ICU Management & Practice, Volume 21 - Issue 3, 2021
Introduction
In the intensive care unit (ICU), ultrasound imaging has become increasingly popular for the diagnosis and to guide treatment in critically ill patients (Volpicelli et al. 2020). The use of ultrasound to evaluate the respiratory muscle function (especially the diaphragm) is relatively new, and remains infrequent due to the supposed difficulty in obtaining adequate ultrasound windows, and the common assumption that ultrasound evaluation of the diaphragm would not alter patient management. However, the ultrasound learning curve focused on this muscle is fast, requiring just a short course (Khurana et al. 2018). Diaphragm ultrasound is able to provide serial, non-invasive data at the bedside of critically ill patients (Grosu et al. 2012; Schepens et al. 2015). Various sources of injury (sepsis, mechanical ventilation, myotoxic drugs...) may alter the contractile function of the diaphragm in critically ill patients (Dres et al. 2017a). The prevalence of diaphragmatic dysfunction in intubated patients can exceed 60% on admission, reaching more than 80% in patients requiring prolonged mechanical ventilation with short and long term consequences (Jung et al. 2016; Dres et al. 2017b). The assessment of diaphragm function with ultrasound provides a first step towards improving the detection of diaphragm dysfunction as well as allowing protective and supportive strategies for its management (Supinski et al. 2018). Different ultrasound methods can be used to monitor diaphragm function, and some new interesting approaches have recently been described. The aim of this review is to provide an overview of the principles and current applications of diaphragm ultrasound and to describe innovative ultrasound-based techniques.
Why Use Diaphragm Ultrasound in Intensive Care?
The main goals of diaphragm ultrasound in critically ill patients are:
- Assessment of diaphragm function over time: normal, reduced (weakness), loss (paralysis) = diagnosis of diaphragmatic weakness.
- Assessment of the risk of successful/failed mechanical ventilation weaning.
- Guidance for management of ventilatory assistance settings.
- Monitoring diaphragm thickness over time for prognostic purposes.
The diaphragm is a fundamental muscle of the respiratory system. In physiological condition during spontaneous breathing, the diaphragm contracts and flattens for the inspiratory phase, decreasing thoracic pressure and causing air to flow into the lungs. This active movement called diaphragm excursion (DE), is possible thanks to the three-dimensional anatomy of the diaphragm with a non-contractile centre and muscular fibres departing from this apical centre and attaching to the ribs from T5 to T12. This area of attachment is referred to as the zone of apposition (ZOA). During contraction, the change in muscle fibres length leads to a thickening in diaphragm wall dimension in the ZOA, making the diaphragm move caudally during inspiration. The expiration phase is a passive relaxation of muscle fibres which returns to initial conformation causing air to flow out of the lungs. In case of increased work of breathing (bronchospasm, respiratory distress…) or in case of diaphragm dysfunction, the force generated by the diaphragm may become insufficient. Therefore, accessory muscles may intervene to allow inspiration, compensating for insufficient diaphragm strength. This can result in a paradoxical movement of the diaphragm (inverse to its physiological action: cranial inspiratory excursion). Exhalation can also become active with the abdominal muscles (McCool and Tzelepis 2012).
1.jpg)
Diaphragm dysfunction is an under-estimated phenomenon that can pre-exist upon ICU admission, or develop early during the first 24 hours of ICU course (Jung et al. 2016). Sepsis and mechanical ventilation are the main risk factors (Vassilakopoulos and Petrof 2004; Jung et al. 2014). Prolonged mechanical ventilation (MV) duration, increased incidence of weaning failure, prolonged ICU stay, and long term mortality are associated with diaphragmatic weakness (Dres and Demoule 2018).
