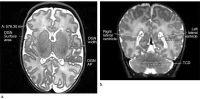
Age and habitual cigarette use factor into the risk assessment for lung cancer in former and current smokers. The implementation of a screening program, which utilises a state-of-the-art prediction model, can be more efficient for the identification of those with a risk high enough to warrant costly and potentially invasive imaging investigations. Advanced software tools can assist in the interpretation of low dose computed tomography (CT) images through improved accuracy and consistency of scan readings.
These recommendations are discussed in a new review published by the British Medical Journal and conducted by Canadian researchers Martin C. Tammemagi, from the Department of Health Sciences at Brock University in Ontario, and Stephen Lam from the British Columbia Cancer Agency and the University of British Columbia in Vancouver. The report coincides with news from the US that lung cancer screening will not be supported by Medicare.
Screening Reduces Lung Cancer Mortality
It is predicted that by 2030, lung cancer will be one of the three leading causes of death in high-income countries, and the fifth highest in middle-income nations. The variation relates to smoking habits around the world, which have changed over time. Incidence of lung cancer began to decline in America toward the very end of the twentieth century, but smoking and lung cancer are on the rise in developing countries.
The National Lung Screening Trial (NLST) published its findings in 2013. The randomised controlled trial discovered that the use of low dose (1.5mSv) CT to annually screen high-risk smokers between the ages of 55 and 74 has the potential to reduce lung cancer mortality by 20 percent. Since diagnoses have traditionally been made at late stages, clinical interventions over the past four decades have only minimally improved the chances of survival. More hospitals and health agencies are therefore considering the use of lung cancer screening to identify high-risk smokers and offer appropriate interventions.
Risk Prediction Models
In many settings, the calculation of cancer risk proceeds through the use of a formula based on age and smoking history, with a ‘pack year’ defined as the equivalent of smoking one pack of 20 cigarettes per day for a year. In the NLST study, high risk was associated with advanced age and at least 30 pack years, and smoking cessation more recent than 15 years for former smokers. New models have shown that additional factors increase predictive accuracy. Algorithms can then assess who among the high-risk smokers should be recommended for additional imaging or invasive procedures.
Identification of High Risk
Lung cancer screening has been most effective in people with higher risk. At least 15 prediction models exist, some developed for and applicable to special populations such as African Americans. Differences in screening methods determine how risk is assessed. In population-based approaches, phone or internet access is sufficient and no direct contact is required. Meanwhile, a medical system approach can incorporate clinical data, including future biomarkers.
Management of Nodules
Early-stage cancer diagnoses are associated with a much higher five-year survival rate: 70 percent, compared to 18 percent or less following the detection of late-stage disease. Advances in imaging technology over the past twenty years have enabled the detection of tiny (as small as one millimetre) lung nodules though the use of low dose CT. When nodules are detected, follow-up should be managed with guidelines based on minimising the number of unnecessary invasive investigations.
Screening and Smoking Cessation
Not only can screening predict outcomes for high-risk smokers, but the process may motivate current smokers to quit their habits. In a report separate from the Canadian review, CT screening for lung cancer was shown to significantly influence smokers who were confronted with their exam results showing suspicious nodules. The Journal of the National Cancer Institute study used data from 15,000 participants of the NLST. Despite the revelation, the Medicare Evidence Development and Coverage Advisory Committee recently voted against Medicare coverage of CT lung cancer screening, opting to support smoking cessation programs.
Future Considerations
The authors of the review cite a series of concerns which exist for the widespread use of such screening. Low dose CT screening for lung cancer may not be as effective at reducing mortality in the general population as it was in the NLST trial, which had participants who were more healthy and better educated than the average American, and who had more access to quality medical care. Furthermore, the reading and interpretation of results is substantially more time consuming than other types of cancer screening.
Furthermore, false positives could cause psychological as well as physical distress, if excess radiation, biopsies and surgeries are performed as follow-up procedures. The same harms are associated with over-diagnosis, in which untreated tumours might not progress to clinical significance. Cost is another consideration, although some analyses suggest that expense of implementing an efficient screening program could be neutralised by the advantages of earlier cancer detection.
However, until a more effective treatment modality is developed, the use of a prediction model, which incorporates the newest data, represents the best means available for reducing lung cancer mortality among high risk smokers. The results of a major Dutch-Belgian trial are expected in 2015, with the hope that the effects of low dose CT screening can be more clearly quantified. Quality assurance remains a priority, with computer vision software likely to assist highly accurate, low-cost scan interpretations.
Photo Credit: Google Images
References:
Tammemagi MC, Lam S (2014) Screening for lung cancer using low dose computed tomography. British Medical Journal (BMJ), 348:g2253. doi: 10.1136/bmj.g2253
Latest Articles
Imaging, Dose, Lung Cancer Screening, lung cancer, CT scans, smoking
Age and habitual cigarette use factor into the risk assessment for lung cancer in former and current smokers. The implementation of a screening program, wh...