Human factors are significant contributors to drug error. To overcome some of these human factors, we propose standardisation and consolidation of agreed drugs and equipment into a compact pre-packed critical care drug pouch (CCP) for use in non-theatre environments.
Introduction
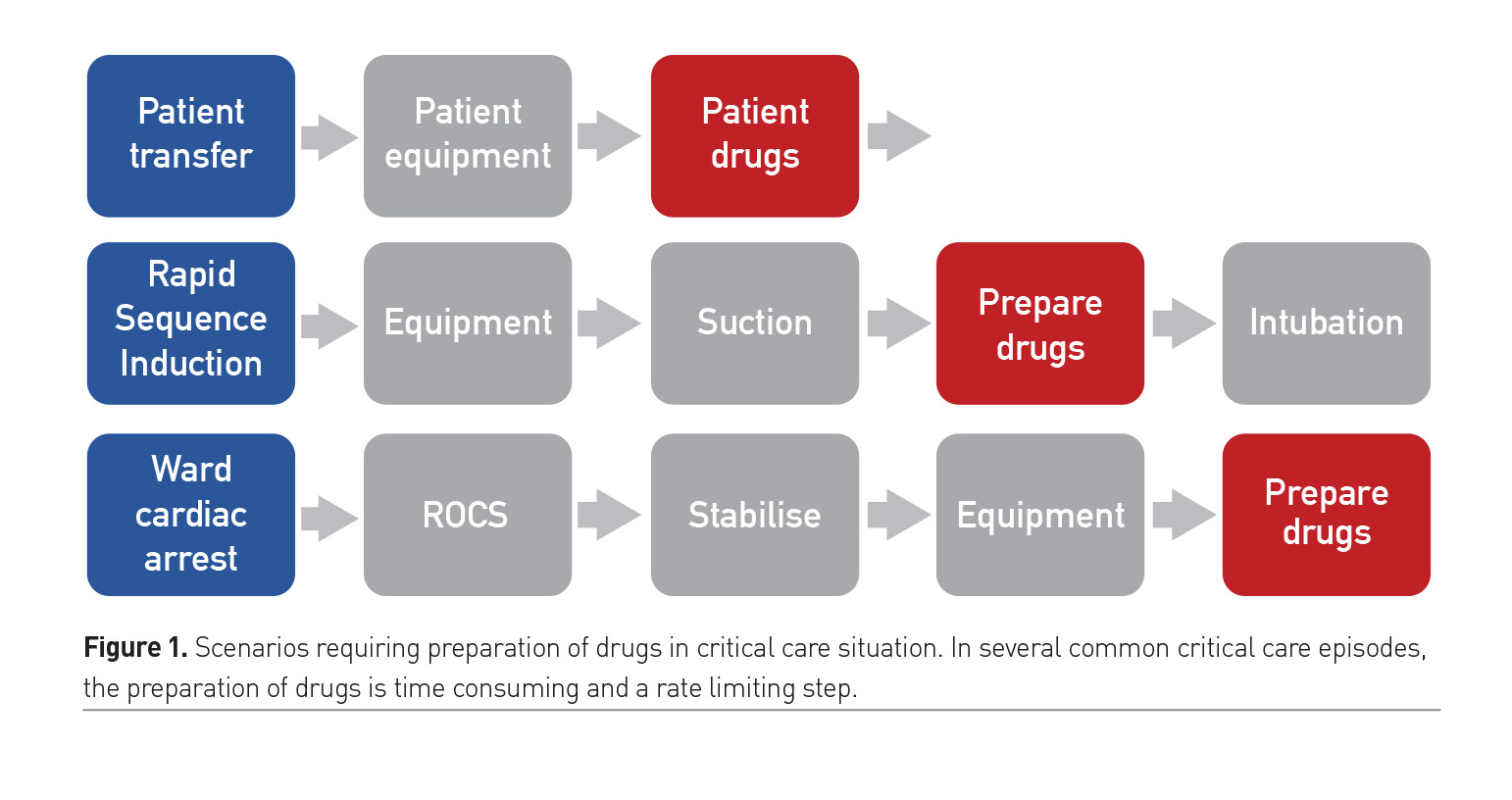
Emergency and sedation drugs availability and preparation represent a challenge in the setting of acute care or resuscitation outside the intensive care unit [ICU] (Glavin 2010; Sakaguchi et al. 2008) (Figure 1). Webster et al (2001) identified an error rate as high as 1 in 133 routine anaesthetics with root causes being syringe swaps (20%) and incorrect doses due to human errors (20%). More stressful environments such as the acute care settings have an even greater risk of preventable errors (i.e. avoidable by any means currently available), reaching 30% (Wilmer et al. 2010).