Ultrasonography has mainly been used to explore diaphragmatic contractile activity by measuring its excursion (DE), thickening fraction (TFdi) ((end-inspiratory thickness - end-expiratory thickness)/end-expiratory thickness), the velocity of diaphragmatic muscle motion (Tissue Doppler Imaging) and diaphragm mass using its thickness as a surrogate (Schepens et al. 2015; Kim et al. 2011; Dres et al. 2018; Umbrello et al. 2015; Soilmezi et al. 2020).
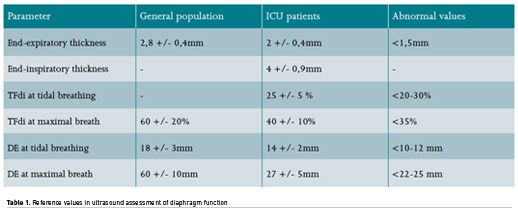
Usually used surrogates for diaphragm weakness are:
- DE of < 10–15 mm during tidal breathing
- (TFdi) < 20% during tidal breathing
The imbalance between load and force-generating capacity of the diaphragm is a main contributor of weaning failure. However, it is important to underline that some patients can be successfully weaned from the ventilator despite having diaphragm weakness. Anyway, diaphragm ultrasound may play an important role to predict weaning success (DiNio et al. 2014). Different parameters under different conditions were associated with weaning success or failure. The more relevant for a clinical perspective are as follows (all reported values are measured under standardised conditions: half-seated position, after 30 minutes of spontaneous breathing trial [SBT] (Tuinman and Jonkman 2020).
- DE: >10-12 mm during a T-piece SBT.
- DE: > 25 mm during a maximal inspiratory effort under T-piece.
- TFdi: > 20-35% during a T-piece SBT or under a pressure support ventilation (PSV) SBT with zero end-expiratory pressure and 7 mmHg PSV.
- Diaphragmatic-rapid shallow breathing index (= respiratory rate/DE): > 1.3 during a T-piece SBT or under a PSV 5 cmH20 and PEEP 5 cmH20 SBT.
- Diaphragmatic time-excursion index (= DE x inspiratory time): An increase in this index between a PSV ventilation and T-piece SBT having a better prognosis than a decrease.
Recently, the diaphragmatic myotrauma concept in which abnormal diaphragm function is associated with either over-assistance (disuse atrophy) or under-assistance myotrauma (muscle overuse leading to inflammation, oedema, and injury) have been developed (Goligher et al. 2019). The authors hypothesised that both ventilator over-assist and ventilator under-assist play an important role in critical illness-associated diaphragm weakness pathophysiology (Goligher et al. 2017). Thus, it may be reasonable to titrate ventilator support for physiological diaphragm, in order to allow a diaphragm protective mechanical ventilation (Goligher et al. 2020). Diaphragm electrical activity and oesophageal pressure swings are often not available and difficult to use in daily practice. Even not specifically studied, TFdi between 15 and 30% during mechanical ventilation has been associated with stable diaphragm muscle thickness and shorter duration of MV (Goligher et al. 2015). Therefore, low TFdi (< 15%) under PSV mode may indicate ventilator over-assist, when high TFdi (> 30-40%) may indicate ventilator under-assist. Clinicians may personalise the level of pressure assist while monitoring other parameters (e.g., respiratory rate and tidal volume).
Diaphragm ultrasound allow prospective monitoring of end-expiratory diaphragm thickness. Although not correlated with diaphragmatic function, this parameter is an easy to obtain marker of severity of critical illness-associated diaphragm weakness (Schepens et al. 2015; Vivier et al. 2019; Sklar et al. 2020). Low baseline diaphragm thickness and significant changes in diaphragm thickness are defined by a change of > 10% over the baseline value. Both decrease and increase of > 10% of thickness are associated with poor outcomes [e.g., longer duration of MV] (Goligher et al. 2017; Goligher et al. 2020; Goligher et al. 2015).
How to Use Diaphragm Ultrasound in Intensive Care?
