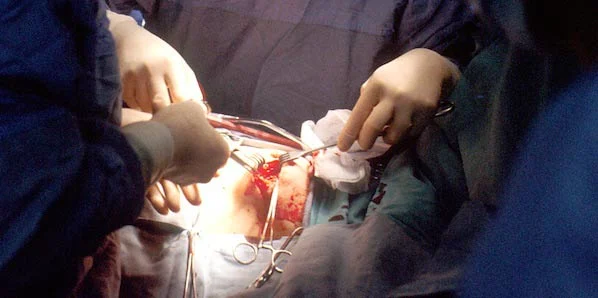
The US FDA has approved an innovative system capable of monitoring surgical blood loss. Created by the mobile health startup business Gauss Surgical, the Triton Fluid Management System will be rolled out in hospitals for use in the operating room as of next month.
Capable of helping anesthesiologists monitor the amount of blood lost in the course of a surgery, this technology combines cloud analytics with an iPad camera. The camera scans blood covered surgical gauze and sends the images to the cloud, where they are images analysed by alrorithms.
The revolutionary app translates this information into real-time estimates of how much blood has been soaked up by the sponges. This data can assist in the decision making process with regards to the need for potential blood transfusions.
Up to now, a patient’s blood loss during surgery was estimated by weighing sponges or by simply guessing the amount, both methods neither accurate nor reliable. Recent studies have consequently suggested that blood transfusions were not always necessary and have exposed receiving patients to complications and risk of infection.
Dr. Aryeh Shander, chief of the Department of Anesthesiology at Englewood Hospital and Medical Center in New Jersey, confirmed that inaccurate blood loss could lead to wrong clinical decision-making. He praised the new technology for providing a more accurate account for blood loss, potentially improving patient outcomes and conserving health resources.
Gauss’ Chief Operating Officer Eric Lindquist highlighted that the app technology had been validated with two clinical studies, demonstrating its superior performance in comparison to the traditional weighing and visual methods.
Having initially submitted an FDA 510(k) approval request back in 2012, the California-based company went trough the 15-months long de novo classification process instead, after it was determined that the app was not substantially equivalent to a predicate device.
The brainchild of graduate of Stanford’s biodesign program Siddarth Satish, and former pro-football player-turned VC-turned medical device entrepreneur Milton McColl, Gauss was founded in 201. They were early participants in the StartX accelerator and followed that up with a $6.2 million Series A round from LifeForce Ventures, Promus Ventures and Taube Investment Partners.
Source: MedCity News Latest Articles
Surgery, Technology, Apps, BloodTransfusion, IT solutions, blood loss, surgeon
The US FDA has approved an innovative system capable of monitoring surgical blood loss. Created by the mobile health startup business Gauss Surgical, the T...