Αdvances in technology and the advent of precision medicine point to a move away from ‘one size fits all’ breast cancer screening.
Breast cancer is the most frequently diagnosed cause of death from cancer in women worldwide and the second leading cause of death from cancer in women in developed countries (Ferlay et al. 2014a; 2014b). Breast cancer mortality rates have decreased due to mammographic screening, and hence earlier detection of cancers, including small noninvasive cancers, and advances in treatment. However, advances in technology and the advent of precision medicine point to a move away from ‘one size fits all’ breast cancer screening.
Breast cancer screening: U.S. and Europe
The screening environments in the U.S. and in Europe are very different. In the U.S., screening is opportunistic, on the recommendation of a physician, or initiated by the woman. Annual screening is commonplace for women aged 40 years and above and recommended by some authorities (Williams et al. 2015). Screening with digital breast tomosynthesis, or 3D mammography, is increasingly popular. Generally mammograms undergo a single reading by a radiologist, and insurance coverage is variable for DBT.
Many European countries have a national screening programme, which generally offer screening every two years to women aged 50 and above. In the UK, double readings by a radiologist are standard. DBT is not as prevalent outside the U.S. yet, and insurance coverage is minimal compared to that in the U.S. However, there are ongoing clinical trials in Europe evaluating DBT in the context of a screening programme in order to assess its cost-effectiveness and performance PROSPECTS trial medphys.royalsurrey.nhs.uk/prospects, the recently reported Reggio Emilia Tomosynthesis Randomized Trial (Pattacini et al. 2018) and the Tomosynthesis Trial in Bergen (TOBE), Norway - clinicaltrials.gov/ct2/show/NCT02835625).
Is ‘one size fits all’ mammographic screening still valid?
Mammography has been the mainstay of breast cancer screening since the 1990s. Although it is the only imaging modality shown to reduce mortality caused by breast cancer by 25-30% in multiple large randomised clinical trials, it has limitations. Clinical trials have found false negative rates of 4-34%, 10-12% call backs for additional imaging studies, and a 20% incidence rate of interval cancers after a negative mammogram and physical examination. Such limitations are most common in younger women and women with dense breasts.
Why should women be informed about breast density?
Dense breast tissue masks breast cancer, so by the time cancer is detected it may have grown to a stage at which more aggressive treatment is required. Women with dense breasts are four to six times more likely to develop breast cancer (Boyd et al. 2007). In an analysis of clinical risk factors for breast cancer in more than 18,000 women with invasive ductal carcinoma (IDC) or ductal carcinoma in situ (DCIS) matched with similar numbers of women without breast cancer, breast density was the most prevalent risk factor in pre- and postmenopausal women: 39.3% (95% CI, 36.6-42%) premenopausal; 26.2% (95% CI, 24.4%-28%) postmenopausal (Engmann et al. 2016).
Screening controversies
There are mixed messages on the efficacy of breast cancer screening—from varying guidelines, articles in the lay press and one large study, the Canadian National Breast Screening study, which cast doubt on the value of annual screening mammograms for women aged 40–59 (Miller et al. 2014). Concerns centre on over-diagnosis and over-treatment as well as radiation dose exposure from more frequent screening (although the latter can be avoided by use of synthesised 2D images obtained using DBT).
Emerging screening modalities
Several modalities are used for additional examination in women where a suspicious lesion has been detected with mammography or as initial examinations in women with an increased risk of breast cancer. Their use as a mass screening modality is still to be proven, however.
Ultrasonography
Handheld US (HHUS) is widely available and well tolerated, and a large multicentre trial demonstrated that screening with HHUS finds significantly more early-stage breast cancers than screening with mammography alone, providing a cancer detection rate of 0.3%-0.5% (Berg et al. 2008; Buchberger et al. 2000; Kolb et al. 2002). However, HHUS has several limitations, including operator dependence and lack of a standardised technique (Berg et al. 2006). In addition, bilateral whole-breast screening using HHUS is time-consuming and has a high number of false positives.
