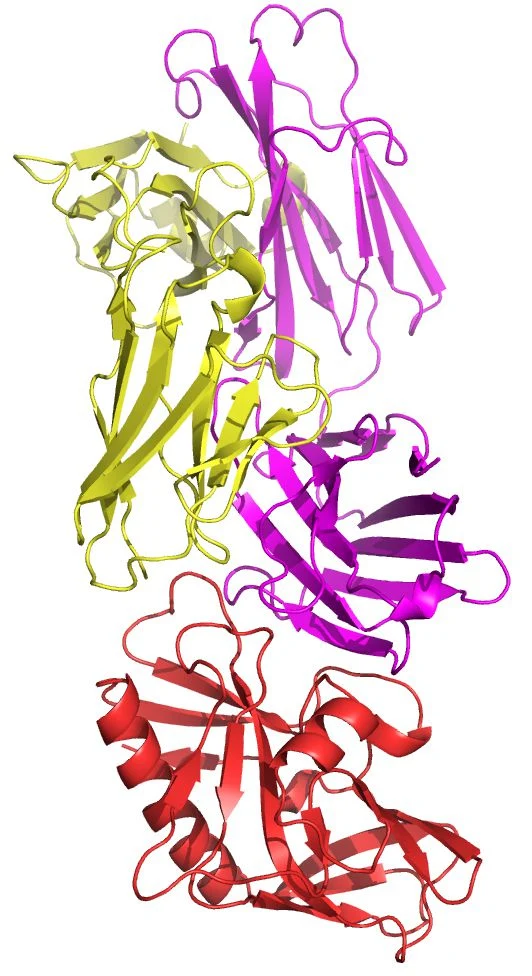
The researchers demonstrated their new technique by engineering a compound that has promise to initiate an otherwise rare immune response against many types of HIV. Here, the germline-targeting immunogen eOD-GT6 (red) is shown bound to its target, the germline VRC01 antibody (magenta and yellow). (Image: Credit TSRI)
A team led by scientists from The Scripps Research Institute (TSRI) and the International AIDS Vaccine Initiative (IAVI) has unveiled a new technique for vaccine design that could be particularly useful against HIV and other fast-changing viruses.
The
report, which appears March 28, 2013, in Science
Express, the early online edition of the journal Science, offers a step toward solving what has been one of the
central problems of modern vaccine design: how to stimulate the immune system
to produce the right kind of antibody response to protect against a wide range
of viral strains. The researchers demonstrated their new technique by engineering
an immunogen (substance that induces immunity) that has promise to reliably
initiate an otherwise rare response effective against many types of HIV.
“We’re
hoping to test this immunogen soon in mice engineered to produce human
antibodies, and eventually in humans,” said team leader William R. Schief, who
is an associate professor of immunology and member of the IAVI Neutralizing
Antibody Center at TSRI.
Seeking a Better Way
For
highly variable viruses such as HIV and influenza, vaccine researchers want to
elicit antibodies that protect against most or all viral strains—not just a few
strains, as seasonal flu vaccines currently on the market. Vaccine researchers have
identified several of these broadly neutralizing antibodies from long-term
HIV-positive survivors, harvesting antibody-producing B cells from blood
samples and then sifting through them to identify those that produce antibodies
capable of neutralizing multiple strains of HIV. Such broadly neutralizing
antibodies typically work by blocking crucial functional sites on a virus that are
conserved among different strains despite high mutation elsewhere.
However,
even with these powerful broadly neutralizing antibodies in hand, scientists
need to find a way to elicit their production in the body through a vaccine. “For
example, to elicit broadly neutralizing antibodies called VRC01-class
antibodies that neutralize 90 percent of known HIV strains, you could try using
the HIV envelope protein as your immunogen,” said Schief, “but you run into the
problem that the envelope protein doesn’t bind with any detectable affinity to
the B cells needed to launch a broadly neutralizing antibody response.”
To
reliably initiate that VRC01-class antibody response, Schief and his colleagues
therefore sought to develop a new method for designing vaccine immunogens.
From Weak to Strong
Joseph Jardine, a TSRI graduate student in the Schief laboratory, evaluated the genes of VRC01-producing B cells in order to deduce the identities of the less mature B cells—known as germline B cells—from which they originate. Germline B cells are major targets of modern viral vaccines, because it is the initial stimulation of these B cells and their antibodies that leads to a long-term antibody response.In
response to vaccination, germline B cells could, in principle, mature into the
desired VRC01-producing B cells—but natural HIV proteins fail to bind or stimulate
these germline B cells so they cannot get the process started. The team thus
set out to design an artificial immunogen that would be successful at achieving
this.
