Among patients with septic shock who had severe acute kidney injury (AKI), there was no significant difference in overall mortality at 90 days between patients who were assigned to an early strategy for the initiation of renal-replacement therapy and those who were assigned to a delayed strategy, according to a randomised controlled trial published in The New England Journal of Medicine.
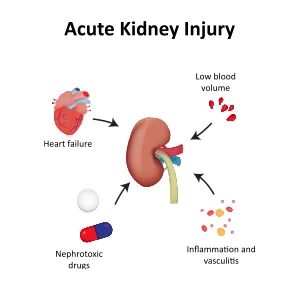
AKI is the most frequent complication in patients with septic shock and is an independent risk factor for death. Although renal-replacement therapy is the standard of care for severe AKI, the ideal time for initiation remains controversial.
The Initiation of Dialysis Early Versus Delayed in the Intensive Care Unit (IDEAL-ICU) trial was a multicentre study that included patients admitted to the ICU with early-stage septic shock who had severe AKI at the failure stage of the risk, injury, failure, loss, and end-stage kidney disease (RIFLE) classification system but without life-threatening complications related to acute kidney injury. Patients were randomly assigned to receive renal-replacement therapy either within 12 hours after documentation of failure-stage acute kidney injury (early strategy) or after a delay of 48 hours if renal recovery had not occurred (delayed strategy).
The failure stage of the RIFLE classification system is characterised by a serum creatinine level 3 times the baseline level (or ≥4 mg per decilitre with a rapid increase of ≥0.5 mg per decilitre), urine output less than 0.3 ml per kilogram of body weight per hour for 24 hours or longer, or anuria for at least 12 hours. The primary outcome was death from any cause at 90 days.
The trial was stopped early for futility after the second planned interim analysis. A total of 488 patients underwent randomisation; there were no significant between-group differences in the characteristics at baseline. Among the 477 patients for whom follow-up data at 90 days were available, 58% of the patients in the early-strategy group (138 of 239 patients) and 54% in the delayed-strategy group (128 of 238 patients) had died (P=0.38). In the delayed-strategy group, 38% (93 patients) did not receive renal-replacement therapy. Criteria for emergency renal-replacement therapy were met in 17% of the patients in the delayed-strategy group (41 patients).
In addition, the delayed strategy resulted in a significantly larger number of days free of renal-replacement therapy (median days, 16; interquartile range, 2 to 28) than the early strategy (median days, 12; interquartile range, 1 to 25) (P=0.006). There were no significant differences between the groups in the other secondary outcomes, namely mortality at 28 days and 180 days, number of days free of mechanical ventilation and vasopressors, and length of ICU and hospital stay.
According to the authors, the results show that initiating renal-replacement therapy too early could unnecessarily expose patients in whom renal dysfunction would have recovered spontaneously to the risks associated with renal-replacement therapy.
"Although our trial did not show any benefit to expediting the initiation of renal-replacement therapy in the absence of emergency criteria, our results cannot be interpreted as encouraging indefinite deferral of renal-replacement therapy," the authors note. "Rather, our data indicate that the risk of death is not increased if renal-replacement therapy is postponed for at least 48 hours, as long as care is taken to identify patients in whom criteria for emergency renal-replacement therapy are likely to be met."
Source: The New England Journal of Medicine
Image credit: iStock
AKI is the most frequent complication in patients with septic shock and is an independent risk factor for death. Although renal-replacement therapy is the standard of care for severe AKI, the ideal time for initiation remains controversial.
The Initiation of Dialysis Early Versus Delayed in the Intensive Care Unit (IDEAL-ICU) trial was a multicentre study that included patients admitted to the ICU with early-stage septic shock who had severe AKI at the failure stage of the risk, injury, failure, loss, and end-stage kidney disease (RIFLE) classification system but without life-threatening complications related to acute kidney injury. Patients were randomly assigned to receive renal-replacement therapy either within 12 hours after documentation of failure-stage acute kidney injury (early strategy) or after a delay of 48 hours if renal recovery had not occurred (delayed strategy).
The failure stage of the RIFLE classification system is characterised by a serum creatinine level 3 times the baseline level (or ≥4 mg per decilitre with a rapid increase of ≥0.5 mg per decilitre), urine output less than 0.3 ml per kilogram of body weight per hour for 24 hours or longer, or anuria for at least 12 hours. The primary outcome was death from any cause at 90 days.
The trial was stopped early for futility after the second planned interim analysis. A total of 488 patients underwent randomisation; there were no significant between-group differences in the characteristics at baseline. Among the 477 patients for whom follow-up data at 90 days were available, 58% of the patients in the early-strategy group (138 of 239 patients) and 54% in the delayed-strategy group (128 of 238 patients) had died (P=0.38). In the delayed-strategy group, 38% (93 patients) did not receive renal-replacement therapy. Criteria for emergency renal-replacement therapy were met in 17% of the patients in the delayed-strategy group (41 patients).
In addition, the delayed strategy resulted in a significantly larger number of days free of renal-replacement therapy (median days, 16; interquartile range, 2 to 28) than the early strategy (median days, 12; interquartile range, 1 to 25) (P=0.006). There were no significant differences between the groups in the other secondary outcomes, namely mortality at 28 days and 180 days, number of days free of mechanical ventilation and vasopressors, and length of ICU and hospital stay.
According to the authors, the results show that initiating renal-replacement therapy too early could unnecessarily expose patients in whom renal dysfunction would have recovered spontaneously to the risks associated with renal-replacement therapy.
"Although our trial did not show any benefit to expediting the initiation of renal-replacement therapy in the absence of emergency criteria, our results cannot be interpreted as encouraging indefinite deferral of renal-replacement therapy," the authors note. "Rather, our data indicate that the risk of death is not increased if renal-replacement therapy is postponed for at least 48 hours, as long as care is taken to identify patients in whom criteria for emergency renal-replacement therapy are likely to be met."
Source: The New England Journal of Medicine
Image credit: iStock
References:
Barbar SD, Clere-Jehl R, Quenot J-P et al. for the IDEAL-ICU Trial
Investigators and the CRICS TRIGGERSEP Network (2018) Timing ofRenal-Replacement Therapy in Patients with Acute Kidney Injury andSepsis. N Engl J Med 379:1431-1442 DOI: 10.1056/NEJMoa1803213
Latest Articles
Sepsis, Renal replacement therapy, acute kidney injury
Among patients with septic shock who had severe acute kidney injury (AKI), there was no significant difference in overall mortality at 90 days between pati...