1. Technical approach
Patient set-up: supine position, half-seated position with a bed inclination at 30-45°.
a) Intercostal approach through the ZOA
- A linear array transducer (10-15 MHz) is positioned perpendicular to the skin between the 8th and 11th intercostal space and between the medial and anterior axillary lines, in the cranio-caudal direction.
- The diaphragm appears at a depth of 2 cm between the shadow cones of the upper and lower ribs.
- Three layered structure: a hypoechoic (dark) central layer corresponding to the diaphragm between 2 hyperechoic (white) lines corresponding to the pleural and peritoneal membrane.
- A hyperechoic (white) line can be visualised within the diaphragmatic muscle corresponding to the aponeurosis between the 2 heads of the diaphragm. It guarantees a reliable cross section of the muscle, and good intra and inter-operator reproducibility.
b) Subcostal approach
- A phased-array or curved-array transducer (2-5 MHz) is positioned below the costal arch at the mid-clavicular line. The probe is angled cranially so as to target a perpendicular cross section of the dome with the ultrasound beam. Liver or spleen can be used as an acoustic window. The liver window is easier to obtain.
- The diaphragm appears as a hyperechoic convex line at 10 to 15 cm depth covering the liver and the spleen that moves toward the probe during inspiration.
2. Measured parameters
a) Diaphragm thickness
- Intercostal approach.
- Measurement in B-mode or in time-motion mode (M-mode).
- Measurement between the internal faces of the pleural and peritoneal membranes, perpendicular to the direction of the muscle fibres.
- Standardised measurement at the end of expiration.
- Reflects the muscular trophicity of the diaphragm.
b) Thickening fraction of the diaphragm (TFdi)
- Intercostal approach.
- Measurement on B-mode or in M-mode.
- Percentage inspiratory increase of diaphragm thickness compared to end-expiratory diaphragm thickness: ([End-inspiratory thickness - End-expiratory thickness]/End-expiratory thickness) x 100.
- Measured during calm (tidal) breathing, or during a maximal inspiratory effort.
- Reflects the contractile activity (function) of the diaphragm. Provides an index of diaphragmatic effort during mechanical ventilation (tidal TFdi) or an index of diaphragmatic function (maximal TFdi).
c) Diaphragm excursion (DE):
- Subcostal approach.
- Measurement in M-mode.
- Activation of the M-mode after visualisation of the hyperechoic diaphragmatic line, M-line placed perpendicular to the direction of the diaphragm movement.
- Measurement of the amplitude of the cranio-caudal movement of the diaphragm during inspiration (towards the probe).
- Measured during calm (tidal) breathing, or during a maximal inspiratory effort.
- Reflects the contractile activity (function) of the diaphragm, in spontaneous ventilation without artificial assistance.
- The excursion of both hemi diaphragms can be compared to identify unilateral weakness or paralysis).
- A negative inspiratory excursion indicates paradoxical diaphragmatic movement and is associated with diaphragmatic paralysis and use of accessory muscles.
When to Use Diaphragm Ultrasound in Intensive Care?
Diaphragm excursion and TFdi are useful to detect diaphragm weakness. One measure alone helps the clinician to easily diagnose the impairment in diaphragm contraction when below known cut-off values. This value at admission is associated with longer duration of MV and poor outcomes (Zamban et al. 2017). There are specific situations where the monitoring with repeated measurements of DE and TFdi as an estimate of the force generated by the diaphragm can be useful.
In patients admitted to emergency department acute exacerbation of chronic obstructive pulmonary disease, severe diaphragm weakness (defined as TFdi <20%) is highly sensitive (85%) and specific (92%) in predicting NIV failure were found to be associated (Kocyigit et al. 2020). In patients admitted to ICU for AECOPD, the same cut-off of TFdi<20% was found in almost 25% of patients. Diaphragm weakness at admission was associated with NIV failure, longer MV, and higher short-term and 90-day mortality (Marchioni et al. 2018). DE can also be used, as a predictor of NIV success in case of improvement (>18mm vs <12mm) after 2 hours of NIV (Cammarota et al. 2019).
