In the past decades there have been significant advances in the diagnosis and management of patients with sepsis and septic shock, and overall awareness has increased significantly (Angus and van der Poll 2013). Emphasis is currently on the early detection of sepsis and rapid initiation of fluid administration and antibiotic therapy, all of which have improved outcomes (Rhodes et al. 2017). Nevertheless, discussion remains about the targets for fluid resuscitation, the optimal type of fluid and many other aspects of sepsis management, and this directs scientific research in the field (Perner et al. 2017).
While there is consensus that antibiotic therapy and source control are the major therapies for severe infections, source control has been consistently ignored by many studies, and its exact role, particularly the timing and methodology used, remains uncertain. Source control is receiving only limited attention in the first hour of sepsis treatment, disproportional to its impact on outcome. It has proved hard to accurately define source control, and quantifying it is even more difficult. But insights into the role of source control are evolving, and both the epidemiology and methodology will surely receive more consideration in the next years. Currently, exact data on the impact of source control, or data that provide adequate guidance on the timing and preferred method for source control remain scarce.
1. Defining source control
The definition of source control has not changed over the years, yet this definition is more a conceptual approach, focusing on the goal of source control rather than the exact method to reach these goals. Source control is defined as the different measures that are used to eliminate the source of an infection, control ongoing contamination and restore premorbid anatomy and function (Schein and Marshall 2002). It is true that source control is most often thought of in patients with abdominal infections because of the ongoing contamination (De Waele 2016) but source control should be considered in every patient with sepsis or septic shock. In fact, up to 45% of patients with sepsis and septic shock require some form of source control (Bloos et al. 2017). Not every patient of course may require a surgical procedure, but also in patients with presumed non-surgical infections source control may be considered e.g. in patients with bloodstream, urinary tract or respiratory infections.
Most of the time the focus is on the first two goals of source control, namely eliminating the source of infection and controlling ongoing contamination. Source control can involve a surgical procedure, percutaneous drainage using a catheter (that either remains in place or not), incision of an abscess, removal of necrotic tissue or removal of an infected device e.g. central venous catheter or external ventricular drain.
Historically, the evidence came from non-randomised controlled trials, mainly in necrotising fasciitis, with multiple case series conducted in the 1990s (Elliott et al. 1996). The data were in favour of an aggressive operative approach. An expert opinion roundtable in the mid 2000s highlighted that appropriate source control should be part of the systematic checklist we have to keep in mind in setting up the therapeutic strategy in sepsis (Marshall et al. 2004).
A practical approach to define source control is rather ambiguous in some guidelines. For instance, in the recent Surviving Sepsis Campaign (SSC) guidelines from 2016 the recommendation was:
We recommend that a specific anatomic diagnosis of infection requiring emergent source control be identified or excluded as rapidly as possible in patients with sepsis or septic shock, and that any required source control intervention be implemented as soon as medically and logistically practical after the diagnosis is made (Rhodes et al. 2017).
Interestingly the recommendation was acknowledged as a “Best Practice Statement”.
2. Source control in critically ill patients
Applying source control and its principles in critically ill patients poses specific challenges. First, the patient is more severely ill, with less room for compensating for the consequences or complications of a procedure; second, the urgency of the need for source control is equally different. Leaving a patient exposed to an untreated infection can have more severe consequences compared to patients who present without sepsis, even if antibiotic therapy and fluid resuscitation have been initiated.
The proportion of patients with sepsis that requires source control clearly depends on the type of infection that is causing sepsis. In the multicentre study by Bloos et al. (2014), 42% of patients with septic shock required source control. In this study, the majority of these procedures was surgical (85%), but this may vary according to the source of the infection, presence of ongoing contamination, surgical history and general condition as well as co-morbidities of the patient.
3. Available methods for source control
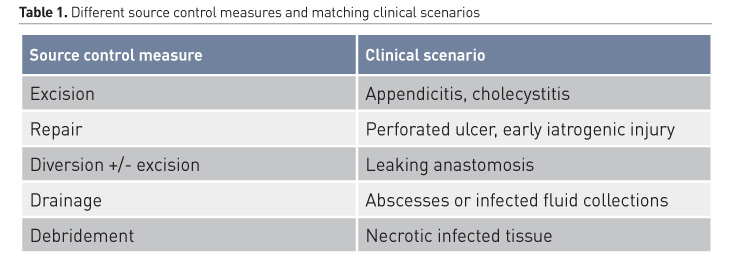
Based on the aspect that source control consists of those definitive measures to control a source of ongoing microbial contamination and to restore anatomy and function, definition of source control can be integrated in five categories (Table 1).
