The body’s immune system is set up much like a home security system; it has sensors on the outside of cells that act like motion detectors — floodlights — that click on when there’s an intruder rustling in the bushes, bacteria that seem suspect. For over a decade researchers have known about one group of external sensors called Toll-like receptors that detect when bacteria are nearby.
Now, researchers at the University of North Carolina School of
Medicine have identified a sensor pathway inside cells. These internal
sensors are like motion detectors inside a house; they trigger an alarm
that signals for help — a response from the immune system. This
research, published in the Sept. 13, 2013 issue of the journal Science,
indicates that both exterior and interior sensors work together to
detect the same component of bacterial cell membranes, a molecule called
lipopolysaccharide or LPS.
By showing how the immune system distinguishes between suspicious
activity and real threats, the study could lead to new therapies for
septic shock — when the immune system overreacts to a bacterial
infection to such an extent that it causes more harm than good.
“During the defense against an infection you want to be able to
differentiate between the bacteria that stay on the outside of the cell
and the ones that get inside,” said senior study author Edward A. Miao,
MD, PhD, assistant professor of microbiology and immunology. “You can
think of the exterior sensors as a yellow alert; they tell us that
bacteria are present. But these bacteria could either be simple ones in
the wrong place, or very dangerous ones that could cause a serious
infection. The interior sensors act as a red alert; they warn us that
there are bacteria with ill intent that have the genetic capacity to
invade and manipulate our cells.”
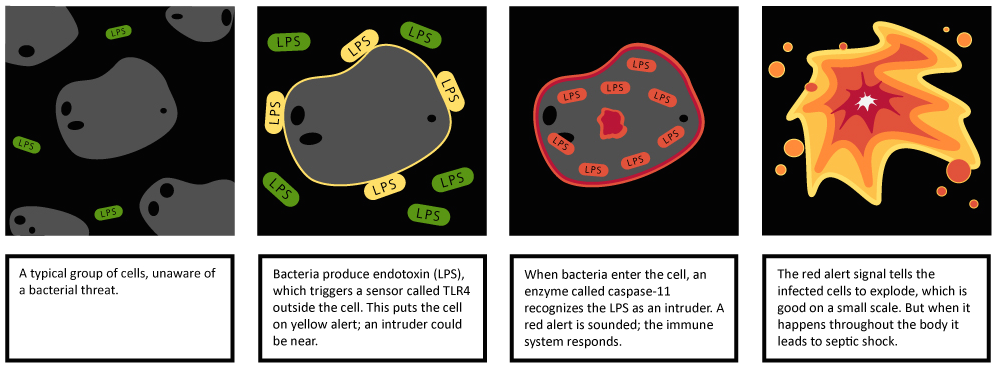
About half of the cases of septic shock are caused by bacteria that
produce LPS, also known as endotoxin. In fact, much of what is known
about endotoxic shock comes from studying animals injected with high
doses of LPS. For example, previous studies pinpointed the role of the
Toll-like receptor 4 gene (TLR4) as a sensor on the outside of cells;
mice without that gene resisted endotoxic shock.
In a study published in January 2013, also in the journal Science,
Miao and his colleagues showed that a sensor called caspase-11 sounds
an alert when bacteria enter a cell. However, it wasn’t clear which of
the thousands of molecules that make up a bacterial cell triggers that
new sensor.
In the current study, Miao and his colleagues investigated which bits
of foreign material were being detected. They took apart and delivered
different chunks of bacteria into the cytoplasmic compartment inside the
cell. To their surprise, they found that the caspase-11 sensor inside
the cell was detecting the same molecule, LPS, as the TLR4 sensor
outside the cell. The researchers wondered whether there was a link
between these two sensors.
Through a number of experiments in animal models of sepsis, Miao’s
team showed that the exterior and interior alarms work together through a
two-step defense mechanism: LPS is first seen on the outside of the
cell by TLR4, which sets the interior caspase-11 alarm into a watchful
state. At very high doses, the LPS crosses into the cell, tripping the
caspase-11 alarm. The end result is the generation of the red alert
signal, which causes the cell to explode, a form of cell death called
pyroptosis. During an infection, the immune system essentially burns the
house down around the invading bacteria, depriving it of a place to
replicate, and exposing it to more potent immune defenses. During
sepsis, however, too much fire leads to the onset of shock.
Miao says that figuring out how these two
sensors get activated in response to a bacterial infection could help
researchers develop new ways of preventing or treating septic shock, a
condition that kills about half its victims.
“The septic shock we see in patients is probably a lot more
complicated than what we see in this experimental system,” said Miao.
“The next question we need to ask is whether these same sensors are
going off in people with septic shock, and if so, is there a way to
block them so we can keep patients from dying.”
Study co-authors from UNC were Jon A. Hagar and Youssef Aachoui. The
work was done in collaboration with Robert K. Ernst and Daniel A.
Powell at the University of Maryland in Baltimore. This work was
supported by NIH grants AI007273 (J.A.H), AI097518 (E.A.M), AI057141
(E.A.M), and AI101685 (R.K.E).
Source: University of North Carolina