Recently, in COVID-19 patients admitted to ICU for acute respiratory failure, a reduced TFdi (<21%) measured during spontaneous breathing with conventional oxygen therapy, was associated with CPAP failure and requirement of invasive ventilation (Corradi et al. 2021). Patients with COVID-19 often present with a silent hypoxaemia which may be especially bewildering to physicians. The lack of dyspnoea in the early stages of the disease is likely related to the absence of increased inspiratory drive due to compensatory mechanisms of hypoxaemia. It seems that COVID-19 patients can then develop patient self-inflicted lung injury even when there are still spontaneously breathing and without any sign of major struggle (Cruces et al. 2020). Assessment of inspiratory effort in spontaneously breathing patients may be difficult without complex monitoring such as oesophageal pressure. TFdi may help by detecting an increase in the generated effort (increase in TFdi) or even by detecting the beginning of exhaustion (decrease in TFdi). Finally TFdi may help to titrate bilevel positive airway pressure support in patients with acute respiratory failure (Laverdure et al. 2019).
In invasively ventilated patients, TFdi can be used to titrate the ventilatory support. It has been postulated that both ventilator over-assist and ventilator under-assist resulting in muscle atrophy and muscle injury, respectively, play an important role in critical illness-associated diaphragm weakness pathophysiology (Goligher et al. 2019). To limit these detrimental consequences, it seems reasonable to titrate ventilator support such that diaphragm effort is within physiological limits. Ultrasound can thus help us to combine the principles of lung protective ventilation (with tidal volume target) and diaphragm protective ventilation (with a TFdi target, as a proxy for effort) (Goligher et al. 2020). Data from Goligher and colleagues demonstrate that a TFdi between 15 and 30% during the first days of mechanical ventilation is associated with stable muscle thickness (Goligher et al. 2015). Accordingly, a low TFdi (< 15%) in a patient on a partially supported ventilatory mode raises the possibility of ventilator over-assist; a high TFdi (>30-40%) may indicate ventilator under-assist. Therefore, pressure support may be titrated targeting physiological levels of TFdi. Diaphragm ultrasound may also help to detect patient ventilator asynchrony which may be associated with worse outcome (Soilemezi et al. 2019).
Another important aspect of diaphragm ultrasound in the ICU is weaning time in which both excursion and thickness provide useful measures for assessing patient effort. TFdi was shown to be strongly correlated with diaphragm strength and support level, while excursion during spontaneous breathing was found to be a reliable measure of an unsuccessful breathing trial (Dubé et al. 2017).
What’s New in Diaphragm Ultrasound in Intensive Care?
Tissue Doppler imaging (TDI) quantifies the velocity of moving structures. Potential applications include assessment of regional diaphragm contractile function at rest and with loading, and measurement of diaphragm relaxation velocity. Diaphragm relaxation abnormalities have been described as a marker of impaired contractility in patients who failed weaning (Soilmezi et al. 2020).
Strain imaging is based on the ability to track ultrasound speckles over time and an excellent feature to quantify motion and deformation of anatomical structures. It was reported that strain and strain rate were highly correlated with transdiaphragmatic pressure (Oppersma et al. 1985).
Shear wave elastography is a technique that allows quantification of the elastic modulus of tissues (Creze et al. 2018). Application of this technique on the diaphragm could be of clinical importance since changes in muscle stiffness may reflect alterations in muscle physiology (e.g., injury, fibrosis). Recently, two studies have evaluated SWE during diaphragmatic contraction and have suggested that SM can be a surrogate for diaphragm contractile activity in healthy volunteers (Chino et al. 2018; Bachasson et al. 2019). Diaphragmatic SWE exploration is feasible in the critically ill population (Flatres et al. 2020). Fossé et al. (2020) investigated changes in SM during diaphragm contraction in mechanically ventilated patients as a potential surrogate to transdiaphragmatic pressure. Therefore, it might offer a new non-invasive method for gauging diaphragm effort.
