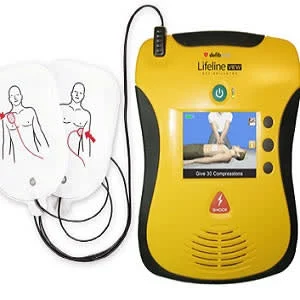
Current resuscitation guidelines recommend that bystander defibrillation be attempted as soon as an automated external defibrillator (AED) is available. Moreover, a new systematic review finds evidence supporting the association between bystander AED use and improved clinical outcomes in out-of-hospital cardiac arrest (OHCA), although the review team says the quality of evidence was low to very low.
"High-quality studies are warranted to confirm these findings, particularly to understand the role of AEDs in patients with non-shockable rhythms which constitutes the majority of OHCA," according to the systematic review and meta-analysis published in the journal Resuscitation.
In about one quarter of cases, patients with OHCA present with an initial shockable rhythm, i.e., ventricular fibrillation (VF) or pulseless ventricular tachycardia (pVT). This patient group is primarily dependent on high-quality CPR, identification of reversible causes, and rapid transport to advanced hospital care. AEDs allow bystanders not trained in rhythm interpretation to provide early defibrillation prior to emergency medical services arrival. However, AED retrieval, AED application, and rhythm analysis could result in delayed or interrupted CPR thereby potentially reducing the likelihood of survival.
In this review, researchers assessed observational and randomised studies comparing bystander AED use to no AED use in regard to clinical outcomes in OHCA, and to provide a descriptive summary of studies on the cost-effectiveness of bystander AED use. They searched Medline, Embase, the Web of Science, and the Cochrane Library for relevant studies published before 1 June 2017. Meta-analyses were performed for patients with all rhythms, shockable rhythms, and non-shockable rhythms.
In total, 44 observational studies, 3 randomised trials, and 13 cost-effectiveness studies were included. AED use was associated with survival when used in patients with all rhythms (OR: 1.73; 95% CI: 1.36, 2.18) and in those with a shockable rhythm (1.66; 95% CI: 1.54, 1.79). There was no association between bystander AED use and neurological outcome for non-shockable rhythms (OR: 0.76 [95% CI: 0.10, 5.87]). The quality of evidence was low for randomised trials and very low for observational studies.
In addition, AEDs were cost-effective in settings with high cardiac arrest incidence, with most studies reporting ratios < $100,000 per quality-adjusted life years. The researchers explain:
"Although some studies explicitly aimed to quantify a 'high incidence', the robustness and generalisability of these findings are unclear. Assumptions regarding the effectiveness of AED use varied substantially across studies. While some of this heterogeneity might be explained by various settings and patient populations, analytical differences and use of potentially biased estimates obtained from observational studies could also be important factors. Only one study used estimates from a randomised clinical trial."
The American Heart Association (AHA) and the European Resuscitation Council (ERC) support allocation strategies to improve AED resuscitation. The AHA 2015 guidelines recommend that AED programmes are “. . . implemented in public locations where there is a relatively high likelihood of witnessed cardiac arrest (e.g., airports, casinos, sports facilities)“, while the ERC has indicated that AED placement in areas with at least one cardiac arrest per five years can be considered cost-effective.
Source: Resuscitation
Image Credit: Axxenpoint
References:
Holmberg, Mathias J. et al. (2017) Bystander automated external defibrillator use and clinical outcomes after out-of-hospital cardiac arrest: A systematic review and meta-analysis. Resuscitation. doi.org/10.1016/j.resuscitation.2017.09.003
Latest Articles
out-of-hospital cardiac arrest, automated external defibrillator, bystander defibrillation, bystander AED
Current resuscitation guidelines recommend that bystander defibrillation be attempted as soon as an automated external defibrillator (AED) is available. Moreover, a new systematic review finds evidence supporting the association between bystander AED use