Demand for cardiovascular imaging (CVI) continues to increase due to changes in CV disease epidemiology and ageing of the population. However, reliable statistics on CVI practice in Europe are lacking.
In 2013, the European Association of Cardiovascular Imaging (EACVI) and the European Society of Cardiology conducted a survey to gather as much current information as possible concerning the practice of CVI in Europe. Results of the survey are summarised in this report published in European Heart Journal - Cardiovascular Imaging.
"While practice variations can reflect patient preferences, local demographics, and a host of other factors, availabilities of techniques, educational platform, training requirements, certification guidelines, and reimbursement systems also significantly contribute to the current differences observed between European countries in terms of CVI practice", the authors note in the journal report.
The survey showed that, overall, there were diverse country-specific regulations to perform CVI and a widespread lack of national certification/accreditation in CVI practice.
Methodology
The 2012 leadership of the EACVI agreed upon the importance of obtaining the latest data on the practice of CVI in Europe. Lead authors Patrizio Lancellotti, President of the EACVI 2012–2014, and Edyta Płońska-Gościniak, chair of the European Communities Committee 2012–2014, with the help of the EACVI board and the Chairs of National Societies prepared a questionnaire with the most relevant items to be featured. After a final revision in 2013, the questionnaires were sent out to all the national imaging/echocardiography societies/working groups in Europe.
Representatives from 41 countries returned the questionnaire. Some countries, however, did not have available data for all of the sections of the survey. A random crosscheck of the data quality was conducted with the help of the EACVI board members and some national volunteers. All data were entered into a central database.
All the information was then compiled into the ‘EACVI Highlight Book’ presented during the 2014 EuroEcho-Imaging in Vienna.
Results and Discussion
The rate of CV disease remained a major healthcare burden in Europe, with substantial differences in CV mortality rates between East and North (Figure 1). A national registry on the practice of CVI was present in a very few countries (Albania, Armenia, Denmark, Germany, Greece, Hungary). There was a wide variety of healthcare systems, which involve public or private insurances, including mixed solutions, and co-payments.
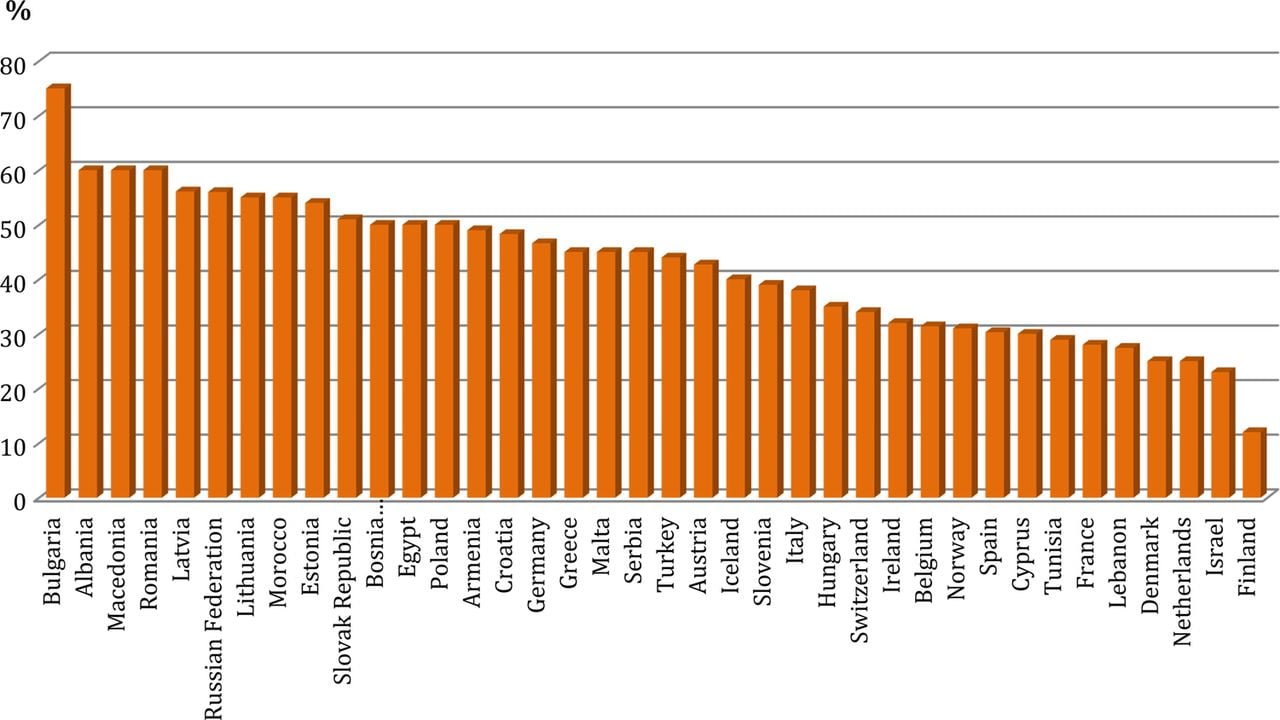
Figure 1.Percentage of death resulting from cardiovascular disease in 2012 (most of the data come from the Ministry of Health).
