ICU Management & Practice, Volume 16 - Issue 2, 2016
Where Are We Now?
Acute kidney injury (AKI) has been recognised as a major public health problem. It affects >50% of patients in the Intensive Care Unit (ICU) and is associated with serious short- and long-term complications, premature death and high financial healthcare costs (Mehta et al. 2015; Hoste et al. 2015; Lewington et al. 2013). The consensus definition of AKI has emerged from the Risk, Injury, Failure, Loss, End-stage (RIFLE) criteria in 2004 and the AKI Network classification in 2007 to the most recent Kidney Disease Improving Global Outcomes (KDIGO) classification in 2012 (Kidney Disease: Improving Global Outcomes 2012). Although these consensus criteria should be considered a major success towards the standardisationof AKI, they are solely based on serum creatinine and urine output, i.e. two markers which are not kidney-specific and have well-known limitations (Thomas et al. 2015). In particular, serum creatinine can take 24–36 hours to rise after a definite renal insult, may increase following administration of medications that inhibit tubular secretion despite no change in renal function, and is not reliable in patients with sepsis, liver disease, muscle wasting or fluid overload. It also does not provide any information regarding the underlying aetiology. As such, diagnosing AKI can be challenging, especially in critically ill patients. The failure to detect AKI early and the inaccuracy of AKI diagnosis are reasons why the management of AKI is often delayed and attempts to develop specific therapies for AKI have not been successful. There is general agreement that better tools are needed to improve risk assessment, early detection and management of AKI.
Types of Novel AKI Biomarkers
Biomarkers are defined as “characteristics that are objectively measured and evaluated as indicators of normal biologic or pathogenic processes, or pharmacologic responses to a therapeutic intervention" (Biomarkers Definition Working Group 2001). An ideal biomarker for AKI should be accurate, easy to measure at the point of care, correlate with severity of injury, be sensitive to early subclinical renal injury and affordable (Belcher et al. 2011).
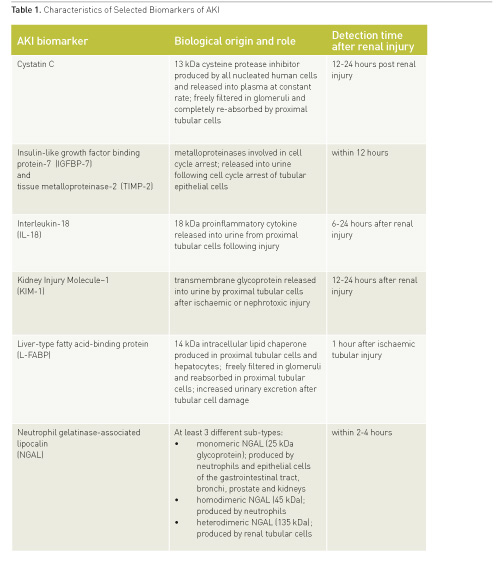
In the last 10 years, numerous different substances in serum and urine have been identified and undergone evaluation as potential biomarkers for AKI (Ostermann et al. 2012; harlton et al. 2014). They vary in their anatomical origin, physiological function, time of release after the onset of renal injury, kinetics and systemic distribution (Table 1). Based on their physiological role, they can be divided into markers of glomerular filtration (i.e. serum creatinine, cystatin C), glomerular integrity (i.e. albuminuria and proteinuria), tubular stress [i.e. insulin-like growth factor binding protein 7 (IGFBP-7), tissue inhibitor metalloproteinase 2 (TIMP-2)], tubular damage [i.e. neutrophil gelatinase-associated lipocalin (NGAL), kidney injury molecule-1 (KIM-1), liver fatty acid-binding protein (L-FAB)] and intra-renal inflammation (i.e. interleukin-18) (Table 1).