Automated breast ultrasound (ABUS) has several advantages over HHUS, including higher reproducibility, less operator dependence, and less time required for image acquisition (Kaplan 2014). In addition, ABUS reduces the need for trained operators and it provides both a coronal view and a relatively large field of view. Recent studies have reported that ABUS is promising in US screening for women with dense breasts and can potentially replace handheld second-look US in a preoperative setting (Kelly K et al. 2010; Golatta M et al. 2015; Lander M, Tabar L 2011; The SOMO-INSIGHT study 2013).
Abbreviated breast MRI
Breast MRI is not a cost-effective modality for screening women at intermediate risk, including those with dense breast tissue as the only risk. Therefore abbreviated MRI protocols (under 10 minutes) have been developed as a way of achieving efficiency and rapid throughput. It is a less expensive option that offers an exceptional supplemental screening modality. However, MRI scanner availability means that access may be an issue.
Mango and colleagues (2015) found that one pre and post-contrast T1W sequence may be adequate for detecting breast cancer. The Abbreviated Breast MRI and Digital Tomosynthesis Mammography in Screening Women With Dense Breasts randomised clinical trial (NCT02933489 - clinicaltrials.gov/ct2/show/NCT02933489) will report on which modality is more effective in cancer detection.
Contrast-enhanced digital mammography
Contrast-enhanced digital mammography (CEDM) is an alternative to abbreviated breast MRI. CEDM can highlight areas that may not be detected with standard 2D full-field digital mammography. As with breast MRI, the administration of intravenous contrast in CEDM highlights areas of increased blood flow that may be associated with tumour growth. CEDM has consistently been demonstrated to be more sensitive than standard mammography for the detection of breast cancer. The installed mammography base makes CEDM an attractive option, and the technology is easily integrated in the clinic.
Can genomics help?
Ongoing global genome characterisation efforts are revolutionising our knowledge of cancer genomics, tumour biology and treatment methods. Information collected from a number of studies on driver cancer gene alterations—mutations, copy number alterations, translocations, and/or chromosomal rearrangements—can be leveraged to develop a cohesive framework for individualised cancer treatment. Genetic information can give an insight into tumour detection and individual risk and costs have come down considerably.
A 2001 study by Sørlie et al. classified breast carcinomas based on variations in gene expression patterns derived from cDNA microarrays and correlated tumour characteristics to clinical outcome. They concluded that classification of tumours based on gene expression patterns can be used as a prognostic marker with respect to overall and relapse-free survival in a subset of patients that had received uniform therapy.
Early cancer genomics detection can indicate treatment approach. More heterogeneous tumours may be more likely to contain treatment-resistant subclones. Resistant subclones survive and propagate to reform a heterogeneous tumour.
When clonal expansion occurs is relevant to selection of a screening technique. If there is early clonal expansion, screening is needed to detect when a tumour is extremely small, whereas in late clonal expansion it may not matter when the tumour is detected as a single clone is easier to treat. Different cancers present varying rates of disease dynamics and progression, with the size at which cancer causes symptoms—and death—varying according to type. We need to be smarter in our screening recommendations and detect before clonal expansion.
Liquid screening for early cancer detection
Liquid screening is a promising technique that looks for cell-free nucleic acid (cfNA), which is released from tumour tissues through secretion, necrosis and mostly apoptosis. A cfNA final platform is likely to include SNP array plus whole genome, whole exome or targeted sequencing. Memorial Sloan Kettering is running the MSK Discovery Study: Use of cfNA To Distinguish Between Benign and Malignant BI-RADS 4 Radiographic Lesions (NCT03372902-clinicaltrials.gov/ct2/show/NCT03372902). The STRIVE Study: Breast Cancer Screening Cohort for the Development of Assays for Early Cancer Detection is also based in the U.S. and will collect blood samples from participants within 28 days of their screening mammogram. There are some challenges in ctNA assays, as detection rates vary.
