
PRINT OPTIMISED
In this article, I highlight that the most important thing intensive care physicians should stop doing is ignoring that they are prone to several cognitive biases. I will first support my statement by looking for conceptual caveats and cognitive bias in routine intensive care unit (ICU) care, and then move to specific patient and structural problems.
Intensive care is an interesting specialty. From all the early excitement in the 1970s, passing through two decades of intensive physiological use at the bedside, intensive care landed on the rough ground of modern randomised controlled trials (RCTs) in the late 1990s and early 2000s. The increasing number of critically ill patients coupled with new monitoring devices and important funding both from governmental and private agencies (including pharmaceutical companies) fostered research. In the early 2000s, the panorama looked promising, with positive trials coming out on a frenetic basis (Bernard 2001; Rivers 2001). Regrettably, the initial enthusiasm was followed by a wave of negative results (Ranieri 2012; PRISM Investigators 2017). Many interventions that seemed promising in the early 2000s were sequentially disproved or proved to be harmful, which has been the basis for the rationale of limiting excesses of interventions and treatments in the critically ill, the so-called “doing less” (Singer 2006).
What are the conceptual caveats in routine ICU care we should stop doing?
This section could be summarised in one sentence: Obtain less (not more) data and reduce treatment exposure considering it part of the disease and not of the healing process. Do so because we are all prone to cognitive bias.
The first part of the sentence brings a concept that is well-known to experts in behavioural science: information overload (Bawden 2008). Excessive information is known to reduce accuracy and increase confidence in the decision-making process (Hall 2007). This association can have disastrous consequences for critically ill patients, worsening the performance of important acute decisions and making physicians less prone to notice their own mistakes.
As the heart might be responsible for generating its own afterload, intensivists are also partially responsible for generating their own information overload. Examples include excessive use of haemodynamic monitoring in otherwise stable patients, pleiads of routine laboratory and imaging tests and inputs from several colleagues and healthcare workers (Manor-Shulman 2008). In the eagerness of having a quick diagnosis and treatment, intensivists generate data that will only aggravate the problem. Coupled with the increasing difficulty in accessing patient´s data due to poorly designed electronic health records, this creates an intensivist that has both information overload and information anxiety; that is, an individual exposed to too much data and that has trouble trying to access it (even the parts that indeed matter!). This results in a nightmare that is well known by most of us. A vignette is shown in Figure 1.
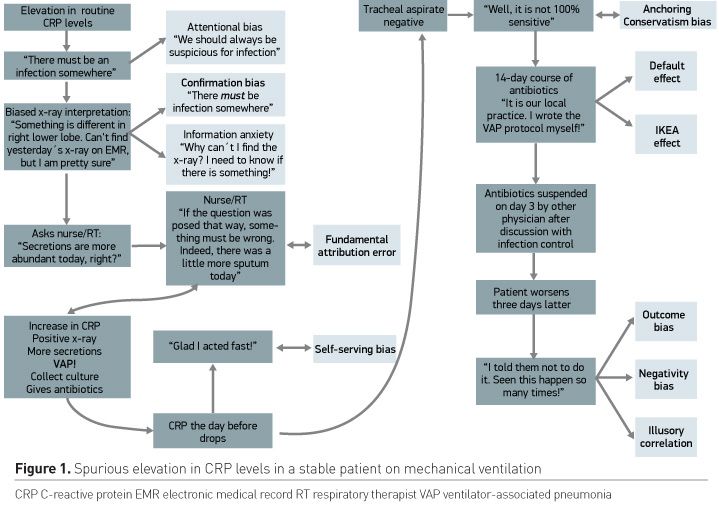
In the left part of this example, a series of types of cognitive bias occurred, triggered by a spurious elevation in C-reactive protein (CRP) levels that were routinely collected. Due to concerns of an untreated infection, the physician tries to find something that suits his/her keenness to explain the laboratory finding and somehow slack his fear of negligence. Secure physicians would probably ignore (or would not even have ordered) CRP levels and would choose a “wait-and-see” approach (Hranjec 2012). However, some physicians would embark on a destructive cycle of cognitive bias aiming at confirming their hypothesis. A similar scenario is conceivable for an apparently stable patient, who presents with an elevated lactate level, low central venous oxygen saturation etc. The problem, therefore, is the attempt to contextualise excessive information inside an otherwise unremarkable situation.