On the contrary, Highly Reliable Organisations (HROs) have the capacity to operate in hazardous situations with consistent, effective, nearly failure free performance, whilst maintaining optimum output (Roberts 1990; La Porte 1996). HROs encourage the use of checklists and pre-packed equipment to reduce variability and errors caused by human factors. There is substantial material published about the ideal content of cardiac arrest trolleys, difficult airway trolleys, and transfer equipment bags (Henderson et al. 2004). Unfortunately, there is little in the literature about the drugs available and desirable during a critical care transfer or emergency intubations outside the ICU. The aim of this project is to illustrate the process used for defining the content through to implementation of a critical care pre-packed resuscitation pouch (CCP).
Methods
A pre-packed “Critical Care Drug Pouch” has been recently introduced in a large London metropolitan hospital. There was no pre-defined standard against which to compare preceding this project. A modified two-round Delphi and Nominal Group method was used to achieve consensus throughout the project as illustrated in Figure 2 (Hasson et al. 2000; Foth et al. 2016). The project was registered as a quality improvement activity by the Hospital Trust and interviews were anonymously conducted only with the staff working in the ICU. Quality improvement projects in the United Kingdom (UK) do not require ethical approval as soon as these are approved by the audit lead and the division leads as there was no direct patient interaction.

The project was divided into a two-round consensus study beginning with a nominal group session followed by the first round of a Delphi approach using electronic questionnaires. These results were further voted upon by the nominal group and a second round of Delphi performed via simulation and implementation to acquire further feedback and consensus. This nominal group (NG) consisted of a range of 4 experts deemed by seniority and experience: Intensive Care consultant, Practice Improvement Lead Nurse, 2 Senior Anaesthetic registrars (who frequently assisted in, or performed emergency intubations on ICU and within the hospital).
Nominal Group Review 1
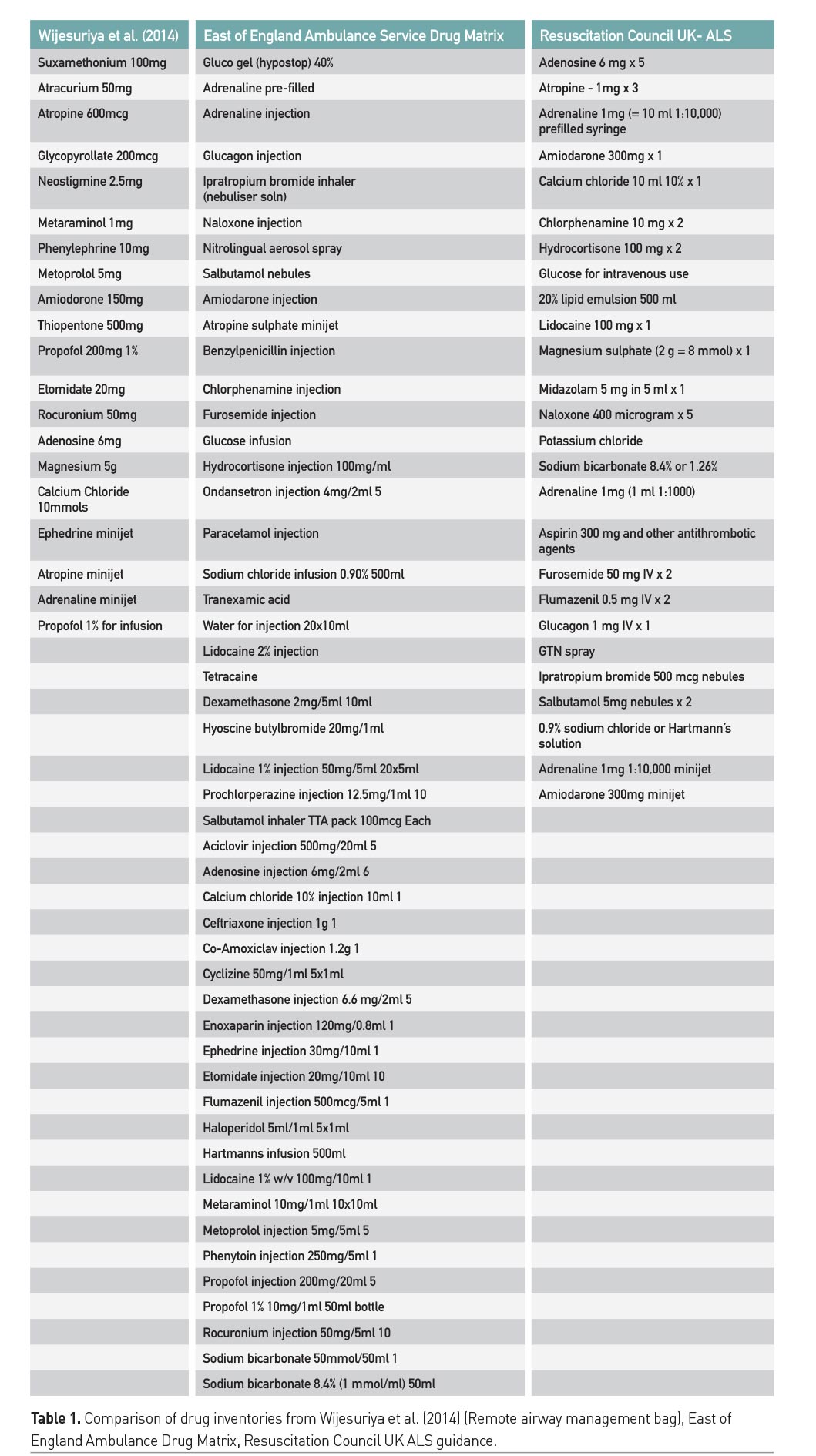
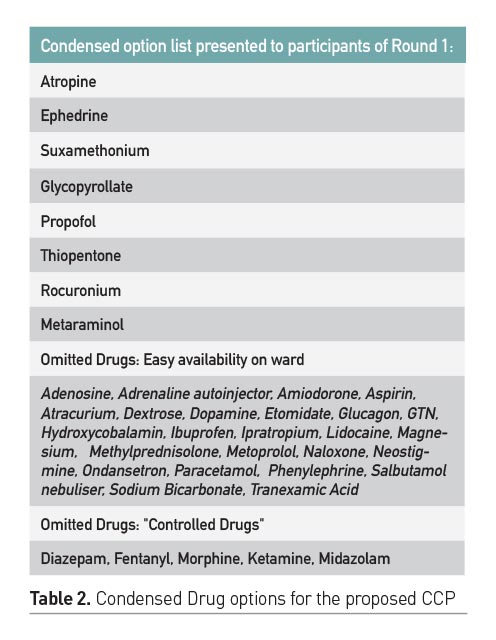
An initial face-to-face brainstorm was performed by the NG after briefing on the project aims over 1 hour. Questions were determined to ask the Delphi group to ascertain feasibility and necessity for the critical care drug pouch. An initial all-inclusive list of drugs was compiled from established models used in advanced life support (ALS) and ambulance service (Wijesuriya and Brand 2014) (eastamb.nhs.uk/policy-libraries/drugs-matrix/274; resus.org.uk/quality-standards/acute-care-equipment-and-drug-lists/; e-lfh.org.uk/e-learning-sessions/rcoa-novice/content/started/theatre.html). The NG excluded “controlled-drugs” such as opiates due to clinical governance constraints. Delphi group respondents were presented with a list of drugs (Table 1) plus a free text option to provide further input (drugs/contents).
