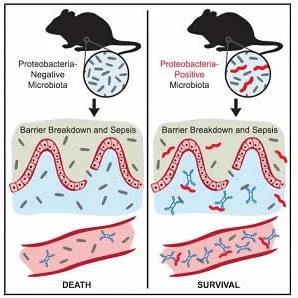
New research published online in the journal Cell Host & Microbe suggests that gut bacteria may help in the fight against sepsis, a life-threatening condition that arises when the body's response to infection causes injury to its own tissues and organs. In the study, mice were given particular microbes which increased blood levels of immunoglobulin A (IgA) antibodies, protecting the mice against polymicrobial sepsis.
There is evidence showing that people with IgA deficiencies are more likely to succumb to sepsis. Also, previous research indicates that immunoglobulin M (IgM) antibodies quickly respond to blood-borne bacteria in sepsis and that gut microbes trigger immunoglobulin G (IgG) antibody responses that can block bacterial infection.
The current study aimed to determine whether gut microbes could trigger IgA responses that protect against sepsis. "We propose that serum IgA and IgG antibodies may play roles similar to the protective role proposed for natural IgM antibodies, with the IgA component providing a non-inflammatory mechanism for keeping invading bacteria in check," says first author Joel Wilmore of the Perelman School of Medicine at the University of Pennsylvania.
To investigate this possibility, Wilmore and colleagues looked at IgA antibodies, which are readily detected in mice and humans but whose role in host protection against sepsis was unknown. The researchers found that exposing mice to a unique but natural microflora that included several members of the Proteobacteria phylum led to increases in IgA levels in the blood. Moreover, shifting the mouse gut to a Proteobacteria-rich microbiota led to IgA-mediated resistance to sepsis in mice.
When the researchers transferred blood lacking IgA into mice with sepsis, all but one animal died within two days. By contrast, mice that received blood enriched in IgA survived much longer. Taken together, the findings suggest that commensal microbes can have a substantial impact on IgA levels in the blood, resulting in protection against bacterial sepsis.
More studies are needed to further dissect the mechanism by which IgA confers protection against sepsis and explore ways to harness the specific properties of these antibodies to develop a treatment that may be applied to human disease. In the meantime, the researchers urge caution against over-interpreting the new findings.
"The study is limited by the fact that the microbiome in every person or animal is unique to some degree, and our study is in the context of the animal facility at the Perelman School of Medicine at the University of Pennsylvania," explains senior author David Allman, also at UPenn's Perelman School of Medicine. "While IgA protected mice in our study, it should not be assumed that IgA could replace standard treatments provided to patients in a clinical setting."
Source: Cell Press
Image Credit: Wilmore et al./Cell Host & Microbe 2018
References:
Wilmore JR, Gaudette BT, Allman D et al. (2018) Commensal Microbes Induce Serum IgA Responses that Protect against Polymicrobial Sepsis. Published online in Cell Host & Microbe, February 22, 2018. DOI: https://doi.org/10.1016/j.chom.2018.01.005
Latest Articles
Sepsis, gut microbes, gut bacteria, immunoglobulin A
New research published online in the journal Cell Host & Microbe suggests that gut bacteria may help in the fight against sepsis.