Conclusion
In the ICU, ultrasound is starting to be acknowledged as a useful tool for studying the diaphragm. Until recently, the monitoring of diaphragmatic function could only be performed with specialised invasive instruments. We are now able to non-invasively assess diaphragmatic function at the bedside and on a daily basis, from ICU admission to discharge. That will surely allow physicians to better understand pathophysiological processes involved in diaphragm injuries in critically ill patients and will lead to improvements in patient outcome.
Conflict of Interest
Dr Aarab has no conflict of interest. Dr De Jong reports receiving consulting fees from Medtronic. Pr. Jaber reports receiving consulting fees from Drager, Medtronic, Baxter, Fresenius-Xenios, and Fisher & Paykel. 
References:
Bachasson D, Dres M, Niérat MC et al. (2019) Diaphragm shear modulus reflects transdiaphragmatic pressure during isovolumetric inspiratory efforts and ventilation against inspiratory loading. J Appl Physiol Bethesda Md, 126:699–707. https://doi.org/10.1152/japplphysiol.01060.2018.
Cammarota G, Sguazzotti I, Zanoni M et al. (2019) al. Diaphragmatic Ultrasound Assessment in Subjects With Acute Hypercapnic Respiratory Failure Admitted to the Emergency Department. Respir Care, 64:1469–77. https://doi.org/10.4187/respcare.06803.
Chino K, Ohya T, Katayama K, Suzuki Y (2018) Diaphragmatic shear modulus at various submaximal inspiratory mouth pressure levels. Respir Physiol Neurobiol, 252–253:52–7. https://doi.org/10.1016/j.resp.2018.03.009.
Corradi F, Vetrugno L, Orso D et al. (2021) Diaphragmatic thickening fraction as a potential predictor of response to continuous positive airway pressure ventilation in Covid-19 pneumonia: A single-center pilot study. Respir Physiol Neurobiol, 284:103585. https://doi.org/10.1016/j.resp.2020.103585.
Creze M, Nordez A, Soubeyrand M et al. (2018) Shear wave sonoelastography of skeletal muscle: basic principles, biomechanical concepts, clinical applications, and future perspectives. Skeletal Radiol, 47:457–71. https://doi.org/10.1007/s00256-017-2843-y.
Cruces P, Retamal J, Hurtado DE et al. (2020) A physiological approach to understand the role of respiratory effort in the progression of lung injury in SARS-CoV-2 infection. Crit Care Lond Engl, 24:494. https://doi.org/10.1186/s13054-020-03197-7.
DiNino E, Gartman EJ, Sethi JM, McCool FD (2014) Diaphragm ultrasound as a predictor of
successful extubation from mechanical ventilation. Thorax, 69:423–7.
https://doi.org/10.1136/thoraxjnl-2013-204111.
Dres M, Goligher EC, Heunks LMA, Brochard LJ (2017) Critical illness-associated diaphragm weakness. Intensive Care Med 2017;43:1441–52. https://doi.org/10.1007/s00134-017-4928-4.
Dres M, Dubé BP, Mayaux J et al. (2017) Coexistence and Impact of Limb Muscle and
Diaphragm Weakness at Time of Liberation from Mechanical Ventilation in Medical Intensive Care Unit Patients. Am J Respir Crit Care Med, 195:57–66.
https://doi.org/10.1164/rccm.201602-0367OC.
Dres M, Demoule A (2018) Diaphragm dysfunction during weaning from mechanical ventilation: an underestimated phenomenon with clinical implications. Crit Care, 22:73.
https://doi.org/10.1186/s13054-018-1992-2.