A 2017 study from MSKCC/GRAIL assessed the use of cell-free nucleic acid (cfNA) for early detection using GRAIL’s high-intensity sequencing approach (Razavi et al. 2017). It demonstrated that liquid screening can be a key approach to detecting cancer. Of 161 patients studied, 124 (39 breast cancer, 41 lung cancer, 44 prostate cancer) had sequencing results available for analysis from both blood and tumour tissue samples. In 89% of patients, at least one of the mutations detected in the tumour tissue was also detected in the blood (97% in patients with breast cancer, 85% in those with lung cancer and 84% in those with prostate cancer). When evaluating all genetic variations, including those present at high levels in tumour tissue (clonal) as well as those at low levels (sub-clonal), 627 of 864 mutations (73%) detected in tumour tissue were also detected in patients’ blood. Most actionable mutations (those with associated targeted treatments) detected in tumour tissue were also detected in cfDNA (54 of 71 actionable mutations detected in tumour tissue were also detected in cfDNA; 76%). In addition, some cancer-related mutations that were detected in the bloodstream had been unidentified in the tumour tissue.
Molecular imaging and ctDNA will enable detection of tumours at the cell and receptor level. Optimal detection of a tumour requires molecular imaging with ctDNA, combined with MRI CEDM and mammography DBT.
Personalised screening
The goals of personalised screening are early identification of women at high risk for breast cancer. Identifying women at high risk includes genetic testing, single nucleotide polymorphisms (SNPs) from saliva, use of the Tyrer Cuzick (TC) risk model and breast density measurement. The TC model has performed the best at breast cancer risk estimation (Amir et al. 2003), as it allows for the presence of multiple genes of differing penetrance. Risk should be assessed at regular intervals, with 5/10-year risk preferred to lifetime risk evaluation.
Single nucleotide polymorphism to detect genetic variation
SNPs detect the most common genetic variation among people. Each SNP is different in a single DNA nucleotide. They occur once in every 300 nucleotides on average, which means there are roughly 10 million SNPs in the human genome. Some are associated with disease and they are useful for comparing regions of the genome between cohorts in genome-wide association studies.
A study by Cuzick et al. in 2017 examined the impact of a panel of 88 SNPs on the risk of breast cancer in high-risk women, looking at the results from two randomised tamoxifen prevention trials. Conditional logistic regression and matched concordance indices (mC) were used to measure the performance of SNP88 alone and with other breast cancer risk factors assessed using the TC model. Results show that SNP88 was predictive of breast cancer risk overall (odds ratio 1.37; 95% CI, 1.14 to 1.66; mC, 0.55), but mainly for oestrogen receptor-positive disease (OR 1.44; 95%} CI, 1.16 to 1.79) versus oestrogen receptor-negative disease. The predictive power was similar to the TC model (OR 1.45; 95% CI, 1.21 to 1.73), but SNP88 was independent of TC and when combined multiplicatively, a substantial improvement was seen (OR 1.64; 95%} CI, 1.36 to 1.97; mC, 0.60). The researchers concluded that a polygenic risk score can refine risk from the TC or similar models, which is important for identifying women who need heightened screening.
