The following is an overview of the DIVINE trial (Dietary Management of Glucose Variabilty in the ICU) as well as a quick summary of the role of glucose control and outcomes in critically ill patients. The DIVINE study was funded by Nestlé.
Clinical studies show that goal nutrition may not result in the best outcomes. Available data suggest that protein may be more important than non-protein calories. Findings from a study conducted by Peter Weijs1 and their group show that early protein intake at a level of ≥1.2 g/kg at day 4 of ICU admission is associated with lower mortality and early energy overfeeding is associated with higher mortality in non-septic mechanically ventilated critically ill patients.
Another study2 shows the association of administered calories/resting energy expenditure with mortality and protein intake. Findings show that the lowest mortality was observed among those who were within 60 to 80% of their goal calories whereas protein mortality was almost linear, thus suggesting that mortality goes down with more delivery of protein.
Hyperglycaemia is common in critically ill patients for a number of reasons, one of which is that critical illness worsens insulin sensitivity and resistance. It is thus associated with the severity of critical illness. This is also a probable cause of worse outcomes. It is not just the levels of glucose, but it’s actually the variability of the glycaemic variability index that accounts for these outcomes. A clinical study3 was conducted with 759 patients to evaluate glycaemic variability and its association with outcomes. Out of the 759 patients, 651 survived, and 108 died. Among the factors that could be associated with death, glycaemic variability was also highlighted, defined in this study as the standard deviation/mean blood glucose x 100. Hyperglycaemia and hypoglycaemia may both worsen outcomes.
In the NICE-SUGAR study4 conducted with 6000 sepsis patients, and two different randomised sugar targets, it was found that in both of those groups, hypoglycaemia was associated with worse outcomes, specifically worse mortality. The more severe the hypoglycaemia, the higher the association with outcomes suggesting a dose-response. The more severe and the longer the hypoglycaemia, the bigger the hazard ratio for mortality.
The objective of the DIVINE5 study was to determine whether blood glucose control could be facilitated by using enteral nutrition formula that contained low carbohydrates, medium-chain triglycerides and very high levels of hydrolysed whey protein to ensure optimal protein delivery. It is an open-label, multi-centre trial at seven academic medical centres in North America. The trial went on for almost two years and included mechanically ventilated, critically ill obese and overweight patients (BMI between 26 and 45) who were thought to require enteral nutrition for at least five days. Patients with hepatic failure or those admitted for trauma or major surgery or pregnant were excluded from the study.
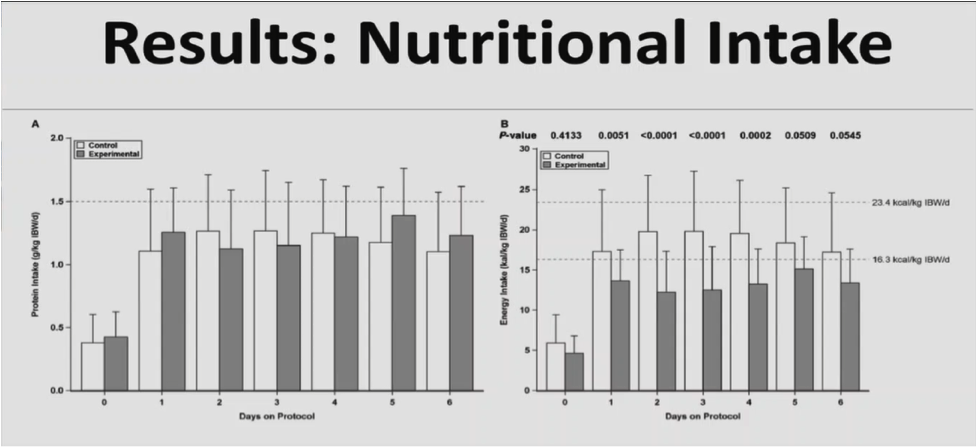
The control group received a high protein formula, and the experimental group received a very high protein formula with low carbohydrates. The control formula had a caloric density of 1, and so did the interventional formula. But it had lower protein and higher carbohydrate with similar amount of fat as the experimental protein. The goal in both of the groups was to try and deliver 1.5 g/kg IBW/day of protein.
The endpoint of the study was the rate of glycaemic events outside of the interval of 6.1 to 8.3 mmol/L in the first seven ICU days. Secondary endpoints included serial blood glucose, markers of nutritional status, urine/serum ketones, insulin, and dextrose administered, and clinical outcomes. A total of 105 patients were randomised. 102 patients had glucose measurements that allowed them to be included in the intention to treat analysis.
Both groups received similar amounts of protein, but the experimental group received fewer carbohydrates. The experimental group got about half as much carbohydrate as the control group, and fat was similar between the two.
Figure 1: Results of the DIVINE study
Both groups got about 70% of their target. There was no difference in the rate of glycaemic events outside of the interval of 6.1 to 8.3 mmol/L. There was a significant increase in the mean rate of glycaemic events that were between 4.4 to 6.1 mmol/L. This was an area of concern and the primary reason why the trial was stopped. There was also a significant decrease in values above 8.3 mmol/L.
The mean glucose was significantly lower in the experimental group: 7.0 versus 7.7 mmol/L. There was no difference in the rates of hypoglycaemia defined as glucose levels less than 4.4 mmol/L. There was a smaller glycaemic dispersion in the experimental group. The experimental group also received less insulin, so there was less insulin administered both in the amounts and the number of administrations in the experimental group and no difference in the amount of rescue dextrose that was given.
There was some increased frequency of abdominal distension in the experimental group, but overall the number of patients with adverse events in both groups weren’t different. Mortality, in general, was low in this trial but it was numerically lower in the intervention group than in the control group but not statistically significant. Why did patients get better control in the experimental group? There could be a number of potential reasons for this, and multiple of these could be at play.
One is that a higher protein load probably improves insulin sensitivity. The second is the type of protein matters, and whey protein improves insulin sensitivity. The third is that if you give fewer carbohydrates, you probably have better glucose control. In general, if you give fewer calories, you actually have better glucose control.
To summarise the findings of the DIVINE study, a very high hydrolysed whey protein low carbohydrate formula facilitated blood glucose control in critically overweight and obese patients. Although it didn’t reduce the number of events outside of the interval of 6.1 to 8.3 mmol/L, it did lower dispersion of blood glucose as measured by standard deviations and had a lower incidence of hyperglycaemia defined as glucose > 8.3 mmol/L.
Nutritional support for critically ill patients needs to be individualised, and that includes individualised plans for obese patients. Current data suggest that moderate permissive underfeeding while administering higher levels of protein may improve outcomes of critically ill obese patients. Avoiding hyper and hypoglycaemia likely does improve outcomes, and as this study suggests, that can be accomplished by specific nutritional formulas. Further research is required to see if these nutritional formulas actually improve clinical outcomes and not just blood sugar control.