well as international recommendation of GOLD (Global Initiative for Chronic Obstructive Lung Disease)].
Although these examples show that oxygen should be applied cautiously, every coin has two sides. In cases of carbon monoxide intoxication, the pathophysiological quandary of a 200-fold affinity of carbon monoxide to haemoglobin as compared to oxygen must be solved to preserve oxygen transport and prevent from tissue hypoxaemia. As carbon monoxide competitively displaces oxygen from haemoglobin the solution is to increase oxygen tension and thus shift the chemical flow equilibrium to the oxygen side. The half-life of the carboxyhaemoglobin is decreased from 320 min in breathing ambient air to 74 min when breathing pure oxygen. The half-life may be further decreased under hyperbaric conditions [e. g. 20 min at 2.5 bar] (Rose et al. 2017; cdc.gov/disasters/co_guidance.html). However, transport of a critically ill patient to a hyperbaric chamber may be a cumbersome challenge and as no consensus could be reached so far, is regarded as optional.
Besides carbon monoxide intoxication, patients with cardiac arrest should be ventilated with pure oxygen as this increases the chances for a return of spontaneous circulation. However, after successful resuscitation, it is recommended to lower oxygen partial pressures to a physiological range to prevent from ROS mediated cell damage.
One question that has received limited attention so far is if oxygen could exhibit a dose-effect relationship. We often create a short-term hyperoxia in the context of preoxygenation before medical procedures as e.g. endotracheal suctioning or endotracheal intubation. This leads to a denitrogenisation of the pulmonary functional residual capacity, creating an “oxygen reserve” to prolong apnoea tolerance and thus prevent hypoxaemia. Currently, it is not known whether this short-term hyperoxia may already have negative effects on the outcome of these patients, but should, on the other hand, be balanced against the deleterious effects of hypoxia.
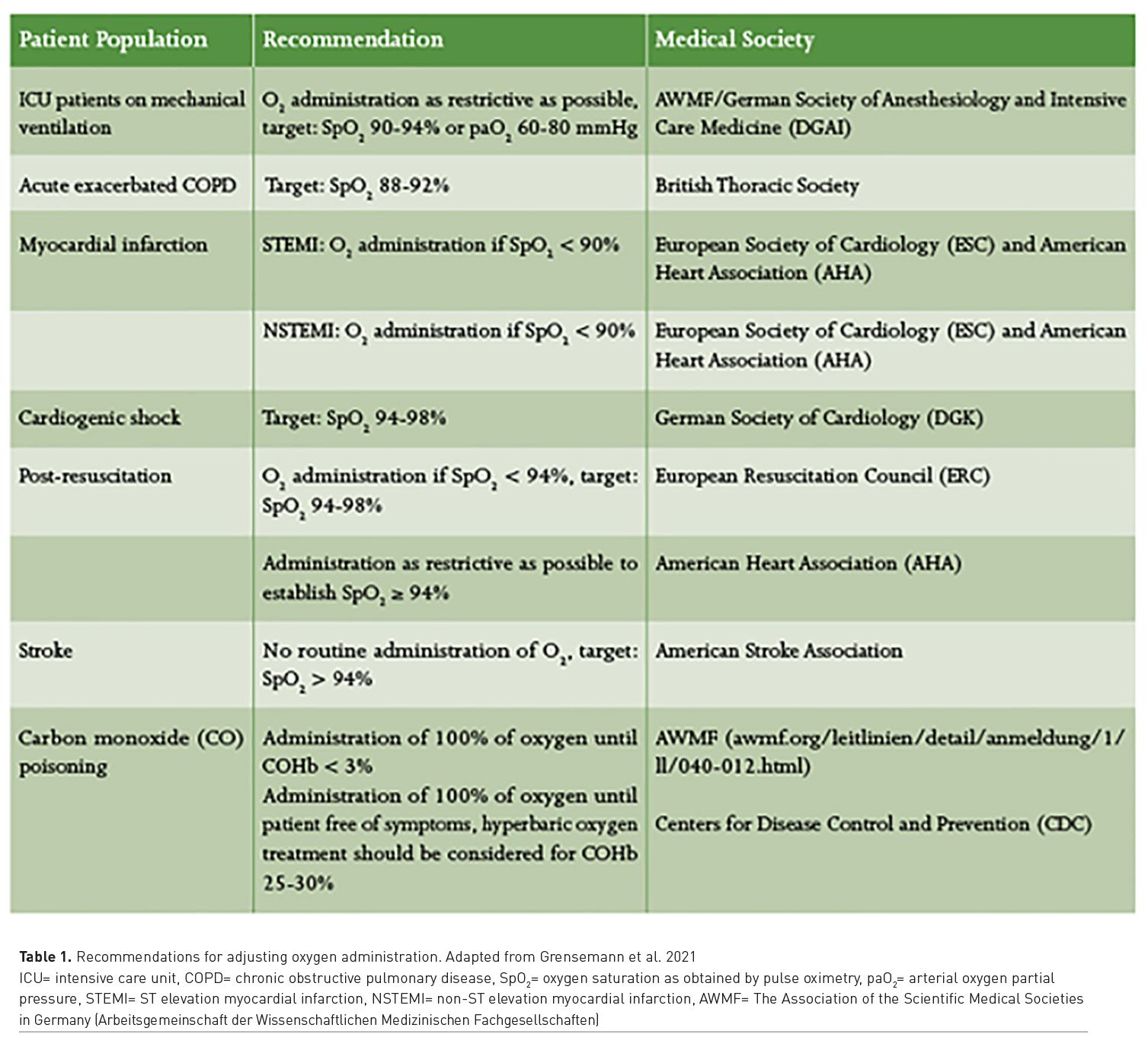