It may be stated that simply collecting less data is a childish suggestion and that all efforts should be made to use more data to improve treatment. I beg to disagree. A probabilistic interpretation of data is well suited in complex scenarios when we are accustomed to information (this applies to most Bayesian inference done in medicine); however, when data is new, time is short, and a decision is crucial, approaches that minimise choices based on less data may outperform complex models (Hardman 2003). This applies to many busy strained ICUs around the world.
Now let´s move to the right part of Figure 1. Damage has been done and our patient with a spurious irrelevant CRP elevation now has a ventilator-associated pneumonia (VAP) diagnosis. VAP has a doubtful attributable mortality but appears to be associated with prolonged mechanical ventilation and, obviously, higher costs (Melsen 2013). A VAP diagnosis leads to serious developments, such as antibiotic exposure, family distress (“Now, above all, he has a pneumonia!”) and even administrative issues (billing and benchmarking). Cognitive bias will not stop there. Despite evidence that guiding antibiotic time using CRP and/or procalcitonin levels are appropriate (de Jong 2016), the physician may now choose to embrace a conservative approach and apply a whole two-week course of antibiotics (the default effect). The same physician that relied on CRP to diagnose VAP is now shaky to stop antibiotics when CRP drops. However, if CRP dares to rise again in the following 48 hours, it is inevitable that concerns about “treatment failure” will arise and the circle of overtreatment will prevail. If physicians would consider that treatment is part of the disease and not an indissociable part of recovery, maybe the pros and cons would favour the first. In fact, using our infection vignette as an example, it is estimated that up to 20% of all patients receiving antibiotics will develop a serious adverse reaction (Tamma 2017). Maybe net benefit would be negative in our vignette?
The right side of the figure continues with a well-known sequence of cognitive biases that preclude proper patient management. The physician becomes emotionally tied to the diagnosis, knotted to the VAP protocol he wrote for the ICU (the “IKEA effect”, Norton 2012) and will fail to see evidence contrary to his hypothesis. If antibiotics are withdrawn in the next days by other physician and the patient eventually worsens, this will only further close the book on cognitive bias. While I used VAP for this example, the reader might find it suitable for many haemodynamic interventions (including the fluid bolus-diuretics conundrum, cardiac output measurements, etc).
Moving to the patient level
