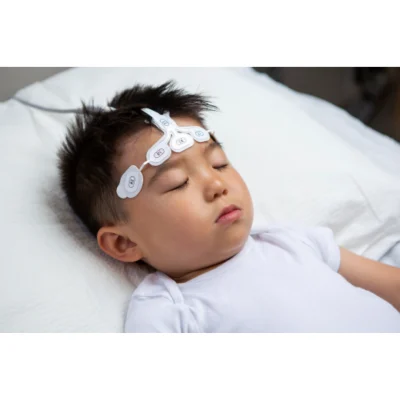
Masimo has announced the FDA clearance of SedLine® brain function monitoring for pediatric patients (1-17 years of age) and the SedLine Pediatric EEG Sensor. With this clearance, the potential benefits of SedLine have been expanded to all patients one year old and above in the United States. Equipped with Masimo's advanced signal processing technology, SedLine helps clinicians monitor brain activity bilaterally by processing electroencephalogram (EEG) signals from Masimo's four-lead SedLine EEG sensors.
This clearance brings Masimo's bilateral brain activity monitoring to children 1 to 17 years old, in conjunction with specially sized pediatric sensors designed for easier application on smaller pediatric foreheads. Brain activity monitoring under anesthesia on pediatric patients is different from that of adults.1-2 Maintaining an appropriate depth of anesthesia is key to preventing anesthesia-related events and enabling faster recovery.3 To aid clinicians in monitoring anesthesia depth on children, SedLine features both the display of EEG signals and the Multitaper Density Spectral Array (DSA) from both sides of the brain, to provide clinicians with a more complete picture of the brain.
Dr. Cristina Verdú of Hospital Universitario La Paz, Madrid, Spain, said, "SedLine is an easy window into a child's electroencephalogram. It helps us to personalize sedoanalgesia. Now, we can choose the appropriate dose according to its effects, not just according to weight or age. But in addition to monitoring anesthetic depth, it allows us to detect warning signs such as asymmetries or seizures; it tells us what is happening to the child's brain."
Joe Kiani, Founder and CEO of Masimo, said, "SedLine is achieving for brain function monitoring what Masimo SET® did for pulse oximetry. We believe SedLine is the best and most advanced way to monitor depth of sedation, crucial to helping ensure patients with even the most challenging and the youngest brains are appropriately anesthetized. We are proud to be able to bring its benefits to children in the United States."
Source: Masimo
References
- Davidson et. Current Anesthesiology Reports 3. 1 (2013): 57-63.).
- Cornelissen L et al. Elife 4 (2015): e06513.
- Musialowicz et al. Current Anesthesiology Reports 4. 3 (2014): 251-260