Regarding this question, one retrospective study analysed approximately 700 patients in an emergency department that were ventilated for a median of approximately 5.5 hours before being transferred to an intensive care unit (Page et al. 2018). Patients were categorised based on partial pressures of oxygen. The highest mortality of approximately 30% was found in the hyperoxia group (pO2 > 120 mmHg); in contrast, mortality in the normoxia group (pO2 60-120 mmHg) was only approximately 20%. The result was statistically significant. Although this is a retrospective evaluation, these data may provide a first indication that even a relatively short exposure to hyperoxia over a few hours could have negative effects on outcome. However, a change in clinical practice is not warranted from these data; presumably, the benefit of preoxygenation on the avoidance of hypoxia outweighs the disadvantage of oxygen toxicity. Most recently, the randomised, prospective HOT-ICU study having enrolled 2928 patients from 35 ICUs in Denmark, Switzerland, Finland, the Netherlands, Norway, UK and Iceland between 2017 and 2020 has been published. Control group (targeted PaO2 of 90 mmHg) and intervention group (targeted PaO2 of 60mmHg) were similar, i.e., pneumonia was the main acute illness: 57.7% vs 57.4%. As main result, 90-day all-cause mortality was not significantly different (42.9% vs 42.4%, p = 0.64). Furthermore, secondary outcomes (alive without life support, alive after hospital discharge) and serious adverse events (shock, myocardial ischaemia, ischaemic stroke and intestinal ischaemia) were comparably often. For illustration, current recommendations for adjusting oxygen administration in different patient populations are summarised in Table 1.
Although the quest for optimal oxygen target values is still ongoing and a possible dose-effect relationship should be elucidated in the future, it is essential to implement oxygen targets in clinical practice as hyperoxia may be harmful or at least, have no benefit (Grensemann et al. 2018). The implementation may prove difficult due to long-time engraved mental models that only hypoxia increases mortality. On the other hand, a step-by-step implementation strategy accompanied by appropriate interprofessional training has been proven safe and feasible (Helmerhorst et al. 2016).
Conflict of Interest
None.