Dres M, Goligher EC, Dubé B-P et al. (2018) Diaphragm function and weaning from mechanical ventilation: an ultrasound and phrenic nerve stimulation clinical study. Ann Intensive Care, 8:53. https://doi.org/10.1186/s13613-018-0401-y.
Dubé B-P, Dres M, Mayaux J et al. (2017) Ultrasound evaluation of diaphragm function in mechanically ventilated patients: comparison to phrenic stimulation and prognostic implications. Thorax, 72:811–8. https://doi.org/10.1136/thoraxjnl-2016-209459.
Flatres A, Aarab Y, Nougaret S et al. (2020) Real-time shear wave ultrasound elastography: a new tool for the evaluation of diaphragm and limb muscle stiffness in critically ill patients. Crit Care.
Fossé Q, Poulard T, Niérat M-C et al. (2020) Ultrasound shear wave elastography for assessing diaphragm function in mechanically ventilated patients: a breath-by-breath analysis. Crit Care, 24:669. https://doi.org/10.1186/s13054-020-03338-y.
Goligher EC, Brochard LJ, Reid WD et al. (2019) Diaphragmatic myotrauma: a mediator of prolonged ventilation and poor patient outcomes in acute respiratory failure. Lancet Respir Med, 7:90–8. https://doi.org/10.1016/S2213-2600(18)30366-7.
Goligher EC, Dres M, Fan E et al. (2017) Mechanical Ventilation–induced Diaphragm Atrophy Strongly Impacts Clinical Outcomes. Am J Respir Crit Care Med, 197:204–13. https://doi.org/10.1164/rccm.201703-0536OC.
Goligher EC, Dres M, Patel BK et al. (2020) Sahetya SK, Beitler JR, Telias I, et al. Lung- and Diaphragm-Protective Ventilation. Am J Respir Crit Care Med, 202:950–61. https://doi.org/10.1164/rccm.202003-0655CP.
Goligher EC, Fan E, Herridge MS et al. (2015) Evolution of Diaphragm Thickness during Mechanical Ventilation. Impact of Inspiratory Effort. Am J Respir Crit Care Med, 192:1080–8. https://doi.org/10.1164/rccm.201503-0620OC.
Grosu HB, Lee YI, Lee J et al. (2012) Diaphragm muscle thinning in patients who are mechanically ventilated. Chest, 142:1455–60. https://doi.org/10.1378/chest.11-1638.
Jung B, Moury PH, Mahul M et al. (2016) Diaphragmatic dysfunction in patients with ICU-acquired weakness and its impact on extubation failure. Intensive Care Med, 42:853–61. https://doi.org/10.1007/s00134-015-4125-2.
Jung B, Nougaret S, Conseil M et al. (2014) Sepsis Is Associated with a Preferential Diaphragmatic AtrophyA Critically Ill Patient Study Using Tridimensional Computed Tomography. Anesthesiol J Am Soc Anesthesiol, 120:1182–91. https://doi.org/10.1097/ALN.0000000000000201.
Khurana J, Gartner SC, Naik L et al. (2018) Ultrasound Identification of Diaphragm by Novices Using ABCDE Technique. Reg Anesth Pain Med, 43:161–5. https://doi.org/10.1097/AAP.0000000000000718.
Kim WY, Suh HJ, Hong S-B et al. (2011) Diaphragm dysfunction assessed by ultrasonography: Influence on weaning from mechanical ventilation. Crit Care Med, 39:2627. https://doi.org/10.1097/CCM.0b013e3182266408.
Kocyigit H, Gunalp M, Genc S et al. (2020) Diaphragm dysfunction detected with ultrasound to predict noninvasive mechanical ventilation failure: A prospective cohort study. Am J Emerg Med. https://doi.org/10.1016/j.ajem.2020.08.014.
Laverdure F, Genty T, Rezaiguia-Delclaux S et al. (2019) Ultrasound Assessment of Respiratory Workload With High-Flow Nasal Oxygen Versus Other Noninvasive Methods After Chest Surgery. J Cardiothorac Vasc Anesth, 33:3042–7. https://doi.org/10.1053/j.jvca.2019.05.020.