Delphi (Round 1): Online Survey
A single-site pre-implementation online survey was conducted following the NG meeting to understand the needs of the end-user groups. The survey link was distributed via email to the doctors and nurses of the ICU team, introducing the aims of the project. Responses were collated over a two-week period with a second round of emails sent after the first week to improve response rate. The survey link contained an in-built validation field to ensure repeated answer by the same user did not occur. Respondents were kept anonymous. The questionnaire was composed of two main parts namely part one questions needed to understand the clinical requirement for the bag and the second to illicit choice of drugs for the CCP as described above. The survey collected also data related to demographic information (role and seniority) and seniority (experience) of respondents.
Nominal Group Review 2
The results of Delphi round 1 were reviewed by the same NG of experts prior to a scheduled 1 hour face-to-face meeting. Pre-defined principles to reduced medication error from Jensen et al. (2014) were applied to determine the contents and form of the prototype CCP. Ideas were shared in a round-robin fashion followed by group discussion. A list of priority drugs was established based on the multiple choice and free text answers from Delphi round 1, and drugs were selected with appropriate support materials.
Delphi (Round 2): Clinical Implementation and Simulation
A prototype pouch bag was prepared in collaboration with the hospital pharmacy containing the drugs and required equipment. Following implementation, feedback was gathered from end-users from two sources:
Questionnaires following clinical use during the first 4 weeks of implementation.
Verbal and written feedback following integration of the CCP into 3 multidisciplinary clinical simulation sessions conducted over a 2 week period.
Written questionnaires were attached to the storage unit of the CCP to permit feedback after each clinical use. Respondents were presented with multiple choice questions on whether or not their experience of drug preparation in critical care was made more efficient, quicker and easier. Free text was also permitted to allow suggestions. Questionnaires remained anonymous. Participants were allowed to respond multiple times given that they may use the bag in different clinical scenarios separated in time and place. This permitted real-time feedback regarding the use of the bag at each use to ascertain safety and functionality issues.
The bag was also incorporated into 3 of the regular multi-disciplinary clinical simulation sessions to troubleshoot problems regarding function and to increase awareness. Feedback was requested verbally and as part of the session feedback in the simulator. This second round of the modified Delphi method allowed the same large end-user group to reach further consensus from real-time experience on useful amendments to be made to the bag.
Feedback Based CCP Improvements
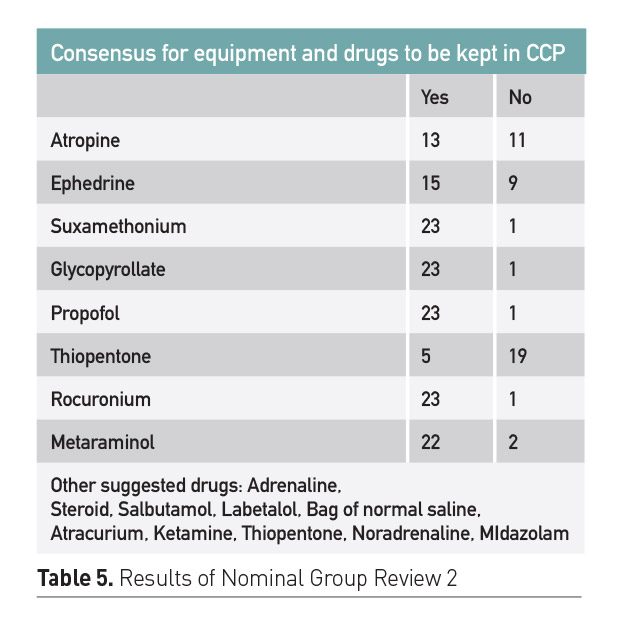
The NG reconvened to review the results of feedback from the clinician questionnaires and from the simulation sessions. Practicality of suggestions were reviewed again using the summary of recommendations from Jensen et al. (2019) to prevent medical errors (Table 5) and restrictions set out by clinical governance and the storage of opiates. This resulted in a final complete CCP which is now in established practice.