After the vignette, I hope that the reader considers that a more pragmatic approach to intensive care may be desirable. The cornerstone is transposing one of the Orwellian rules of writing to the ICU: “If it is possible to cut a word out, always cut it out” (Orwell 2013). Let´s replace “word” for “treatment” or “practice” and see what we can do. Examples are shown in Table 1.
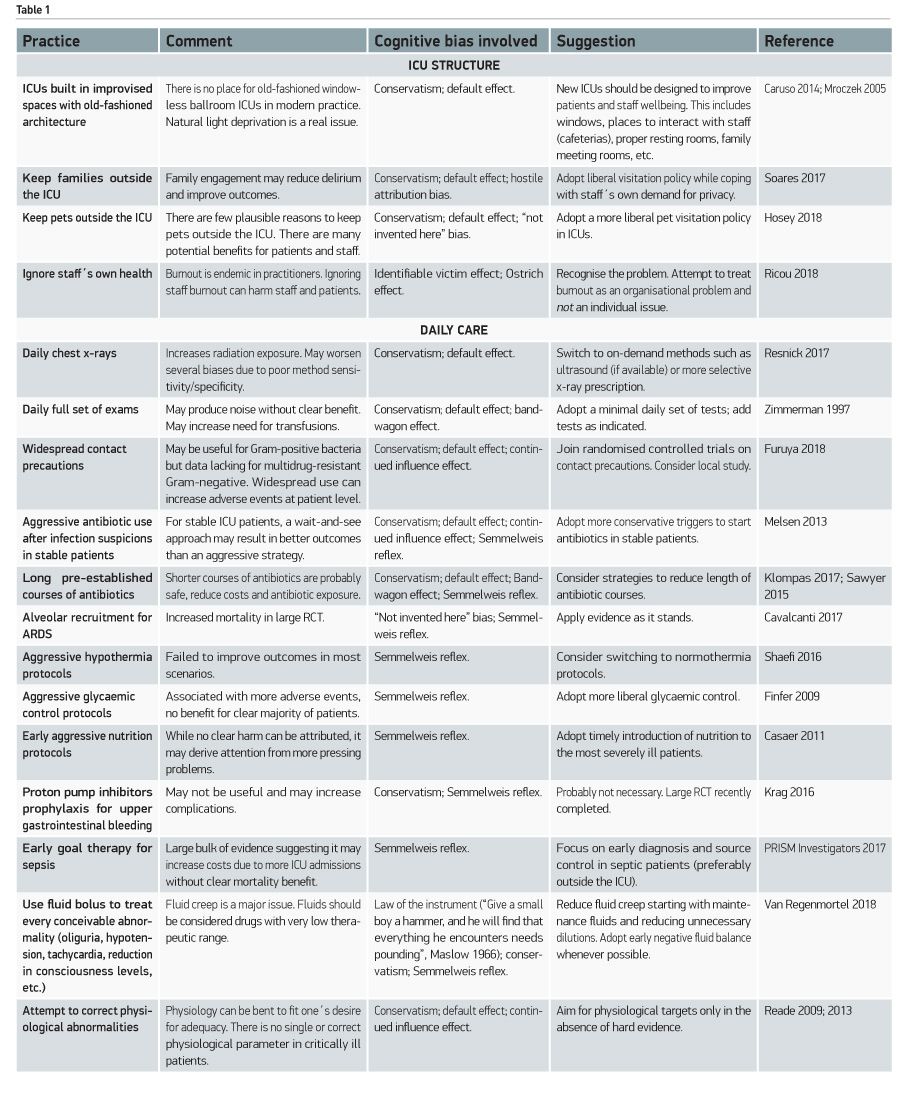
For each intervention, procedure and treatment shown in Table 1, one cognitive bias will have to be overthrown. This is not an easy process, since most of the teaching in medicine is indeed based on passing bias and abstract concepts from generation to generation. It is commonplace to hear that we should aim to keep a patient “normovolaemic”, “well-nourished”, etc., while it remains underappreciated that these terms are closer to a linguistic trick than to a medical practice. The first step to embrace a modern ICU is understanding that much of what we did and believed was part of habit and not science. This is the very reason why conservatism, Semmelweis reflex (Leary and Wilson 1991) and default effect are the most frequent cognitive bias shown in Table 1.
In the dawn of civilization in ancient Eridu, architects were more interested in rebuilding structures from scratch than preserving previous buildings. The Eridu fortress was rebuilt eleven times. As Paul Kriwaczek stated, ancient Eridu habitants were impatient with what was old and receptive to the new (Kriwaczek 2012). Intensive care should remember its roots but allow the new to be built upon its ground.
Abbreviations
CRP C-reactive protein
ICU intensive care unit
VAP ventilator-associated pneumonia
References:
Bawden D, Robinson L (2008) The dark side of information: overload, anxiety and other paradoxes and pathologies. J Inf Sci, 35(2): 180-91.
Bernard GR, Vincent JL, Laterre PF et al; Recombinant human protein C Worldwide Evaluation in Severe Sepsis (PROWESS) study group (2001) Efficacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med, 344(10): 699-709.
Caruso P, Guardian L, Tiengo T et al. (2014) ICU architectural design affects the delirium prevalence: a comparison between single-bed and multibed rooms. Crit Care Med, 42(10): 2204-10.
Casaer MP, Mesotten D, Hermans G et al. (2011) Early versus late parenteral nutrition in critically ill adults. N Engl J Med, 365(6): 506-17.
de Jong E, van Oers JA, Beishuizen A et al. (2016) Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis, 16(7): 819-27.
Furuya EY, Cohen B, Jia H et al. (2018) Long-term impact of universal contact precautions on rates of multidrug-resistant organisms in ICUs: a comparative effectiveness study. Infect Control Hosp Epidemiol, 39(5): 534-40.