Marchioni A, Castaniere I, Tonelli R et al. (2018) Ultrasound-assessed diaphragmatic impairment is a predictor of outcomes in patients with acute exacerbation of chronic obstructive pulmonary disease undergoing noninvasive ventilation. Crit Care, 22:109. https://doi.org/10.1186/s13054-018-2033-x.
McCool FD, Tzelepis GE (2012) Dysfunction of the diaphragm. N Engl J Med, 366:932–42. https://doi.org/10.1056/NEJMra1007236.
Oppersma E, Hatam N, Doorduin J et al. (2017) Functional assessment of the diaphragm by speckle tracking ultrasound during inspiratory loading. J Appl Physiol Bethesda Md 1985, 123:1063–70. https://doi.org/10.1152/japplphysiol.00095.2017.
Schepens T, Verbrugghe W, Dams K et al. (2015) The course of diaphragm atrophy in ventilated patients assessed with ultrasound: a longitudinal cohort study. Crit Care, 19:422. https://doi.org/10.1186/s13054-015-1141-0.
Sklar MC, Dres M, Fan E et al. (2020) Rubenfeld GD, Scales DC, Herridge MS, et al. Association of Low Baseline Diaphragm Muscle Mass With Prolonged Mechanical Ventilation and Mortality Among Critically Ill Adults. JAMA Netw Open, 3:e1921520. https://doi.org/10.1001/jamanetworkopen.2019.21520.
Soilemezi E, Savvidou S, Sotiriou P et al. (2020) Tissue Doppler Imaging of the Diaphragm in Healthy Subjects and Critically Ill Patients. Am J Respir Crit Care Med. https://doi.org/10.1164/rccm.201912-2341OC.
Soilemezi E, Vasileiou M, Spyridonidou C et al. (2018) Understanding Patient-Ventilator Asynchrony Using Diaphragmatic Ultrasonography. Am J Respir Crit Care Med 2019;200:e27–8. https://doi.org/10.1164/rccm.201901-0054IM.
Supinski GS, Morris PE, Dhar S et al. (2018) Diaphragm Dysfunction in Critical Illness. Chest, 153:1040–51. https://doi.org/10.1016/j.chest.2017.08.1157.
Tuinman PR, Jonkman AH, Dres M et al. (2020) Respiratory muscle ultrasonography: methodology, basic and advanced principles and clinical applications in ICU and ED patients-a narrative review. Intensive Care Med, 46:594–605. https://doi.org/10.1007/s00134-019-05892-8.
Umbrello M, Formenti P, Longhi D et al. (2015) Diaphragm ultrasound as indicator of respiratory effort in critically ill patients undergoing assisted mechanical ventilation: a pilot clinical study. Crit Care, 19:161. https://doi.org/10.1186/s13054-015-0894-9.
Vassilakopoulos T, Petrof BJ (2004) Ventilator-induced diaphragmatic dysfunction. Am J Respir Crit Care Med, 169:336–41. https://doi.org/10.1164/rccm.200304-489CP.
Vivier E, Roussey A, Doroszewski F et al. (2019) Atrophy of Diaphragm and Pectoral Muscles in Critically Ill Patients. Anesthesiol J Am Soc Anesthesiol. https://doi.org/10.1097/ALN.0000000000002737.
Volpicelli G, Mayo P, Rovida S (2020) Focus on ultrasound in intensive care. Intensive Care Med, 46:1258–60. https://doi.org/10.1007/s00134-020-06027-0.
Zambon M, Greco M, Bocchino S et al. (2017) Assessment of diaphragmatic dysfunction in the critically ill patient with ultrasound: a systematic review. Intensive Care Med 2017;43:29–38. https://doi.org/10.1007/s00134-016-4524-z.