Results
Nominal Group Review 1 Results
The initial NG review produced an all-inclusive list of drugs from sources as described. Oral tablets, rectal suppositories and enemas are removed from the list for the sake of brevity. Drugs unrelated to critical care have also been removed. The remaining all-inclusive list is demonstrated in Table 1. By discussion the NG consensus led to removing large bags of intravenous fluids to ensure the CCP would be compact. Drugs which were easily available on the local wards and existing resuscitation trolleys were eliminated, therein nebulisers and antibiotics excluded. We described previously the constraints from clinical governance regarding the storage of controlled drugs such as opiates, ketamine and midazolam would not be permissible in an unsupervised clinical area. Table 2 demonstrates the list of drug options presented to the participants via the web link questionnaire for Delphi round 1.
Delphi Round 1 Results
In the local pre-implementation questionnaire survey, 25 responses were received from 45 recipients following two email notifications containing the web link (3 Consultants, 3 Specialist registrars, 6 Senior House Officers, 12 ICU nurses) (Table 3). Only 1 respondent failed to complete all elements; their responses have been omitted from the analysis. There was a uniform agreement amongst responders that drawing up drugs for emergency rapid sequence induction and transfer was subject to errors and delays, and a significant proportion (32%) felt that the currently available drugs were insufficient for delivery of optimal care. The consensus opinion felt delays were present in finding both equipment and medications with potential patient compromise, and 96% of respondents felt that the drug pouch would improve this experience. Respondents felt that their knowledge of the drugs required for intubation was incomplete. Table 4 demonstrates which drugs from the reduced list respondents felt were vital. Frequent mentions were seen for additional agents in the free text box: adrenaline, atracurium, ketamine, thiopentone, noradrenaline.
