New guidelines on the prevention and control of multidrug-resistant Gram-negative bacteria in hospitals have been published by the Journal of Hospital Infection. These guidelines are accredited by the National Institute for Health and Care Excellence in the UK.
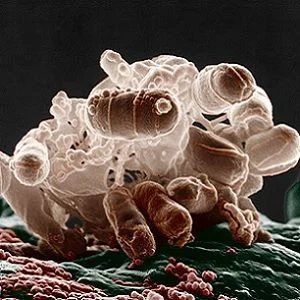
Gram-negative bacteria (MDRGNB) include common types such as E. coli that are present in human bowels. When these bacteria get into other parts of the body they can cause infections, including common infections such as cystitis or urinary tract infections. The bacteria can also be readily spread from person to person. Multidrug-resistant strains are resistant to most, or all, of the common antibiotics that are used to treat infections.
Professor Peter Wilson, professor of microbiology at University College London Hospitals NHS Foundation Trust and Chairman of the Working Group that developed the guidelines, explained: "Multi-resistant Gram negative bacteria have spread rapidly in some areas of Europe and threaten to do so in the UK. We conducted a wide ranging review of the evidence for preventing spread of these infections and have produced straightforward practical advice which we hope can be used by all grades of staff in every healthcare setting. If widely adopted we can prevent the problem becoming widespread."
The release of the new guidelines coincided with the first World Health Organization World Antibiotic Awareness Week. Much of the focus of the WHO's new campaign was on proper use of antibiotics to help prevent, or slow down, the emergence of MDRGNB. As these bacteria are already seen in the UK, authors of the guidelines said it is important to have systems in place to identify patients with these bacteria, and to make sure that they do not spread further in hospitals.
Dr. Jim Gray, Editor of the Journal of Hospital Infection, noted on the release of the guidelines, "Five or six years ago multi-resistant Gram-negative bacteria were a rarity in many UK hospitals. Now every hospital is seeing these bacteria regularly, as is evidenced by the large number of papers on the subject of MDRGNB that are published in the Journal of Hospital Infection."
As these guidelines are based on a synthesis of the best available evidence, Dr. Gray said the document "will play a crucial part in ensuring that all hospitals have ready access to a comprehensive toolkit to manage the very real threat posed by MDRGNB."
Source: Elsevier
Image credit: Flickr.com
Gram-negative bacteria (MDRGNB) include common types such as E. coli that are present in human bowels. When these bacteria get into other parts of the body they can cause infections, including common infections such as cystitis or urinary tract infections. The bacteria can also be readily spread from person to person. Multidrug-resistant strains are resistant to most, or all, of the common antibiotics that are used to treat infections.
Professor Peter Wilson, professor of microbiology at University College London Hospitals NHS Foundation Trust and Chairman of the Working Group that developed the guidelines, explained: "Multi-resistant Gram negative bacteria have spread rapidly in some areas of Europe and threaten to do so in the UK. We conducted a wide ranging review of the evidence for preventing spread of these infections and have produced straightforward practical advice which we hope can be used by all grades of staff in every healthcare setting. If widely adopted we can prevent the problem becoming widespread."
The release of the new guidelines coincided with the first World Health Organization World Antibiotic Awareness Week. Much of the focus of the WHO's new campaign was on proper use of antibiotics to help prevent, or slow down, the emergence of MDRGNB. As these bacteria are already seen in the UK, authors of the guidelines said it is important to have systems in place to identify patients with these bacteria, and to make sure that they do not spread further in hospitals.
Dr. Jim Gray, Editor of the Journal of Hospital Infection, noted on the release of the guidelines, "Five or six years ago multi-resistant Gram-negative bacteria were a rarity in many UK hospitals. Now every hospital is seeing these bacteria regularly, as is evidenced by the large number of papers on the subject of MDRGNB that are published in the Journal of Hospital Infection."
As these guidelines are based on a synthesis of the best available evidence, Dr. Gray said the document "will play a crucial part in ensuring that all hospitals have ready access to a comprehensive toolkit to manage the very real threat posed by MDRGNB."
Source: Elsevier
Image credit: Flickr.com
References:
Wilson APR et al. (2015) Prevention and control of multi-drug-resistant Gram-negative bacteria: recommendations from a Joint Working Party.
Journal of Hospital Infection; published online 19 October 2015. DOI:
10.1016/j.jhin.2015.08.007
Latest Articles
healthmanagement, antibiotics, infections, guidelines, antibiotic resistance, E. coli
New guidelines on the prevention and control of multidrug-resistant Gram-negative bacteria in hospitals have been published by the Journal of Hospital Infection. These guidelines are accredited by the National Institute for Health and Care Excellence in t