Potential Benefits of Novel AKI Biomarkers
Biomarkers for AKI have been studied in various different patient cohorts, ranging from those with a clearly defined renal insult (i.e. coronary angiography or cardiac surgery) to patients presenting to the emergency department or critically ill patients in ICU where the onset of renal injury is less clear. Most investigations have focused on their ability to detect AKI earlier than serum creatinine. The results are most impressive in paediatric cohorts without comorbidities suffering from an illness with a defined onset of AKI, for instance in children after cardiac surgery. In more heterogeneous populations, where the onset of renal injury is not usually known and comorbid factors exist, the performance is more variable and sometimes equivalent to clinical evaluation and standard laboratory measurements (Vanmassenhove et al. 2013). Some biomarkers have also been shown to correlate better with severity of renal injury and important outcomes like mortality and need for renal replacement therapy than serum creatinine.
An important finding has been the identification of patients with elevated biomarker levels but no detectable change in serum creatinine (Haase et al. 2011). These injury biomarkerpositive, creatinine-negative patients appear to have a greater risk of complications, a longer stay in ICU and a higher mortality compared to patients without elevated biomarker level, which implies the existence of a state of ‘subclinical AKI’ where renal injury has occurred but the glomerular function is still preserved. Whether this phase of AKI represents a golden window for effective therapeutic interventions will need to be investigated in future studies.
Finally the discovery of new functional and damage markers has broadened our understanding and provided invaluable insight into the pathophysiological processes involved in AKI from early injury to recovery. For instance, validation studies of the stress biomarkers IGFBP-7 and TIMP2 have highlighted the role of cell cycle and cell cycle arrest in the development and progress of AKI (Gomez et al. 2014).
Limitations of Novel Biomarkers
As indicators of normal biologic and pathogenic processes, the release of biomarkers following the original renal insult is dynamic and temporary. Therefore, the timing of measurement is very important and affects the interpretation of biomarker levels in serum or urine. Studies also vary in their chosen cut-offs for negative and positive predictive events related to AKI, which again contributes to the differences seen between reports. Another important limitation of biomarker research relates to the fact that in most studies the performance of novel biomarkers was compared with that of serum creatinine and oliguria, two markers which are not renal-specific and are considered to be inadequate for the diagnosis of AKI. So far, newer imaging techniques or methods to measure realtime glomerular filtration have not been used for the purpose of evaluating new biomarkers.
Use of Novel AKI Biomarkers in Clinical Practice
Some studies have shown very impressive results and clear indications that novel biomarkers have the potential to transform the way clinicians diagnose and manage patients with AKI. Commercial kits for measurement of cystatin C, NGAL, IGFBP7, and TIMP-2 are now available. However, some biomarkers of AKI, though approved for clinical utilisation, have not been extensively employed in the clinical setting. While reasonably good results are seen in the research setting, their performance in routine clinical practice is influenced by patient case mix, comorbidities, aetiology of AKI, the timing of renal insult, timing of biomarker measurement and the selected thresholds for diagnosis. The scarcity of evidence that biomarkers improve patient outcomes, the prohibitive cost and unavailability of point-of-care testing are additional barriers to their widespread routine use.
In the right setting, the new biomarkers have great potential. However, it is crucial to identify those patients who would benefit most. Some studies advertise the use of biomarkers in situations where the outcome already seems predictable based on clinical evaluation and standard physiological parameters (Vanmassenhove et al. 2013). Clearly, in this case, there is limited added benefit. Biomarkers should be regarded as a complement to routine assessment and be part of a decision tree. Indiscriminate application in patients at low risk of AKI would render the biomarker useless, as well as unnecessarily increase healthcare costs.
Future Roles of AKI Biomarkers
The discovery of new markers of glomerular and tubular function, tubular damage and inflammation allows a much better description and characterisation of AKI than traditional markers of renal function can offer. It is therefore very likely that they will be incorporated into future definitions and classifications of AKI, as proposed at the 10th Acute Dialysis Quality Initiative Consensus (ADQI) Conference (Murray et al. 2014) (Fig. 1).

As indicators of specific pathophysiological processes within the kidney, some of the new biomarkers also offer the opportunity to be used as diagnostic tools to identify the aetiology of AKI. However, a single biomarker is unlikely to be useful. Instead, a panel of functional and damage biomarkers in combination with traditional markers of renal function and clinical judgement will provide best results.