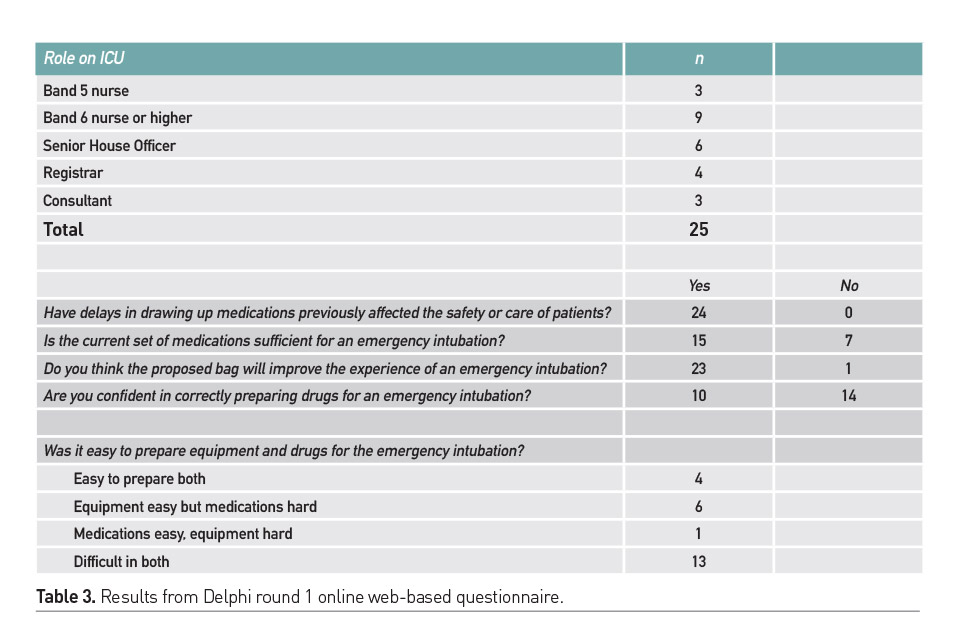
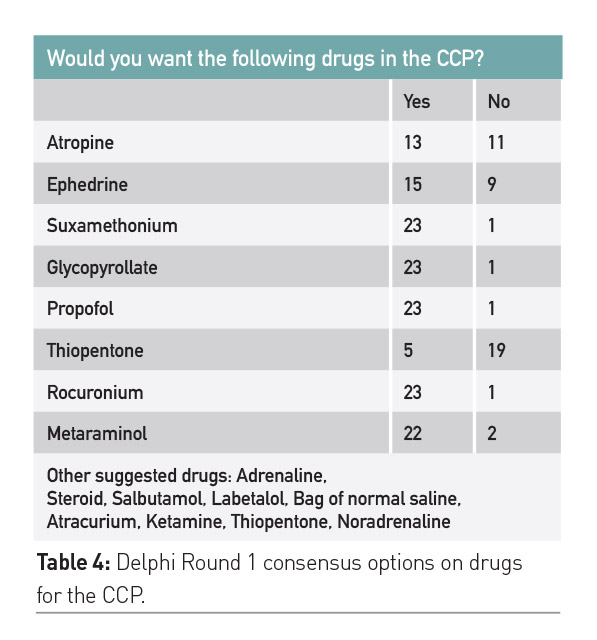
Nominal Group Review 2
The same nominal group reviewed the results of the first Delphi round. A consensus was obtained that the pouch should be minimalistic in size and contents, to permit easy and mobile use, daily check and reduction of waste. It was agreed that it should be in keeping with infection control guidelines and have clinical governance procedures in place to ensure sustained quality and safety.
The resulting pouch contains the minimum syringes (akin to routine anaesthetic practice) and drawing up needles. It was agreed that colour coded syringe labels would be contained in harmony with UK regulations and common anaesthetic practice as per the Association of Anaesthetist of Great Britain and Ireland guidelines to prevent “wrong drug” administration (Woodcock 2014). To facilitate standardisation, two small bags (20cm x 20cm x 7cm) with elasticated vial and mini-jet compartments, were introduced across the 25-bedded unit. The bag also contained a fixed inventory and audit form permitting accountability and ease of daily check. The expert consensus panel agreed from review of the pre-implementation questionnaire and free-text suggestions, that the bag should contain sedatives, hypnotics, neuromuscular blockade, inotropic and chronotropic agents to facilitate emergency intubation on the ICU/remote location or stabilisation of a patient prior to transfer. A diagrammatic/photographic representation of the layout, contents and inventory is provided (Figure 3). The pilot bag contained the following agents as per the consolidation of consensus reached: atropine, ephedrine, glycopyrollate, metaraminol, propofol, rocuronium, suxamethonium, adrenaline, and noradrenaline.
