A Massachusetts General Hospital (MGH)-led study has shown that a set of characteristics, including differences in gene expression, may indicate which patients recovering from severe burns are at greatest risk for repeat infections. The finding is published online in Annals of Surgery.
"Our approach is the first to enable the prediction of repeat infections days or even weeks before they occur," according to Laurence Rahme, MS, PhD, director of the Molecular Surgical Laboratory in the MGH Department of Surgery and senior author of the study. "All current approaches that use biomarkers identify infections after they have occurred."
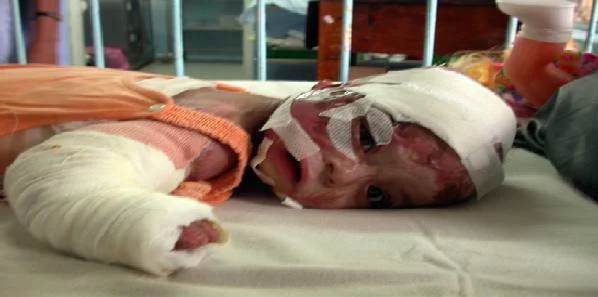
The ability to predict infection would allow more effective prevention and treatment, help stem the emergence of antibiotic-resistant microorganisms, and reduce healthcare costs, Rahme explains. Infection is the leading cause of death in patients with serious burns, and multiple infections stress patients' ability to heal, delaying their recovery. The usual practice is to initiate antibiotic treatment when symptoms of infection develop.
However, the inability to quickly identify the infecting organism can result in patients' receiving powerful medications that target all likely infecting agents. Antibiotics also may be given prior to surgeries or other procedures that could introduce infection. Notably, the ability to select antibiotic treatment based on a patient's individual susceptibility to infection would open a new pathway to improved treatment, Rahme points out.
Aiming to identify factors associated with increased infection risk, Rahme and colleagues analysed data from patients enrolled in the Inflammation and the Host Response to Injury programme, a multi-institutional NIH-sponsored collaboration investigating the body's response to serious traumatic injury. Out of the more than 570 patients studied in the overall project, all of whom had burns over 20 percent or more of their bodies, Rahme et al. focused on 113 adults for whom gene expression data were collected within a week of their injury. They searched for factors — both clinical characteristics such as age and extent of burns, and gene expression patterns — that differed between the 66 patients with two or more infection episodes and the 47 with one or none.
The investigators found that factors currently used to estimate infection risk — patients' age, the extent of burn injury and lung injury from smoke inhalation — could help predict multiple infections. Moreover, they found that a biomarker model based on observed differences in gene expression correctly predicted the infection susceptibility of over 80 percent of patients.
Analysis of risk-associated differences indicated that the expression of genes involved with the immune response, overall metabolism, and epigenetic functions (such as regulating the expression of other genes) was significantly different in patients who experienced multiple infection episodes, implying that impairment of those functions could be behind increased risk of infection.
"Using genomic signatures to understanding why some patients are more susceptible to infection may allow the design of novel, personalised therapies that would support our fight against the antibiotic resistance crisis," says Shuangchun Yan, PhD, of the MGH Department of Surgery and lead author of the current study.
"Our findings also may have very broad applicability to causes of infection other than burns, particularly in the care of military personnel, among whom multidrug-resistant infection of blast and other combat injuries has become a major cause of illness and death. These infections cause grave suffering, severe pain, emotional and physical stress, and considerable financial burdens to patients, their families and to health care systems," explains Rahme, who is also an associate professor of Surgery, Microbiology and Immunobiology at Harvard Medical School.
She adds, "With our focus on innovative ways to counter these infections through the development of biomarker panels and our pioneering work on anti-virulence therapies that block pathogenesis but not cell viability, we hope to open new avenues to treat and prevent infection that do not contribute to antibiotic resistance."
Source: Massachusetts General Hospital
Image Credit: Flickr.com
"Our approach is the first to enable the prediction of repeat infections days or even weeks before they occur," according to Laurence Rahme, MS, PhD, director of the Molecular Surgical Laboratory in the MGH Department of Surgery and senior author of the study. "All current approaches that use biomarkers identify infections after they have occurred."
The ability to predict infection would allow more effective prevention and treatment, help stem the emergence of antibiotic-resistant microorganisms, and reduce healthcare costs, Rahme explains. Infection is the leading cause of death in patients with serious burns, and multiple infections stress patients' ability to heal, delaying their recovery. The usual practice is to initiate antibiotic treatment when symptoms of infection develop.
However, the inability to quickly identify the infecting organism can result in patients' receiving powerful medications that target all likely infecting agents. Antibiotics also may be given prior to surgeries or other procedures that could introduce infection. Notably, the ability to select antibiotic treatment based on a patient's individual susceptibility to infection would open a new pathway to improved treatment, Rahme points out.
Aiming to identify factors associated with increased infection risk, Rahme and colleagues analysed data from patients enrolled in the Inflammation and the Host Response to Injury programme, a multi-institutional NIH-sponsored collaboration investigating the body's response to serious traumatic injury. Out of the more than 570 patients studied in the overall project, all of whom had burns over 20 percent or more of their bodies, Rahme et al. focused on 113 adults for whom gene expression data were collected within a week of their injury. They searched for factors — both clinical characteristics such as age and extent of burns, and gene expression patterns — that differed between the 66 patients with two or more infection episodes and the 47 with one or none.
The investigators found that factors currently used to estimate infection risk — patients' age, the extent of burn injury and lung injury from smoke inhalation — could help predict multiple infections. Moreover, they found that a biomarker model based on observed differences in gene expression correctly predicted the infection susceptibility of over 80 percent of patients.
Analysis of risk-associated differences indicated that the expression of genes involved with the immune response, overall metabolism, and epigenetic functions (such as regulating the expression of other genes) was significantly different in patients who experienced multiple infection episodes, implying that impairment of those functions could be behind increased risk of infection.
"Using genomic signatures to understanding why some patients are more susceptible to infection may allow the design of novel, personalised therapies that would support our fight against the antibiotic resistance crisis," says Shuangchun Yan, PhD, of the MGH Department of Surgery and lead author of the current study.
"Our findings also may have very broad applicability to causes of infection other than burns, particularly in the care of military personnel, among whom multidrug-resistant infection of blast and other combat injuries has become a major cause of illness and death. These infections cause grave suffering, severe pain, emotional and physical stress, and considerable financial burdens to patients, their families and to health care systems," explains Rahme, who is also an associate professor of Surgery, Microbiology and Immunobiology at Harvard Medical School.
She adds, "With our focus on innovative ways to counter these infections through the development of biomarker panels and our pioneering work on anti-virulence therapies that block pathogenesis but not cell viability, we hope to open new avenues to treat and prevent infection that do not contribute to antibiotic resistance."
Source: Massachusetts General Hospital
Image Credit: Flickr.com
Latest Articles
Infections, Biomarkers, antibiotic resistance, burn, gene expression
A Massachusetts General Hospital (MGH)-led study has shown that a set of characteristics, including differences in gene expression, may indicate which pati...