Why and how we need to assess patient centricity.
Focusing on the issues and opportunities in quantifying patient engagement in pharma.
It is an exciting time to be a member of the ‘patient economy’. For close to a decade pharma has worked relentlessly – sometimes seeming to run in circles – chasing the ‘perfect’ infrastructure to implement a company shift to a patient-centric focus. Now, whether the system is ‘C-level’, grassroots or truly embedded in a multistakeholder model across an organisation, the gap between wanting and doing is closed and, like it or loathe it, every pharma and biotech company is now firmly committed to at least some sort of patient-centred initiative. As such, we are now able to demonstrate what ‘good engagement’ looks like – to deliver return on investment (ROI) data that substantiate and ratify that patient centricity delivers better services for patients and a more sustainable business promise for industry. We can benchmark, assess and audit the success or redundancy of the patient-centred initiatives we choose to undertake.
Having disentangled ourselves from ethical ruminations – ‘should we?’ ‘could we?’ – the momentum now underpins an attitude of ‘can we?’ and if so, when? The sweet spot of better outcomes for patients and more business success for pharma lies in the implementation of systematic inclusion of the patient voice at the translational level; garnering insights from patients about living with a condition as early as possible and anchored within the Medical Affairs department but permeated across the entire product lifecycle. The huge challenge is bringing together expertise and knowledge within companies that are usually multi-therapeutic, made up of thousands of employees, multiple geographies and differing cultural expectations; a model not wildly dissimilar from many given patient populations. Pharma companies have built resources, strategies, approaches and even pipelines more in tune with what patients are looking for. The next step therefore is to measure, share and innovate – to keep up the momentum that took so long to build.
Pharma still has some way to go
There will always be a disconnect between what patients need and what pharma is able to deliver within the highly-regulated world of drug development, where products are investigational but patients are desperate for answers, solutions and even ‘cures’. Perhaps we should accept and embrace the differences as arguably they drive collaboration and innovation. Recently published results from Ipsos/The Aurora Project show that whilst 72% of biopharmaceutical and medical device company employees agreed with the statement, ‘My company communicates with care and compassion, transparent and unbiased information on diseases, treatment options, and available resources’, only 32% of the patients surveyed agreed – even when considering the company that they interact with the most (Ipsos & The Aurora Project 2018). There is also divergence in expectations between doctors and patients – again this might be ‘normal’ as each party enters a dialogue with a different need. Patients seek efficacy first, but pharma and doctors are risk averse and seek safety as a priority. Here, therefore, is the opportunity to provide patient services that have a therapeutic affordance in their own way. It is universally accepted that people come to pharma for holistic solutions applicable to their entire lives and the very nature of the R&D and clinical trial process turns them into patients. Furthermore, language or poor health literacy continues to be a barrier that healthcare professionals must hurdle to reach a more impactful relationship with (NHS 2018). In essence, a patient wants to be seen as a person, not as a number; accordingly the increasing popularity of physician, pharma and even patient rating sites, co-collaboration platforms and research hubs continue to develop apace.
Challenges in implementing and measuring the success of patient engagement strategies
There is a risk of being overwhelmed by an urgency for any patient initiative ahead of one that delivers a quantifiable benefit. Quantifying patient engagement is difficult for a number of reasons. There is a lack of shared guidelines and instruments, meaning that even identifying a baseline is near impossible. Evaluating the direct impact of engagement initiatives on patient outcomes requires time and therefore a long-term rather than short-term focus. For this reason, we primarily see patient engagement strategies run by commercial marketing sectors, and research and development (R&D) departments are often forgotten about to focus on basic science. However, it is imperative that the patient voice is heard from the very beginning of drug development, not just when it is time to market the finished product.
It is also tricky to distinguish between those who hold responsibility for patient engagement and those who are accountable; setting up patient affairs departments or appointing chief patient officers may seem like a good idea, but there is an argument that doing so reduces the personal responsibility that other employees feel in being patient-centric.
Approaches for quantifying patient engagement
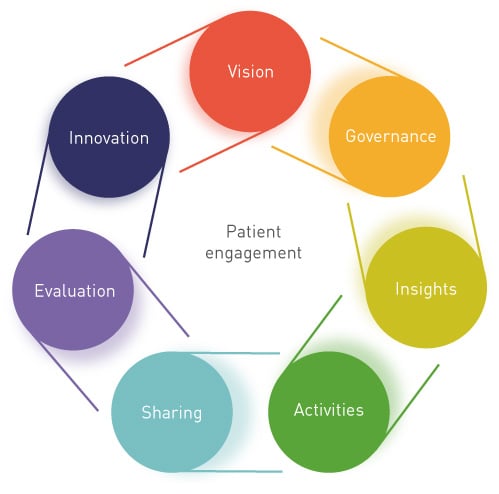
At NexGen Healthcare Communications we have developed a patient engagement audit called ‘HaloGEN’ that is based on our collective experience as pharma executives working with more than 100 organisations – pharma, patient advocacy groups (PAGs), non-governmental organisations (NGOs), academia and individual patient opinion leaders (POLs) that seeks to be both broad and deep in reach and impact. By taking the temperature of patient engagement across all departments to seek to consolidate ‘islands of excellence’, we are able to provide a given company with both a measure of work done, and a blueprint of priorities that will deliver greater return on engagement (ROE). HaloGEN comprises seven interlinking domains that are proven essentials to build a competent attitudinal and practical shift for demonstrable patient centricity. Those sections deliberately vary in depth of dissection according to the ‘feel’ and specific obstacles an organisation might be facing but always include the core elements: Vision, Governance, Insights, Activities, Sharing, Evaluation and Innovation.
Vision is the opening ‘scene’ of the audit as it sets a clear precedent both for the individual participating in the interview and a macro message that HaloGEN is intended to deliver a ‘what next’ set of results – to keep pharma execs rolling their sleeves up – not putting their feet up. Importantly, this also liberates participants from the expectation that they must know every aspect of patient advocacy and lobbying, which is and should remain a specialist function within a company – the HaloGEN audit is a business assessment and improvement tool. Once the Vision is set, the remaining sections are a natural segue and when an employee enters the HaloGEN dialogue, they rapidly release all that is known and desired as they work through the sections.
As experienced practitioners who have run hundreds of individual interviews, we have yet to experience a diminishing of interest as the interview progresses; more often than not, we vastly overrun the allocated time. There is impressive commitment across all departments to contribute to this and to get it right. Governance follows as how much a company can do often relies on their internal processes and the resources dedicated to it. Insights are needed for a company to understand their patients, their needs and opinions, before organising activities to involve patients directly in their business. Once the organisation has gathered knowledge and expertise, it needs to share these both internally and externally, and then evaluate the impact and success on its business, particularly as it relates to patients. Finally, the company needs to continually innovate to stay ahead and enhance patient products and services.
Companies that excel in all domains are poised for success in an environment where patients are becoming more knowledgeable and demanding more of a say in their own treatment. By providing more patient-centred care, we can drive better health and outcomes and empower patients to be active in their own care, ultimately feeding back into a more motivated and efficient workforce and an improved financial picture.
Having disentangled ourselves from ethical pharma ruminations – ‘should we?’ ‘could we?’ – the momentum now underpins an attitude of ‘can we?’
Key Points
- Patient engagement is a buzzword that is thrown around a lot, but less frequently acted on with truly positive effects
- There are many challenges, both in implementing and in measuring the impact of patient engagement strategies
- Surveying the right people can reveal the success of patient engagement strategies across several key domains










