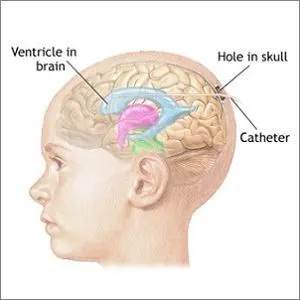
Seven consensus statements from a panel of leading international experts in the treatment of severe traumatic brain injury (sTBI) provide a clear interpretation of the results from the BEST TRIP trial, which have led to ongoing debate over the value of intracranial pressure monitoring in sTBI. These new consensus statements, published in Journal of Neurotrauma, are intended to help guide practising physicians and researchers.
The "Benchmark Evidence from South American Trials: Treatment of Intracranial Pressure (BEST TRIP)" randomised controlled trial compared two management protocols for treatment of severe TBI: one involving ICP monitoring and the other involving serial CT imaging and neurologic examination. However, publication of the trial results has caused much controversy in the treatment of sTBI.
In response to this dissonance, 23 international opinion leaders in acute care sTBI management met to draft a consensus statement to interpret this study. A Delphi method-based approach employed iterative pre-meeting polling to codify the group's general opinions, followed by an in-person meeting wherein individual statements were refined. Statements required an agreement threshold of more than 70 percent by blinded voting for approval. Seven precisely-worded statements resulted, with agreement levels of 83 to 100 percent. These new consensus statements help clarify the trial protocols, the different patient outcomes with and without ICP monitoring, the validity of the trial, and the main implications of the trial results.
"We hope that these statements will serve as a reference to practising physicians, researchers, and clinical associations in guiding the interpretation of the BEST TRIP results and mitigating what one committee member termed 'collateral damage' from its misunderstanding. Further consensus recommendations are presently in preparation regarding clinical and research approaches to ICP monitoring and treatment in light of current data," the authors conclude.
"This brief but eloquent consensus report helps revisit the overall implications and interpretations derived from the BEST TRIP trial," says John T. Povlishock, PhD, Editor-in-Chief of Journal of Neurotrauma and Professor, Medical College of Virginia Campus of Virginia Commonwealth University, Richmond. "As noted by the authors, this consensus document reframes many of the controversial issues generated in this initial trial, calling for a more critical evaluation of the study and its overall interpretation. From my perspective as Editor-in-Chief, I believe the consensus positions detailed therein help place this trial in the appropriate intellectual framework, while highlighting the continued need for more rigorous evaluation of intracranial hypertension, its monitoring, and its implications for traumatically brain-injured patients."
Source: Mary Ann Liebert, Inc., Publishers
Image credit: U.S. National Library of Medicine
The "Benchmark Evidence from South American Trials: Treatment of Intracranial Pressure (BEST TRIP)" randomised controlled trial compared two management protocols for treatment of severe TBI: one involving ICP monitoring and the other involving serial CT imaging and neurologic examination. However, publication of the trial results has caused much controversy in the treatment of sTBI.
In response to this dissonance, 23 international opinion leaders in acute care sTBI management met to draft a consensus statement to interpret this study. A Delphi method-based approach employed iterative pre-meeting polling to codify the group's general opinions, followed by an in-person meeting wherein individual statements were refined. Statements required an agreement threshold of more than 70 percent by blinded voting for approval. Seven precisely-worded statements resulted, with agreement levels of 83 to 100 percent. These new consensus statements help clarify the trial protocols, the different patient outcomes with and without ICP monitoring, the validity of the trial, and the main implications of the trial results.
"We hope that these statements will serve as a reference to practising physicians, researchers, and clinical associations in guiding the interpretation of the BEST TRIP results and mitigating what one committee member termed 'collateral damage' from its misunderstanding. Further consensus recommendations are presently in preparation regarding clinical and research approaches to ICP monitoring and treatment in light of current data," the authors conclude.
"This brief but eloquent consensus report helps revisit the overall implications and interpretations derived from the BEST TRIP trial," says John T. Povlishock, PhD, Editor-in-Chief of Journal of Neurotrauma and Professor, Medical College of Virginia Campus of Virginia Commonwealth University, Richmond. "As noted by the authors, this consensus document reframes many of the controversial issues generated in this initial trial, calling for a more critical evaluation of the study and its overall interpretation. From my perspective as Editor-in-Chief, I believe the consensus positions detailed therein help place this trial in the appropriate intellectual framework, while highlighting the continued need for more rigorous evaluation of intracranial hypertension, its monitoring, and its implications for traumatically brain-injured patients."
Source: Mary Ann Liebert, Inc., Publishers
Image credit: U.S. National Library of Medicine
References:
Chesnut R et al. (2015) A Consensus-Based Interpretation of the Benchmark Evidence from South American Trials: Treatment of Intracranial Pressure Trial. Journal of Neurotrauma. Published online August 31,
2015. doi: 10.1089/neu.2015.3976
Latest Articles
healthmanagement, trauma, brain injury, neurology, CT, TBI, computed tomography
Seven consensus statements from a panel of leading international experts in the treatment of severe traumatic brain injury (sTBI) provide a clear interpretation of the results from the BEST TRIP trial, which have led to ongoing debate over the value of in