Some of these biomarkers also have the potential to facilitate the development of new drugs by indicating renal injury earlier than conventional methods. Collaborations between international centres and major pharmaceutical companies, the U.S. Food and Drug Administration and the European Medicines Agency have already begun, and rodent urinary and plasma biomarkers have been accepted as surrogates for renal histology for initial evaluation and monitoring of nephrotoxicity in drug development. Finally, there is some hope that some of the novel molecules may not only serve as diagnostic tools but also as potential therapeutic targets for the treatment of AKI.
Conclusion
Numerous novel functional and damage biomarkers for AKI have been discovered and validated. Current evidence supports the concept that they have potential to facilitate the early detection, differential diagnosis and management of AKI in appropriately selected patients. More research including intervention studies based on biomarker results, identification of the most appropriate patient groups and standardization of testing is necessary to incorporate utilization of biomarkers into routine clinical practice.
Conflict of Interest
Marlies Ostermann declares that she has no conflict of interest. Kianoush Kashani declares that he has no conflict of interest.
Abbreviations
AKI acute kidney injury
IGFBP-7 insulin-like growth factor binding protein 7
ICU intensive care unit
KDIGO Kidney Disease Improving Global Outcomes
KIM-1 kidney injury molecule-1
L-FAB liver fatty acid-binding protein
NGAL neutrophil gelatinase-associated lipocalin
RIFLE Risk, Injury, Failure, Loss, End-stage
TIMP2 tissue inhibitor metalloproteinase 2
References:
Belcher JM, Edelstein CL, Parikh CR (2011) Clinical application of biomarkers for acute kidney injury. Am J Kidney Dis, 57(6): 930-40. PubMed ↗
Biomarkers Definitions Working Group (2001) Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin Pharmacol Ther, 69(3): 89-95. PubMed ↗
Charlton JR, Portilla D, Okusa MD (2014) A basic science view of acute kidney injury biomarkers. Nephrol Dial Transplant, 29(7): 1301–11.PubMed ↗
Gomez H, Ince C, De Backer D et al. (2014) A unified theory of sepsis-induced acute kidney injury: inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaption to injury. Shock, 41(1): 3-11.PubMed ↗
Haase M, Devarajan P, Haase-Fielitz A et al. (2011) The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J Am Coll Cardiol, 57(17): 1752–61.PubMed ↗
Hoste EA, Bagshaw SM, Bellomo R et al. (2015) Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med, 41(8): 1411-23. PubMed ↗
Kidney Disease: Improving Global Outcomes (2012) KDIGO clinical practice guideline for acute kidney injury. Kidney Int Supplements, 2(1):1–138. Article ↗
Lewington AJ, Cerdá J, Mehta RL. (2013) Raising awareness of acute kidney injury: a global perspective of a silent killer. Kidney Int, 84(3): 457-67.PubMed ↗
Mehta RL, Cerdá J2, Burdmann EA et al. (2015) International Society of Nephrology's 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): a human rights case for nephrology. Lancet, 385(9987): 2616-43.PubMed ↗
Murray PT, Mehta RL, Shaw A et al. (2014) Potential use of biomarkers in acute kidney injury: report and summary of recommendations from the 10th Acute Dialysis Quality Initiative consensus conference. Kidney Int, 85(3): 513-21.PubMed ↗
Ostermann M, Philips BJ, Forni LG (2012) Clinical review: biomarkers of acute kidney injury: where are we now? Crit Care, 16(5): 233. PubMed ↗
Thomas ME, Blaine C, Dawnay A et al. (2015) The definition of acute kidney injury and its use in practice. Kidney Int, 87(1): 62-73. PubMed ↗
Vanmassenhove M, Vanholder R, Nagler E et al. (2013) Urinary and serum biomarkers for the diagnosis of acute kidney injury: an in-depth review of the literature. Nephrol Dial Transplant, 28(2): 254-73. PubMed ↗