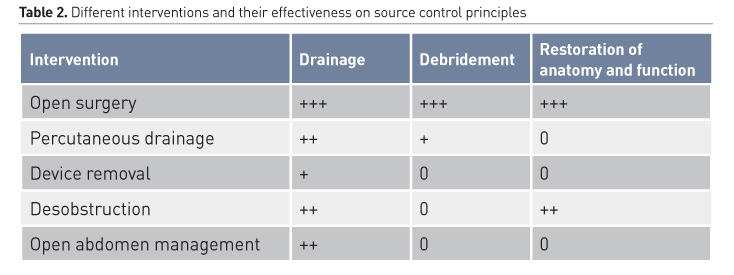
Source control has long been synonymous with a surgical procedure, mostly a laparotomy or other open intervention, but this is changing significantly. Firstly, laparoscopy and minimally invasive procedures have replaced open surgical procedures, although in critically ill patients laparoscopy may be less tolerated. The main evolution regarding source control has been the rise of percutaneous drainage of abscesses in many locations, either ultrasound or CT-guided (Soop et al. 2017). Table 2 provides an overview of the different interventions and how effective they are in regard to the different source control principles.
Open or endoscopic surgery is still the most controlled method of source control. It is very effective in completely draining collections or abscesses and debriding necrotic tissue. Also, restoration of anatomy and function is straightforward.
Percutaneous drainage (PCD) is effective in draining a large part of most collections, although mostly a small residual amount will remain. This is often managed by rinsing the catheter and collection in order to remove the last remainders of the infection. For multiloculated infections multiple catheters may be needed, and also more residual infection may have to be tolerated. Implicitly the residual infection may continue to produce signs and symptoms of infection and the response to the treatment may be more difficult to monitor. Debriding necrotic tissue is even more difficult and again a considerable amount of necrosis may need to be tolerated. Repairing anatomical lesions is not possible using PCD.
But also simple interventions such as device removal can be considered for source control. Removing an obstruction such as a urinary tract lithiasis or choledocholithiasis can be effective in draining the infection, but again no meaningful debridement is possible. Anatomy and function are mostly adequately restored after such a procedure. Open abdomen management can also be part of a source control procedure, effective mostly for draining the abdominal cavity. Restoration of anatomy and function will only follow later.
Strategies for source control are improving continuously and no doubt will do so in the future. The trend towards minimally invasive procedures will continue, with more advanced endoscopic (ultrasound-guided) procedures as the most important development. As an example, transgastric endoscopic drainage and necrosectomy for infected pancreatitis (van Brunschot et al. 2018) is a promising technique that obviates the need for open surgery, often fraught with complications.
It remains to be demonstrated that patient condition makes no difference in selecting the best source control procedure, and in studies reporting on new techniques severity of illness should definitely be considered to make sure that the most vulnerable patient benefits from the most optimal procedure.
4. Importance of source control
Data supporting the role of source control are limited although several studies have recently focused on this important issue. From these data, two relevant aspects are consistently reported: source control adequacy and timing of the intervention.
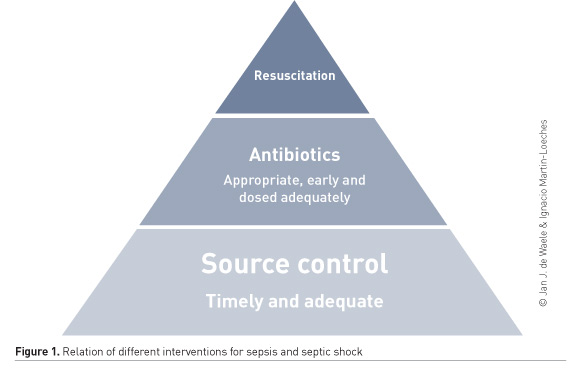
The impact of source control seems to be unrelated to the administration of appropriate antibiotics. Several studies found that both are independent predictors of mortality (Bloos et al. 2015; Tellor et al. 2015), but there is consensus that without adequate source control, antibiotic therapy may have little if any effect (Figure 1).
a) Adequacy of source control
Inadequate source control seems to be a relatively frequent problem, but source control has been inconsistently defined in the literature, and often there is even no definition or description provided. Source control adequacy is determined by source of infection, source control intervention, patient type, definition used, methodology applied among other factors.
