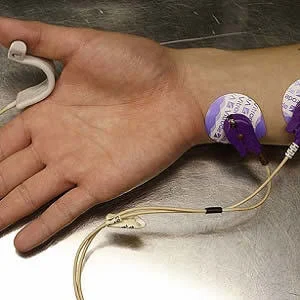
Changes in the delivery of critical care – including protocolised care pathways, increased monitoring techniques and improvements in reducing immobility – have created a modern ICU environment whereby neuromuscular blocking agents (NMBAs) may be administered safely, according to a review published in the journal CHEST.
See Also: Evidence on Monitoring - Capnography, Neuromuscular Blocking
Clinical practice guidelines published in 2002 cautioned against the use of NMBAs, suggesting they only be used when “all other means have been tried without success.” However, new evidence from subsequent randomised trials and studies provides a more optimistic outlook about the use of NMBAs in the ICU.
NMBAs cause skeletal muscle relaxation by blocking the transmission of nerve impulses at the neuromuscular junction. The pharmacokinetics and pharmacodynamics of NMBAs are influenced by the drugs used concomitantly, and other features of critical illness, such as organ dysfunction, and may affect the duration or depth of paralysis. For example, older patients may have reduced drug elimination, while patients with liver or renal dysfunction are at risk for aminosteroid NMBA accumulation.
Furthermore, specific drugs, such as antiepileptics (e.g., phenytoin, carbamazepine), ranitidine, caffeine, and theophylline, may cause resistance to NMBA actions. Other drugs may enhance or prolong NMBA action, including antibiotics (e.g., aminoglycoside, clindamycin, vancomycin), cardiovascular agents (e.g., furosemide, beta blockers, calcium channel blockers), immunosuppressants (e.g., cyclosporine, corticosteroids), lithium, and inhaled and local anaesthetics.
In addition to facilitating endotracheal intubation, clinical guidelines recommend NMBA use in the adult ICU setting as an adjuvant for managing mechanical ventilation, decreasing oxygen consumption, managing increased intracranial pressure (ICP), and treating muscle spasms. NMBAs have also been used to manage increased intra-abdominal pressure (IAP) and for therapeutic hypothermia after out-of-hospital ventricular fibrillation-associated cardiac arrest.
"Although the indications for NMBA use are expanding, their use in the context of intubation and facilitating mechanical ventilation in patients with respiratory failure dominates the clinical picture," the authors say. "Further research is required to optimise NMBA administration in patients with acute respiratory failure to achieve lung-protective ventilation."
The authors also note that uncertainties about the importance of monitoring and the appropriate duration of NMBA administration will need to be addressed.
"However, at present, in the context of adequate sedation and analgesia, monitored NMBA use, whether continuous (< 48 hours) or bolus administration, should be considered safe and efficacious for the small number of clinical indications in critically ill patients for which the current evidence is robust," the authors add.
Source: CHEST
Image Credit: ignis
References:
deBacker, Julian et al. (2017) Neuromuscular Blockade in the 21st Century Management of the Critically Ill Patient. CHEST; doi.org/10.1016/j.chest.2016.10.040
Latest Articles
critical illness, neuromuscular blockade, protocolised care pathways, modern ICU environment
Changes in the delivery of critical care – including protocolised care pathways, increased monitoring techniques and improvements in reducing immobility – have created a modern ICU environment whereby neuromuscular blocking agents (NMBAs) may be administe