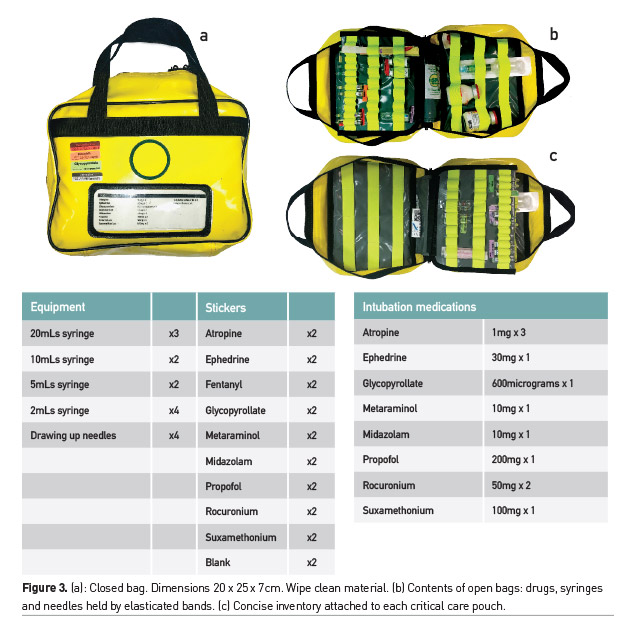
Delphi Round 2 Results
Multidisciplinary clinical simulation sessions provided positive feedback and in which staff felt that the speed of drug preparation was increased with chances of errors reduced, permitting a greater bandwidth for team focus on the clinical situation. Specifically, staff requested a crib sheet detailing the drug dilutions and syringe sizes pertinent to each drug to improve error prevention. Overwhelmingly, respondents felt that a drug bag with a quick reference guide would improve clinical care and that an associated simulation session would be beneficial. Nurses (n=5) and doctors (n=10) who completed the questionnaire following a real clinical episode unanimously felt that the new bag permitted:
a) Greater efficiency in sourcing necessary medications
b) Quicker commencement of the procedure/transfer
c) Contained all medications and equipment required
Free-text answers once again saw suggestions for additional drugs as mentioned previously. In particular midazolam, fentanyl and ketamine were identified. Thiopentone, adrenaline and atracurium were requested by one respondent.
Feedback Based CCP Improvements
Midazolam was added to the bag in subsequent iterations as this was not classified as a controlled drug on the unit. Adrenaline and thiopentone were also added as requested. Ketamine and fentanyl, though deemed desirable by many respondents, would require impractical layers of bureaucracy, reducing the bag’s accessibility in an emergency. Atracurium was deemed unnecessary as this would not permit a safe emergency intubation, given the lack of a reliable reversal agent at the time of writing. As requested, a “dilutions and brief instructional crib sheet” was laminated and attached to each bag.
Discussion
The concept of a “pre-packed drug pouch” is needed for enhancing standardisation and creating a single set containing all necessary drugs and devices. This initiative was started in order to reduce errors and improve patient safety. The preparation of these drugs is time consuming and can often mean the loss of your best team members (critical care nurse, anaesthetic nurse, trainee doctors) whilst they retrieve syringes, needles, drug vials and labels from various locations. The CCP consolidates this search to one place.
Paramedic services and military experience have established practices for delivering acute care to patients in remote, unpredictable and stressful environments (Swinton et al. 2018; Burgess et al. 2018; Woolley et al. 2017). These professions deliver critical care to patients in remote, unpredictable and stressful environments and the presence of pre-packed systematic drug bags provide a portable, systems-based approach. The drug bags are standardised and minimalistic allowing the right drug to be found and prepared quickly and marked clearly. The presence of pre-packed systematic drug bags provides a portable, systems-based approach (nwas.nhs.uk/media/950397/foi-263-web-response.pdf). The drug bags are standardised and minimalistic to easily allow the right drug to be found and drawn up quickly.
“To err is human” and studies have shown that the rate of drug related error is independent of clinician experience (Llewellyn et al. 2009). The solution is cheap, considering that studies show that the additional costs of medication errors can result in an extra $347 (Jiang et al. 2008) to $6647 (Nuckols et al. 2008) per patient secondary to medico-legal costs and increased bed-stay. Similarly Bates et al (1997) found an average increase of hospital costs by $4700 per admission or about $2.7 million annually for a 700 bed hospital as a result of medication errors (Bates et al. 1997). This is in comparison to the total cost of implementation of single bag and its contents: £86.00.
Several techniques to reduce errors attributable to wrong syringe, wrong dilution, wrong dose or wrong medication have been described by Glavin (2010) which emphasise standardisation and minimisation. Many of these are simple behavioural or visual aids and these have been incorporated into the proposed CCP.
Jensen et al. (2004) lays out 12 recommendations to develop systems and associated culture to reduce drug error and evidence based solutions. We share our implementation of these recommendations to reduce errors in preparation and delivery of drugs in our critical care unit (Table 6).