Jardine
used a protein modeling software suite called Rosetta to improve the binding of
VRC01 germline B cell antibodies to HIV’s envelope protein. “We asked Rosetta
to look for mutations on the side of the HIV envelope protein that would help
it bind tightly to our germline antibodies,” he said.
Rosetta
identified dozens of mutations that could help improve binding to germline
antibodies. Jardine then generated libraries that contained all possible
combinations of beneficial mutations, resulting in millions of mutants, and
screened them using techniques called yeast surface display and FACS. This
combination of computational prediction and directed evolution successfully
produced a few mutant envelope proteins with high affinity for germline VRC01-class
antibodies.
Jardine
then focused on making a minimal immunogen—much smaller than HIV envelope—and
so continued development using the “engineered outer domain (eOD)” previously
developed by Po-Ssu Huang in the Schief lab while Schief was at the University
of Washington. Several iterative rounds of design and selection using a panel
of germline antibodies produced a final, optimized immunogen—a construct they
called eOD-GT6.
A Closer Look
To
get a better look at eOD-GT6 and its interaction with germline antibodies, the
team turned to the laboratory of Ian A. Wilson, chair of the Department
of Integrative Structural and Computational Biology and a member of the IAVI Neutralizing
Antibody Center at TSRI.
Jean-Philippe
Julien, a senior research associate in the Wilson laboratory, determined the 3D
atomic structure of the designed immunogen using X-ray crystallography—and, in
an unusual feat, also determined the crystal structure of a germline VRC01
antibody, plus the structure of the immunogen and antibody bound together.
“We
wanted to know whether eOD-GT6 looked the way we anticipated and whether it bound
to the antibody in the way that we predicted—and in both cases the answer was
‘yes’,” said Julien. “We also were able to identify the key mutations that conferred
its reactivity with germline VRC01 antibodies.”
Mimicking a Virus
Vaccine researchers know that such an immunogen typically does better at stimulating an antibody response when it is presented not as a single copy but in a closely spaced cluster of multiple copies, and with only its antibody-binding end exposed. “We wanted it to look like a virus,” said Sergey Menis, a visiting graduate student in the Schief laboratory.
Menis
therefore devised a tiny virus-mimicking particle made from 60 copies of an
obscure bacterial enzyme and coated it with 60 copies of eOD-GT6. The particle
worked well at activating VRC01 germline B cells and even mature B cells in the
lab dish, whereas single-copy eOD-GT6 did not.
“Essentially
it’s a self-assembling nanoparticle that presents the immunogen in a properly
oriented way,” Menis said. “We’re hoping that this approach can be used not
just for an HIV vaccine but for many other vaccines, too.”
The
next step for the eOD-GT6 immunogen project, said Schief, is to test its
ability to stimulate an antibody response in lab animals that are themselves
engineered to produce human germline antibodies. The difficulty with testing
immunogens that target human germline antibodies is that animals typically used
for vaccine testing cannot make those same antibodies. So the team is
collaborating with other researchers who are engineering mice to produce human germline
antibodies. After that, he hopes to learn how to drive the response, from the activation
of the germline B cells all the way to the production of mature, broadly
neutralizing VRC01-class antibodies, using a series of designed immunogens.
Schief
also hopes they will be able to test their germline-targeting approach in
humans sooner rather than later, noting “it will be really important to find
out if this works in a human being.”
The first authors of
the paper, “Rational HIV immunogen
design to target specific germline B cell receptors,” were Jardine, Julien and
Menis. Co-authors were Takayuki
Ota and Devin Sok of the Nemazee and Burton laboratories at TSRI, respectively;
Travis Nieusma of the Ward laboratory at TSRI; John Mathison of the Ulevitch laboratory
at TSRI; Oleksandr Kalyuzhniy and Skye MacPherson, researchers in the Schief laboratory
from IAVI and TSRI, respectively; Po-Ssu Huang and David Baker of the
University of Washington, Seattle; Andrew McGuire and Leonidas Stamatatos of
the Seattle Biomedical Research Institute; and TSRI
principal investigators Andrew B. Ward, David Nemazee, Ian A. Wilson,
and Dennis R. Burton, who is also head of the IAVI Neutralizing Center at TSRI.
Source: The Scripps Research Institute