Logically, not controlling the source of infection should be included in a definition of inadequate source control, but there is no consistency in other elements of source control adequacy. Some definitions include both timing (e.g. within 24h) as well as pure technical considerations (did the surgical procedure result in control of the ongoing infectious process?) (Tellor et al. 2015), whereas others fail to have a clear definition of source control (Bloos et al. 2014; Coccolini et al. 2015; Martínez et al. 2017).
Data on the extent of the problem in critically ill patients is unclear. In patients with complicated diverticulitis, source control adequacy was reported to be as high as 91% (Coccolini et al. 2017). Bloos et al. (2014) reported 86.7% source control adequacy although no clear definition was provided (“unsuccessful procedure”). In some studies source control adequacy is evaluated by a panel of surgeons (Tellor et al. 2015), whereas in other studies this is not specified (Coccolini et al. 2015; Bloos et al. 2014).
Given the importance of source control, a clear definition of source control adequacy and methodology used is essential in order to better understand the role and develop the best approach to patients requiring source control interventions.
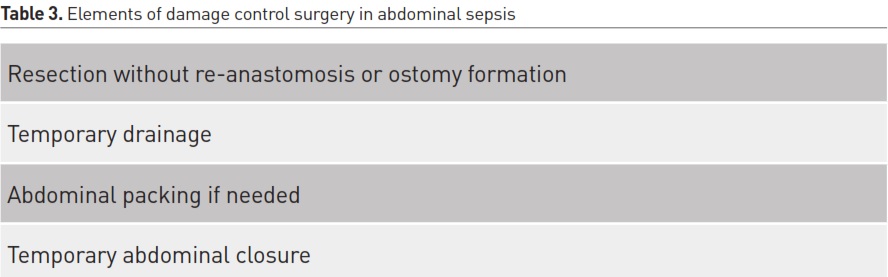
A final comment would be related to the integration of the new concepts in surgery such as damage control, as on many occasions source control is not feasible from the start (Leppäniemi et al. 2015). This concept has been advocated based on experience of damage control laparotomy patients with severe abdominal trauma and it is a limited procedure to control the infection with four practical strategies (Table 3). This approach has also been named rapid source control laparotomy (Becher et al. 2016).
The antibiotic strategy differs depending on the success in achieving source control. Whilst in patients with adequate source control, antibiotics can be used as an adjunct to source control, to prevent dissemination of pathogenic microorganisms during source control procedures and to eradicate residual pathogens after those procedures, in patients with incomplete source control, antibiotics remain as the primary modality for the treatment of the infection.
b) Timing of source control
Delayed source control can be caused by a delay in diagnosis or in intervention after a correct diagnosis has been made; evidently both require different interventions. An accurate and rapid diagnostic process in patients with sepsis or septic shock, which runs in parallel with the resuscitation and other interventions is key for the former; for the latter, often organisational issues such as operating room or interventional radiology availability may be the primary reason.
The rationale for rapid source control is straightforward (Figure 2), yet few guidelines provide clear and evidence-based guidance on the timing of source control in patients with sepsis or septic shock. The SSC guidelines recommend controlling the source of the infection as soon as medically and logistically practical after the diagnosis is made (with the suggestion to do so within a 6-12-hour window after diagnosis) (Rhodes et al. 2017), whereas the English Royal College of Surgeons recommends controlling the source of the infection within 6 hours in patients with sepsis and immediately in patients with septic shock (Royal College of Surgeons of England 2011). The latest update of the Surgical Infection Society guideline on intra-abdominal infections cites 24 hours as the window in which the source needs to be controlled, unless when patients have sepsis or septic shock, when the intervention needs to be undertaken in a more urgent manner (Mazuski et al. 2017).
