The new RAPID (Rapid Assessment Prostate Imaging and Diagnosis) programme accelerates the pathway which could lead to a ‘one-stop shop’ for men with prostate cancer by providing a multi-parametric MRI together with ultrasound imaging and biopsy on the same day. This will reduce unnecessary biopsies and provide accurate targeting whilst reducing the number of samples needed.
Hitachi Medical Systems Europe, manufacturer of high resolution ultrasound imaging equipment and a European distributor for BiopSee® , the image guided prostate biopsy system from MedCom, has been awarded the contract for the six high resolution ARIETTA ultrasound and BiopSee® fusion systems that together offer revolutionary technology for combining mp-MRI data with real-time ultrasound.
The pilots have been developed through funding from RM Partners, the Cancer Alliance for west London hosted by The Royal Marsden. Three trusts are piloting RAPID – Epsom and St Helier University Hospitals NHS Trust, Imperial College Healthcare NHS Trust, and St George’s University Hospitals NHS Foundation Trust.
The RAPID approach involves the patient having an mp-MRI scan in the morning and reported immediately. If it shows areas of abnormality, the patient will then undergo a transperineal ultrasound fusion biopsy, all within the same day.
The transperineal approach, in which samples are taken through the perineum rather than the rectum, is important as it confers a comparatively low risk of sepsis and can obviate the need for broad spectrum antibiotics and the possible development of antibiotic resistance.
Hashim Ahmed, Professor & Chair of Urology, Imperial College London, said “After trialling various systems on the market we felt that the HitachiARIETTA and MedCom BiopSee® combination offered us the best in high resolution imaging together with precise, accurate needle placement and an easy streamlined fusion software interface. This will help deliver a faster diagnosis and treatment for prostate cancer and reduce waiting times. The safer transperineal biopsy route will also reduce infection and life-threatening sepsis rates.”
Nicola Hunt, Managing Director at RM Partners, said: ‘We are delighted to be working with such inspirational clinicians who are at the forefront of delivering this innovative approach to prostate cancer diagnosis. In less than a year since the evidence was published to support this new approach, we have three sites across west London rolling out this investigative pathway. This is going to make a real difference to men through quicker diagnosis, reducing the anxious time waiting for scan results, allowing for earlier treatment if needed, and reducing the number of unnecessary biopsies undertaken. We are also delighted to be working with Hitachi to provide this world leading technology to our population.”
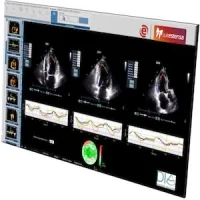
The BiopSee® system fuses the mp-MRI images with the Hitachi ultrasound images in realtime, allowing highly accurate placement of the biopsy needle in the prostate to target thetumour with exceptional precision. 3D ultrasound image acquisition, rigid/elastic fusion capabilities together with precise stepper tracking and reporting software deliver an exceptional diagnostic platform supporting the RAPID programme in London.
BiopSee® records and stores the location of each tissue sample, providing comprehensive documentation and reporting of biopsy core locations for accurate management of patients requiring further treatment and therapy planning in cryotherapy, brachytherapy, HIFU, RF or other treatment programmes.
The combined forces of Hitachi and MedCom BiopSee® offer the “best of both worlds“ to Urologists and patients alike: the superior quality of Hitachi ultrasound together with BiopSee’s® intuitive workflow open up new possibilities for patient management.
Mr Jean-Luc Budillon, President & COO, Hitachi Medical Systems Europe comments, “We’re very excited to have been selected as part of the RAPID programme and look forward to the roll-out of the project as Hitachi’s revolutionary technology is installed across these three sites. Our technological systems are designed by a team of highly skilled personnel, with years of expertise invested to bring this new hybrid system to the clinical environment. We are also fortunate to be working with some of the world’s leading medical experts on prostate cancer as part of the programme. It’s wonderful to see the adoption of such a forwardthinking approach to better managing the patient pathway in prostate cancer. It will be fascinating to analyse the data from RAPID and to fully understand its potential impact on diagnosis rates and care in the UK. We’re confident that patients will experience huge benefits from this innovative care pathway.”
Prostate cancer diagnosis has become a national health priority, with Prime Minister, Theresa May recently (9th April 2018) announcing the Government’s £75m prostate cancer drive to be administered via the National Institute for Health Research clinical research network. This initiative will see 40,000 men recruited for research into the disease, which kills more than 11,000 a year and causes great anxiety and sometimes suffering for the 47,000 men who get a diagnosis.
Find more about Hitachi Medical Systems