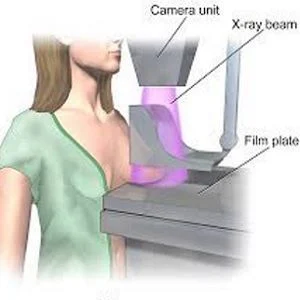
A new UK study shows that screening for and treatment of an early form of breast cancer — ductal carcinoma in situ (DCIS) — is effective in preventing subsequent invasive cancer. Results of the study led by Queen Mary University of London (QMUL) are published in The Lancet Oncology.
Ongoing public debate about the harm caused by mammography screening through overdiagnosis has led to controversy over the value of screen detection and treatment of DCIS. A major question has been the extent to which diagnosis and treatment of DCIS may prevent the occurrence of invasive breast cancer in the future.
For this study, researchers reviewed data on 5,243,658 women aged 50-64 who were screened over a four year period across 84 screening units in the UK's National Health Service Breast Screening Programme in England, Wales and Northern Ireland.
The researchers found that increased prior screen detection of DCIS was associated with a reduction in invasive cancers occurring in the subsequent three-year interval. In 90 percent of the screening units, for every three screen-detected cases of DCIS, there was one fewer case of invasive cancer in the following three years.
See Also - Study: High-Grade DCIS Detection Increases with Age
"There has been controversy over whether ductal carcinoma in situ will ever become invasive cancer. This is the first study from the screening programme which suggests that a substantial proportion of DCIS will become invasive if untreated, and it is therefore worth detecting and treating early," says lead researcher Professor Stephen Duffy from QMUL. "People can be reassured that detection of DCIS in the breast screening programme is benefitting the patients."
The findings suggest that, overall, detection and treatment of DCIS is worthwhile in the prevention of subsequent invasive disease. The authors note, however, that it cannot be known for certain what the outcome would have been for any individual DCIS if it had not been treated.
The average rate of DCIS detected at screening was 1.6 per 1,000 women screened, while the subsequent average rate of invasive cancers found within 36 months of their last screen was 2.9 per 1,000 screened.
The retrospective study was funded by the Department of Health Policy Research Programme and NHS Cancer Screening Programmes.
Source: Queen Mary University of London
Image credit: Wikimedia Commons
Ongoing public debate about the harm caused by mammography screening through overdiagnosis has led to controversy over the value of screen detection and treatment of DCIS. A major question has been the extent to which diagnosis and treatment of DCIS may prevent the occurrence of invasive breast cancer in the future.
For this study, researchers reviewed data on 5,243,658 women aged 50-64 who were screened over a four year period across 84 screening units in the UK's National Health Service Breast Screening Programme in England, Wales and Northern Ireland.
The researchers found that increased prior screen detection of DCIS was associated with a reduction in invasive cancers occurring in the subsequent three-year interval. In 90 percent of the screening units, for every three screen-detected cases of DCIS, there was one fewer case of invasive cancer in the following three years.
See Also - Study: High-Grade DCIS Detection Increases with Age
"There has been controversy over whether ductal carcinoma in situ will ever become invasive cancer. This is the first study from the screening programme which suggests that a substantial proportion of DCIS will become invasive if untreated, and it is therefore worth detecting and treating early," says lead researcher Professor Stephen Duffy from QMUL. "People can be reassured that detection of DCIS in the breast screening programme is benefitting the patients."
The findings suggest that, overall, detection and treatment of DCIS is worthwhile in the prevention of subsequent invasive disease. The authors note, however, that it cannot be known for certain what the outcome would have been for any individual DCIS if it had not been treated.
The average rate of DCIS detected at screening was 1.6 per 1,000 women screened, while the subsequent average rate of invasive cancers found within 36 months of their last screen was 2.9 per 1,000 screened.
The retrospective study was funded by the Department of Health Policy Research Programme and NHS Cancer Screening Programmes.
Source: Queen Mary University of London
Image credit: Wikimedia Commons
References:
Duffy SW et al. (2015) Screen detection of ductal carcinoma in situ and subsequent incidence of invasive interval breast cancers: a retrospective population-based study. The Lancet Oncology. Published
online 04 December 2015. DOI:
http://dx.doi.org/10.1016/S1470-2045(15)00446-5
Latest Articles
healthmanagement, breast cancer, mammography, NHS, screening, early detection
A new UK study shows that screening for and treatment of an early form of breast cancer — ductal carcinoma in situ (DCIS) — is effective in preventing subsequent invasive cancer.