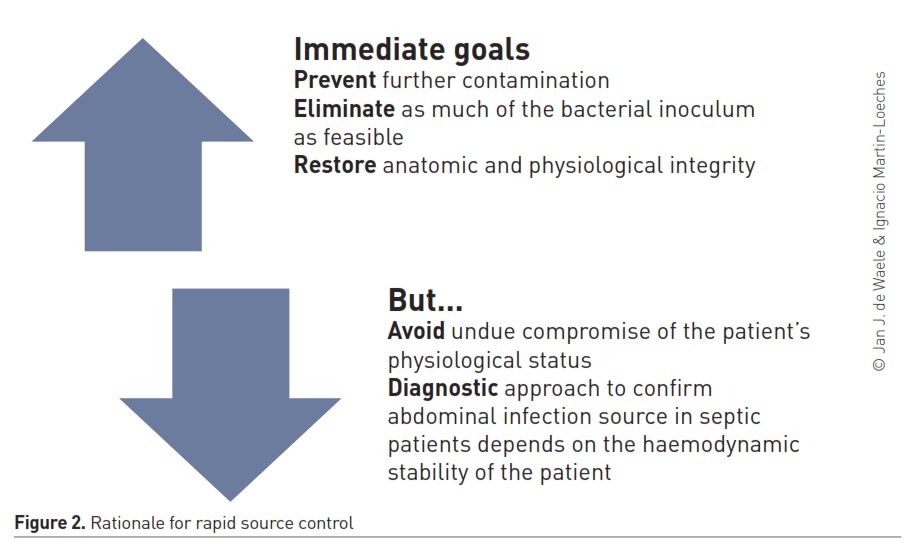
Timing is a critical aspect of source control. A UK study found that in general surgery patients, the median time to surgical source control was 19.8 hours, with no difference in patients with sepsis and septic shock compared to patient without sepsis (as per current definitions) (UK National Surgical Research Collaborative 2017). In a study on patients with complicated diverticulitis of different degrees of severity, one third of source control procedures was delayed for more than 24h (Coccolini et al. 2015). In another large-scale observational study including more than 4500 patients, delay of source control beyond 24h was present in almost half of the survivors, and more than 80% of non-survivors (Sartelli et al. 2015).
In a Korean study on emergency department (ED) patients with septic shock, the majority of patients received source control within 12 hours after ED arrival; in this study the timing of source control did not impact outcome (Shin et al. 2017). A Spanish multicentre study also could not link source control timing (interval between sepsis or septic shock diagnosis to intervention) to worse outcome (Martínez et al. 2017). Again, the majority of the patients’ time to source control was short: median time to source control was 4.6 hours, with 76% of patients receiving source control within 12 hours.
Bloos et al. (2014) found that the median time from onset of severe sepsis or septic shock to source control in a large sample of German ICU patients was 2 hours in survivors and 5.7 hours in non-survivors. Time to source control of more than 6 hours was independently associated with increased mortality (as were age and disease severity) in patients who required source control (Bloos et al. 2014). Time to interventional source control was twice that of surgical source control (6 hours vs. 3 hours).
Bloos et al. (2017) found that source control was significantly related to 28-day mortality and reported a 1% increase in mortality per hour delay of surgical source control. In patients with abdominal sepsis and associated bacteraemia, inadequate and delayed source control was more frequent in non-survivors; inadequate source control and inappropriate antibiotics were independently associated with mortality, but the adjusted odds ratio for inadequate source control was twice as high as for inappropriate antibiotics (Tellor et al. 2015).
Coccolini et al. found delayed source control (>24h) as the sole factor associated with worse outcome in patients with complicated diverticulitis (Coccolini et al. 2017).
Based on the available evidence it could be concluded that the biggest gain in improving outcome is in patients in whom source control is delayed beyond 12-24 hours. It would be very challenging to reduce the timing to source control further in many situations, as also the delay in source control may be explained by other patient factors rather than institutional factors alone.
6. Where are we going from here?
It is clear that source control is an important determinant of outcome, but its exact role in critically ill patients, and the relevance of aspects such as timing and methodology of source control requires more attention. This will help us to provide evidence-based recommendations and may also inform targeted randomised studies on this topic. In the context of severely ill patients, not only the factors related to the intervention but also the patient condition, site of infection and relevant co-morbidities may be highly significant in determining outcomes.
Based on the complexity of severe illness, the range of infections where source control is relevant and the choice of source control interventions at our disposal, it is clear that a generalised approach will be inadequate and a highly personalised, carefully timed approach is the best path to follow.