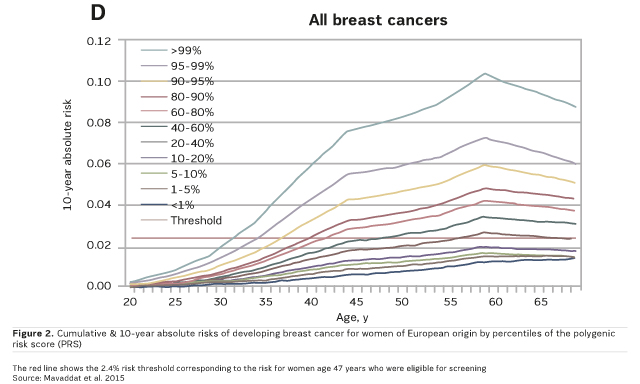
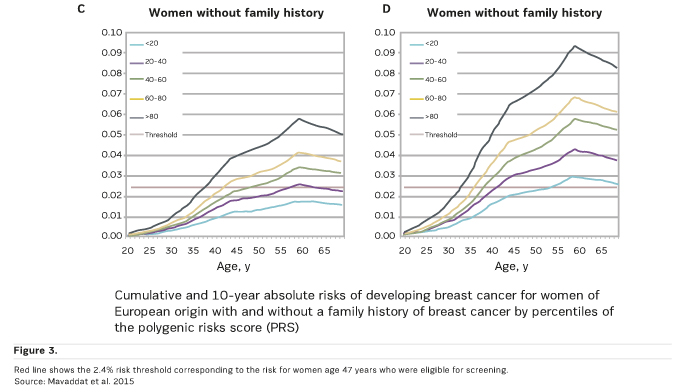
A study carried out by Mavaddat et al. in 2015 predicted breast cancer risk based on profiling with common genetic variants and concluded that the SNP77 percentiles of the polygenic risk score (PRS) stratifies breast cancer risk in women both with and without a family history of breast cancer (Figure 2, Figure 3). This level of risk discrimination can potentially inform targeted screening.
Managing risk
To offer risk-based screening, there are questions about risk estimation and management. Who should be responsible for estimating and managing women’s breast cancer risk? Who will do the risk counselling? What is the best age to obtain initial risk assessment? What should risk assessment include? Some current clinical trials, for example, will be using personal questionnaires on medical and family history, blood and saliva sampling in order to estimate breast cancer risk. There is also the issue of identifying women at sufficiently low risk for breast cancer. When is the risk so low that screening becomes inappropriate?
The American College of Radiology recently published recommendations for breast cancer screening in women at higher-than-average risk (Monticciolo et al. 2018), which include use of breast MRI and contrast-enhanced breast MRI and ultrasound to be considered for women who cannot undergo MRI. “All women, especially black women and those of Ashkenazi Jewish descent, should be evaluated for breast cancer risk no later than age 30, so that those at higher risk can be identified and can benefit from supplemental screening”, state the recommendations.
A number of trials are in progress in Europe and the U.S. to compare risk-adaptive screening regimens with standard ‘one size fits all’ screening programmes. The hypotheses for most of these trials are that a risk-based screening programme is safe (non-inferior in terms of Stage II+ breast cancer detected); superior (decreased incidence of Stage II+ breast cancers); equally or more cost-effective; and more acceptable to women, resulting in wider coverage and better compliance with screening invitations.
Rethinking breast cancer screening
In future women will likely need to be screened based on their risk of breast cancer. Radiologists will be responsible for assessing risk using risk models, breast density measurements, SNPs as well as lifestyle and hormonal factors (reproductive history, BMI, height, alcohol, oral contraceptives).
DBT is the appropriate modality for low risk women, who should be screened every 1-2 years (from age 40?) based on their risk. US is the best modality for women with dense breasts and low risk. MRI is appropriate for women with intermediate and high risk of developing breast cancer (5 or 10 year risk). Abbreviated MRI is a proven technique, although CEDM appears equivalent to abbreviated MRI and is a very promising modality.
Conclusion
Breast cancer screening in the future will no longer be ‘one size fits all’. Personalised screening based on a woman’s individual risk profile holds the potential to find ‘relevant’ cancers, those that could potentially be fatal, and thus decrease over diagnosis and over treatment as well as offer preventive treatment to eligible women. Ongoing clinical trials around the world will evaluate the effectiveness of risk based screening, its acceptability to women as well as its cost-effectiveness. Population-based screening has been a great achievement in healthcare, but precision medicine offers even more.