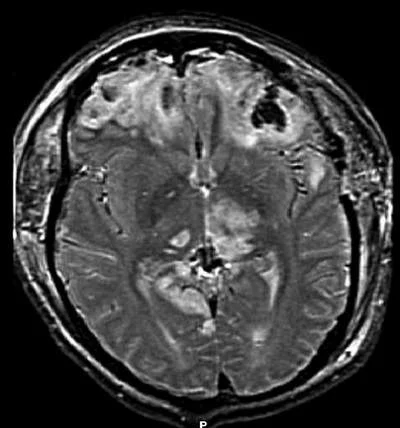
A simple blood test to measure brain-specific proteins released after a person suffers a traumatic brain injury (TBI) has been shown to predict both evidence of TBI on radiographic imaging and injury severity. A multicentre study, published in Journal of Neurotrauma, describes the potential benefit of adding detection of glial fibrillary acidic protein breakdown products (GFAP-BDP) to clinical screening with computed tomography (CT) and magnetic resonance imaging (MRI).
The study was conducted by Paul McMahon, University of Pittsburgh Medical Center, and a team of international researchers, including TRACK-TBI investigators. The researchers analysed blood levels of GFAP-BDP from 215 patients (aged 16-93 years) treated at multiple trauma centres for suspected TBI. They assessed the ability of the blood-based biomarker to predict intracranial injury as compared to the findings on an admission CT and a delayed MRI scan.
Based on the results, the researchers cite a net benefit for the use of GFAP-BDP above imaging-based screening alone and a net reduction in unnecessary scans by 12-30 percent.
"The study demonstrates that serum measurement of GFAP-BDP as a biomarker possesses the necessary characteristics (accuracy, discrimination, calibration, and clinical utility) for improved prediction of radiographically evident injury across the spectrum of TBI," the authors write in the journal report. "Additionally, GFAP-BDP levels were able to discriminate severity of intracranial injury independent of other classic injury predictors. GFAP-BDP also accurately predicted persistence of intracranial injury on imaging performed in the subacute period, again independent of other markers of injury risk."
John T. Povlishock, PhD, Editor-in-Chief of Journal of Neurotrauma and Professor, Medical College of Virginia Campus of Virginia Commonwealth University, Richmond, comments on the findings: "This impressive multicentre study joins with other streams of emerging evidence supporting the use of biomarkers as an important tool in the clinical decision making and prediction process.
"Importantly, this study significantly expands upon other studies that speak to the usefulness of GFAP and, specifically, serum-derived GFAP-BDP in identifying those traumatically brain injured patients whose clinical course is complicated by intracranial injury, demonstrating that GFAP-BDP offers good predictive ability, significant discrimination of injury severity, and net benefit in reducing the need for unnecessary scans, all of which have significant implications for the brain injured patient."
Source: Mary Ann Liebert, Inc., Publishers
Image credit: Wikimedia Commons
The study was conducted by Paul McMahon, University of Pittsburgh Medical Center, and a team of international researchers, including TRACK-TBI investigators. The researchers analysed blood levels of GFAP-BDP from 215 patients (aged 16-93 years) treated at multiple trauma centres for suspected TBI. They assessed the ability of the blood-based biomarker to predict intracranial injury as compared to the findings on an admission CT and a delayed MRI scan.
Based on the results, the researchers cite a net benefit for the use of GFAP-BDP above imaging-based screening alone and a net reduction in unnecessary scans by 12-30 percent.
"The study demonstrates that serum measurement of GFAP-BDP as a biomarker possesses the necessary characteristics (accuracy, discrimination, calibration, and clinical utility) for improved prediction of radiographically evident injury across the spectrum of TBI," the authors write in the journal report. "Additionally, GFAP-BDP levels were able to discriminate severity of intracranial injury independent of other classic injury predictors. GFAP-BDP also accurately predicted persistence of intracranial injury on imaging performed in the subacute period, again independent of other markers of injury risk."
John T. Povlishock, PhD, Editor-in-Chief of Journal of Neurotrauma and Professor, Medical College of Virginia Campus of Virginia Commonwealth University, Richmond, comments on the findings: "This impressive multicentre study joins with other streams of emerging evidence supporting the use of biomarkers as an important tool in the clinical decision making and prediction process.
"Importantly, this study significantly expands upon other studies that speak to the usefulness of GFAP and, specifically, serum-derived GFAP-BDP in identifying those traumatically brain injured patients whose clinical course is complicated by intracranial injury, demonstrating that GFAP-BDP offers good predictive ability, significant discrimination of injury severity, and net benefit in reducing the need for unnecessary scans, all of which have significant implications for the brain injured patient."
Source: Mary Ann Liebert, Inc., Publishers
Image credit: Wikimedia Commons
References:
McMahon PJ et al. (2015) Measurement of the Glial Fibrillary Acidic Protein and Its Breakdown Products GFAP-BDP Biomarker for the Detection of Traumatic Brain Injury Compared to Computed Tomography and Magnetic Resonance Imaging. Journal of Neurotrauma. April 15, 2015, 32(8):
527-533. doi:10.1089/neu.2014.3635.
Latest Articles
healthmanagement, trauma, brain injury, computed tomography, MRI, biomarkers
A simple blood test to measure brain-specific proteins released after a person suffers a traumatic brain injury (TBI) can predict both evidence of TBI on radiographic imaging and injury severity.