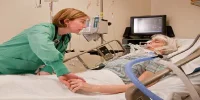
Royal Philips has announced a new initiative with Massachusetts Institute of Technology (MIT) to give medical researchers access to one of the largest data sources available for research into critical care. The data from more than 100,000 patients have been collected and anonymised through the Philips Hospital to Home eICU telehealth programme. These data sets from patient stays in eICU centres account for about 10 percent of all adult ICU beds in the United States.
The Laboratory of Computational Physiology within the MIT Institute for Medical Engineering and Science will serve as the academic research hub for the initiative. It will provide and maintain access, as well as help educate healthcare researchers on the database and offer a platform for collaboration.
The secure database will include anonymised and detailed clinical data such as vital signs, pharmacy medication orders, laboratory results, diagnoses and severity of illness scores, giving researchers comprehensive insights into a patient stay. Currently, most inpatient multicentre data sets available to researchers are limited to insurance claims data, which provide just a summary of a patient's stay.
"Researchers are always looking for better, more accurate and comprehensive data that enables a holistic representation of the patient experience," according to Leo Anthony Celi of MIT. "The quality and resolution of the data Philips has been collecting in the critical care domain is unprecedented. This kind of access will provide researchers with data that will enable investigations otherwise unimaginable."
The data will be available to researchers via PhysioNet before the end of the year. PhysioNet offers free web access to large collections of physiologic signals and related open-source software. Researchers will be able to contribute to the expanding knowledge base by sharing their methodologies and findings with the larger research community to facilitate faster discoveries in the future.
"We're proud to be leading the charge in the industry by opening up our data to independent researchers to allow them to conduct more meaningful analyses related to critical care medicine," said Derek Smith, senior vice president, Hospital to Home, Philips Healthcare. “This initiative will make it easier for researchers to share methods and findings, bypassing the need to reinvent the wheel with each new research project. We hope this will lead to better, faster breakthroughs — and ultimately better medicine.”
The data set is compiled from records shared by hundreds of ICUs across the United States, and is managed by the Philips eICU Research Institute. This is one of the most extensive ICU-centric longitudinal data sets in existence and has been used by participants in the Philips eICU Research Institute to produce valuable peer-reviewed critical care research.
Source: www.newscenter.philips.com
Image Credit: Philips N.V.
The Laboratory of Computational Physiology within the MIT Institute for Medical Engineering and Science will serve as the academic research hub for the initiative. It will provide and maintain access, as well as help educate healthcare researchers on the database and offer a platform for collaboration.
The secure database will include anonymised and detailed clinical data such as vital signs, pharmacy medication orders, laboratory results, diagnoses and severity of illness scores, giving researchers comprehensive insights into a patient stay. Currently, most inpatient multicentre data sets available to researchers are limited to insurance claims data, which provide just a summary of a patient's stay.
"Researchers are always looking for better, more accurate and comprehensive data that enables a holistic representation of the patient experience," according to Leo Anthony Celi of MIT. "The quality and resolution of the data Philips has been collecting in the critical care domain is unprecedented. This kind of access will provide researchers with data that will enable investigations otherwise unimaginable."
The data will be available to researchers via PhysioNet before the end of the year. PhysioNet offers free web access to large collections of physiologic signals and related open-source software. Researchers will be able to contribute to the expanding knowledge base by sharing their methodologies and findings with the larger research community to facilitate faster discoveries in the future.
"We're proud to be leading the charge in the industry by opening up our data to independent researchers to allow them to conduct more meaningful analyses related to critical care medicine," said Derek Smith, senior vice president, Hospital to Home, Philips Healthcare. “This initiative will make it easier for researchers to share methods and findings, bypassing the need to reinvent the wheel with each new research project. We hope this will lead to better, faster breakthroughs — and ultimately better medicine.”
The data set is compiled from records shared by hundreds of ICUs across the United States, and is managed by the Philips eICU Research Institute. This is one of the most extensive ICU-centric longitudinal data sets in existence and has been used by participants in the Philips eICU Research Institute to produce valuable peer-reviewed critical care research.
Source: www.newscenter.philips.com
Image Credit: Philips N.V.
Latest Articles
Telehealth, Philips, Critical Care, eICU, MIT
Royal Philips has announced a new initiative with Massachusetts Institute of Technology (MIT) to give medical researchers access to one of the largest data...