The performance and reimbursement of CVI strongly depended on the type of imaging technique used. The highest costs were found for positron emission tomography (PET), computed tomography (CT), and cardiovascular magnetic resonance imaging (CMR). Transoesophageal echocardiography (TOE) and stress echocardiography costs were, however, in the highest range in about one-fifth of the countries (>200€). Vascular ultrasound was by far the cheapest examination (<50€).
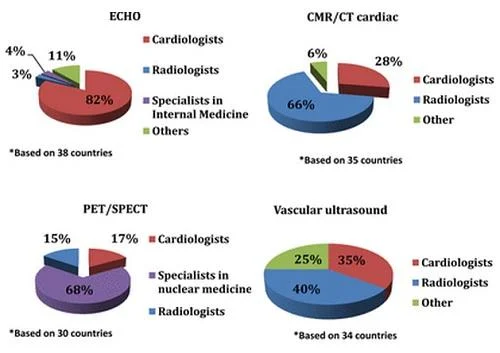
In most countries, echocardiography is in the hands of cardiologists while radiologists mainly performed/reported the CMR and CT examinations. However, medical imaging performance required a specialist license (eg, cardiologist, radiologist, nuclear imaging specialist) in most countries. Unexpectedly, a predefined period of training in CVI during the specialty was absent in one-third of the countries.
A few countries offered official national certification guidelines to perform CVI examinations. Interestingly, the adherence to ESC/EACVI guidelines in CVI was reported in a high number of European countries. Finally, the access to CVI examinations in public healthcare system was marked by a long waiting period in some countries.
Image credit: European Heart Journal - Cardiovascular Imaging
In 2013, the European Association of Cardiovascular Imaging (EACVI) and the European Society of Cardiology conducted a survey to gather as much current information as possible concerning the practice of CVI in Europe. Results of the survey are summarised in this report published in European Heart Journal - Cardiovascular Imaging.
"While practice variations can reflect patient preferences, local demographics, and a host of other factors, availabilities of techniques, educational platform, training requirements, certification guidelines, and reimbursement systems also significantly contribute to the current differences observed between European countries in terms of CVI practice", the authors note in the journal report.
The survey showed that, overall, there were diverse country-specific regulations to perform CVI and a widespread lack of national certification/accreditation in CVI practice.
Methodology
The 2012 leadership of the EACVI agreed upon the importance of obtaining the latest data on the practice of CVI in Europe. Lead authors Patrizio Lancellotti, President of the EACVI 2012–2014, and Edyta Płońska-Gościniak, chair of the European Communities Committee 2012–2014, with the help of the EACVI board and the Chairs of National Societies prepared a questionnaire with the most relevant items to be featured. After a final revision in 2013, the questionnaires were sent out to all the national imaging/echocardiography societies/working groups in Europe.
Representatives from 41 countries returned the questionnaire. Some countries, however, did not have available data for all of the sections of the survey. A random crosscheck of the data quality was conducted with the help of the EACVI board members and some national volunteers. All data were entered into a central database.
All the information was then compiled into the ‘EACVI Highlight Book’ presented during the 2014 EuroEcho-Imaging in Vienna.
Results and Discussion
The rate of CV disease remained a major healthcare burden in Europe, with substantial differences in CV mortality rates between East and North (Figure 1). A national registry on the practice of CVI was present in a very few countries (Albania, Armenia, Denmark, Germany, Greece, Hungary). There was a wide variety of healthcare systems, which involve public or private insurances, including mixed solutions, and co-payments.

Figure 1.Percentage of death resulting from cardiovascular disease in 2012 (most of the data come from the Ministry of Health).
The performance and reimbursement of CVI strongly depended on the type of imaging technique used. The highest costs were found for positron emission tomography (PET), computed tomography (CT), and cardiovascular magnetic resonance imaging (CMR). Transoesophageal echocardiography (TOE) and stress echocardiography costs were, however, in the highest range in about one-fifth of the countries (>200€). Vascular ultrasound was by far the cheapest examination (<50€).
In most countries, echocardiography is in the hands of cardiologists while radiologists mainly performed/reported the CMR and CT examinations. However, medical imaging performance required a specialist license (eg, cardiologist, radiologist, nuclear imaging specialist) in most countries. Unexpectedly, a predefined period of training in CVI during the specialty was absent in one-third of the countries.
A few countries offered official national certification guidelines to perform CVI examinations. Interestingly, the adherence to ESC/EACVI guidelines in CVI was reported in a high number of European countries. Finally, the access to CVI examinations in public healthcare system was marked by a long waiting period in some countries.
Image credit: European Heart Journal - Cardiovascular Imaging
References:
Lancellotti P, Gościniak EP et al. (2015) Cardiovascular imaging practice in Europe: a report from the European Association of Cardiovascular Imaging. European Heart Journal - Cardiovascular Imaging,
5 May 2015. doi: 10.1093/ehjci/jev116
Latest Articles
healthmanagement, cardiovascular imaging, Europe, echocardiography, cardiologists, radiologists
Demand for cardiovascular imaging (CVI) continues to increase due to changes in CV disease epidemiology and ageing of the population.