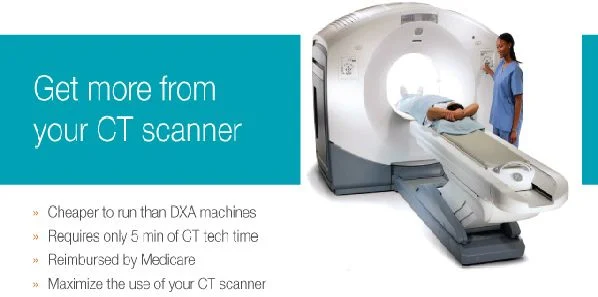
Mindways Software, Inc. (Austin, TX, USA) has obtained 510(k) clearance from the US Food and Drug Administration (FDA) to market its asynchronous Quantitative Computed Tomography (QCT) calibration technology for CT-based bone mineral density measurement. This latest QCT software innovation makes it possible to assess bone mineral density (BMD) using QCT without the need to scan a calibration phantom together with the patient, the company said.
The new technology works by using an earlier acquired calibration scan to transform a CT image into bone mineral density values. This translates to easier QCT workflow. More importantly, now any non-intravenous (IV)-contrast abdominal or pelvic CT scan ordered for another reason can be used for additional bone density measurement with no further patient radiation exposure or time, the manufacturer exlained.
Combined CTC and Osteoporosis Evaluation Screening Programme
In addition to standard CT scans, Mindways revealed that a successful pilot study has focused on the practicability of a combined CT colonography (CTC) and osteoporosis evaluation screening programme. Since patients undergoing CTC screening are typically over the age of 50, they will be at risk of low bone mass, which is a specific concern for postmenopausal women. The dual use of a CTC scan, therefore, can offer these patients “no-dose” BMD testing without the need to plan a dual-energy X-ray absorptiometry (DXA) test, according to the Texas-based software company.
“We are very excited about getting this innovative technology into healthcare practice. Because there is a great deal of overlap between patients getting a CT scan and those at risk of bone loss and consequent fragility fractures, the dual-use of CT scans provides for a very efficient method of screening for low bone mass and early identification patients that would benefit from treatment. This is very important because of the catastrophic nature of osteoporotic hip fractures,” said Alan Brett, PhD, VP of business development at Mindways Software Inc.
The new technology can also offer retrospective analysis of previously acquired images, by interrogating a picture archiving and communication system (PACS) store for CT scans of appropriate patient groups. The new QCT technology will be available sometime in the last quarter of 2014, Mindways said.
Mindways, an industry leader and technology innovator, develops QCT bone densitometry products for spine and hip bone density measurements. The QCT Pro uses QCT images to derive DXA-like to obtain hip BMD measurements and volumetric spine BMD measurements. QCT Pro hip BMD measurements are interpreted in the same manner as DXA and are included in the World Health Organization FRAX online fracture risk calculator, while spine measurements provide excellent sensitivity to short-term bone density changes, Mindways noted.
QCT Pro is fast, easy to use, and provides the same clinical utility as DXA at less cost, the company added.
Source: MedImaging.net
Image Credit: Mindways Software, Inc.
The new technology works by using an earlier acquired calibration scan to transform a CT image into bone mineral density values. This translates to easier QCT workflow. More importantly, now any non-intravenous (IV)-contrast abdominal or pelvic CT scan ordered for another reason can be used for additional bone density measurement with no further patient radiation exposure or time, the manufacturer exlained.
Combined CTC and Osteoporosis Evaluation Screening Programme
In addition to standard CT scans, Mindways revealed that a successful pilot study has focused on the practicability of a combined CT colonography (CTC) and osteoporosis evaluation screening programme. Since patients undergoing CTC screening are typically over the age of 50, they will be at risk of low bone mass, which is a specific concern for postmenopausal women. The dual use of a CTC scan, therefore, can offer these patients “no-dose” BMD testing without the need to plan a dual-energy X-ray absorptiometry (DXA) test, according to the Texas-based software company.
“We are very excited about getting this innovative technology into healthcare practice. Because there is a great deal of overlap between patients getting a CT scan and those at risk of bone loss and consequent fragility fractures, the dual-use of CT scans provides for a very efficient method of screening for low bone mass and early identification patients that would benefit from treatment. This is very important because of the catastrophic nature of osteoporotic hip fractures,” said Alan Brett, PhD, VP of business development at Mindways Software Inc.
Retrospective Analysis of Images Through PACS
The new technology can also offer retrospective analysis of previously acquired images, by interrogating a picture archiving and communication system (PACS) store for CT scans of appropriate patient groups. The new QCT technology will be available sometime in the last quarter of 2014, Mindways said.
Mindways, an industry leader and technology innovator, develops QCT bone densitometry products for spine and hip bone density measurements. The QCT Pro uses QCT images to derive DXA-like to obtain hip BMD measurements and volumetric spine BMD measurements. QCT Pro hip BMD measurements are interpreted in the same manner as DXA and are included in the World Health Organization FRAX online fracture risk calculator, while spine measurements provide excellent sensitivity to short-term bone density changes, Mindways noted.
QCT Pro is fast, easy to use, and provides the same clinical utility as DXA at less cost, the company added.
Source: MedImaging.net
Image Credit: Mindways Software, Inc.
Latest Articles
X-ray, Osteoporosis, CT scan, densitometry, bone
Mindways Software, Inc. (Austin, TX, USA) has obtained 510(k) clearance from the US Food and Drug Administration (FDA) to market its asynchronous Quantitat...