Limitations
Our study has several limitations:
- The Critical Care pouch must be useful and cannot carry controlled drugs such as opioids.
- Clinician’s choice will vary. But a minimalistic principle was agreed and thus a common inventory generated: containing essentials for management of life-threatening emergencies and offer time to find more sophisticated solutions for the clinician’s needs.
Following feedback, a clear learning curve was identified. 37% of staff felt they had some knowledge in drug preparation and 13% felt they were unable to prepare the drugs. Further multidisciplinary simulation training was implemented to overcome this alongside inclusion of the CCP in the induction of new staff and also crib sheets held within the bag to assist unfamiliar staff with the correct syringes and dilutions.
Overall the aim of the project is the reduction of potential errors secondary to human factors. It achieves this through several avenues:
- Avoid the sourcing of drugs, syringes and labels from various locations in a time of emergency- often resulting in the loss of a valuable team member.
- Portable solution to drugs required for intubation, stabilisation and transfer on the ICU or a remote location.
- Clear, concise and colour coded labelling of syringes in harmony with existing anaesthetic practice, with guaranteed legibility.
Conclusion
The implementation of the Critical Care Pouch (CCP) demonstrates a patient-centred, safe, effective and sustainable remedy to reduce staff anxiety during preparation of emergency drugs during common and high stress situations in critical care environments. This successful pilot project was achieved through multiple systematic rounds of large (end user) and small (expert) groups of consensus using the modified Delphi method and nominal group technique. Its clinical success warrants further expansion to a regional level to reach consensus on a standardised bag with potentially greater impact on patient safety by reduction of drug administration errors and streamlined organisation. Progress has been made to create a complement to the already successful Critical Care Equipment Transfer bags established across the Northwest London Critical Care Network (Van Zwanenberg et al. 2016).
Acknowledgements
Gezz Zwanenberg, Northwest London Critical Care; Melissa Dransfield, Consultant Intensivist, Hillingdon Hospital.
For full review of contents of the bag and clinical governance please contact the critical care department at Charing Cross Hospital.
Bates DW, Spell N, Cullen DJ, Burdick E, Laird N, Petersen LA et al.(1997) The costs of adverse drug events in hospitalized patients. Adverse Drug Events Prevention Study Group. JAMA, 277(4):307–11.
Burgess MR, Crewdson K, Lockey DJ, Perkins ZB (2018) Prehospital emergency anaesthesia: an updated survey of UK practice with emphasis on the role of standardisation and checklists. Emerg Med J, 35(9):532–7.
Foth T, Efstathiou N, Vanderspank-Wright B, Ufholz L-A, Dütthorn N, Zimansky M et al. (2016) The use of Delphi and Nominal Group Technique in nursing education: A review. Int J Nurs Stud, 60:112–20.
Glavin RJ (2010) Drug errors: consequences, mechanisms, and avoidance. Br J Anaesth, 105(1):76–82.
Hasson F, Keeney S, McKenna H (2000) Research guidelines for the Delphi survey technique. J Adv Nurs, 32(4):1008–15.
La Porte TR (1996) High Reliability Organizations: Unlikely, Demanding and At Risk. J Contingencies Cris Manag, 4(2):60–71.
Roberts KH (1990) Some Characteristics of One Type of High Reliability Organization. Organ Sci, 1(2):160–76.
Sakaguchi Y, Tokuda K, Yamaguchi K, Irita K (2008) Incidence of anesthesia-related medication errors over a 15-year period in a university hospital. Fukuoka Igaku Zasshi, 99(3):58–66.
Webster CS, Merry AF, Larsson L, McGrath KA, Weller J (2001) The frequency and nature of drug administration error during anaesthesia. Anaesth Intensive Care, 29(5):494–500.
Wilmer A, Louie K, Dodek P, Wong H, Ayas N (2019) Incidence of medication errors and adverse drug events in the ICU: a systematic review. Qual Saf Health Care, 19(5):e7.
For full references, please email [email protected] or visit https://iii.hm/x8c