Masimo Announces FDA 510(k) Clearance for Root® with Noninvasive Blood
- ICU
- 07/03/2016
Masimo Announces FDA 510(k) Clearance for Root® with Noninvasive Blood Pressure and Temperature
READ MORE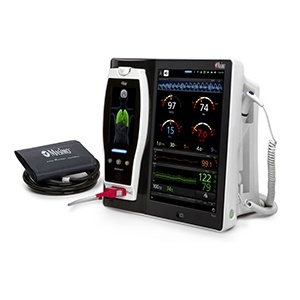
Masimo Announces FDA 510(k) Clearance for Root® with Noninvasive Blood Pressure and Temperature
READ MORE
Evaluation of a central catheter maintenance bundle developed to prevent central line-associated bloodstream infections (CLABSIs) found that it led...
READ MORE
Use of starch for fluid resuscitation was suspended by European and U.S. regulators following publication in the New England Journal of Medicine...
READ MORE
A critical care resuscitation unit (CCRU) at the University of Maryland Medical Center (UMMC) has significantly improved transfer times for non-trauma
READ MORE
Monitoring patients at risk of deterioration 3 times a day instead of twice a day did not significantly reduce the proportion of clinical deterioration...
READ MORE
Prof. Jean-Louis Vincent highlights integration and personalisation as key themes in the future of intensive care medicine, writing in Critical Care...
READ MORE
ResMed to Acquire Brightree® for $800 Million
READ MORE
Combined 13th Annual Critical Care Symposium and CCM-L meeting
READ MORE
Masimo's rainbow® Acoustic Monitoring was Associated with Fewer Alarm Events in New Clinical Study
READ MORE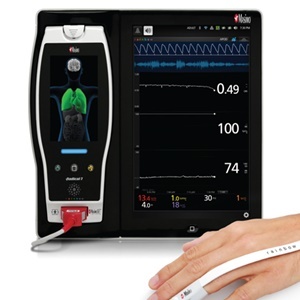
Masimo’s Oxygen Reserve Index Helps Clinicians Detect Impending Desaturation in Pediatric Patients
READ MORE