Hall CC, Ariss L, Todorov A (2007) The illusion of knowledge: when more information reduces accuracy and increases confidence. Organ Behav Hum Decis Process, 103(2): 277-90.
Hosey MM, Jaskulski J, Wegener ST et al. (2018) Animal-assisted intervention in the ICU: a tool for humanization. Crit Care, 22(1): 22.
Hranjec T, Rosenberger LH, Swenson B et al. (2012) Aggressive versus conservative initiation of antimicrobial treatment in critically ill surgical patients with suspected intensive-care-unit-acquired infection: a quasi-experimental, before and after observational cohort study. Lancet Infect Dis, 12(10): 774-80.
Klompas M, Li L, Menchaca JT et al; Centers for Disease Control and Prevention Epicenters Program (2017) Ultra-short-course antibiotics for patients with suspected ventilator-associated pneumonia but minimal and stable ventilator settings. Clin Infect Dis, 64(7): 870-6.
Krag M, Perner A, Wetterslev J et al; SUP-ICU investigators (2016) Stress ulcer prophylaxis with a proton pump inhibitor versus placebo in critically ill patients (SUP-ICU trial): study protocol for a randomised controlled trial. Trials, 17(1): 205.
Kriwaczek P (2012) Babylon: Mesopotamia and the birth of civilization. New York: St. Martin's Griffin.
Leary T, Wilson RA (1991) The Game of Life. Phoenix, AZ: New Falcon Publications 1991.
Manor-Shulman O, Beyene J, Frndova H et al. (2008) Quantifying the volume of documented clinical information in critical illness. J Crit Care, 23(2): 245-50.
Martignon L, Vitouch O, Takezawa M et al. (2003) Naive and yet enlightened: from natural frequencies to fast and frugal decision trees. In: Hardman D, Macchi L, eds. Thinking: psychological perspectives on reasoning. Chichester: John Wiley, pp.189-2011.
Maslow AH (1966) The psychology of science. New York: Harper & Row, p. 15.
Melsen WG, Rovers MM, Groenwold RH et al. (2013) Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis, 13(8): 665-71.
Mroczek J, Mikitarian G, Vieira EK et al. (2005) Hospital design and staff perceptions: an exploratory analysis. Health Care Manag (Frederick), 24(3): 233-44.
NICE-SUGAR Study Investigators, Finfer S, Chittock DR et al. (2009) Intensive versus conventional glucose control in critically ill patients. N Engl J Med, 360(13): 1283-97.
Norton MI, Mochon D, Ariely D (2012) The IKEA effect: when labor leads to love. J Consum Psychol, 22(3): 453-60.
Orwell G (2013) Politics and the English language. London: Penguin.
PRISM Investigators, Rowan KM, Angus DC et al. (2017) Early, goal-directed therapy for septic shock - a patient-level meta-analysis. N Engl J Med, 376(23): 2223-34.
Ranieri VM, Thompson BT, Barie PS et al; PROWESS-SHOCK Study Group (2012) Drotrecogin alfa (activated) in adults with septic shock. N Engl J Med, 366(22): 2055-64.
Reade MC (2009) Should we question if something works just because we don’t know how it works? Crit Care Resusc, 11(4): 235-6.
Reade MC (2013) The pursuit of oxygen euboxia. Anaesth Intensive Care, 41(4): 453-5.
Resnick S, Inaba K, Karamanos E et al. (2017) Clinical relevance of the routine daily chest X-Ray in the surgical intensive care unit. Am J Surg, 214(1): 19-23.
Ricou B, Zampieri FG, Brown SM (2018) Should all ICU clinicians regularly be tested for burnout? No. Intensive Care Med, May 7. doi: 10.1007/s00134-018-5139-3.
Rivers E, Nguyen B, Havstad S et al; Early Goal-Directed Therapy Collaborative Group (2001) Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med, 345(19): 1368-77.
Sawyer RG, Claridge JA, Nathens AB et al; STOP-IT Trial Investigators (2015) Trial of short-course antimicrobial therapy for intraabdominal infection. N Engl J Med, 372(21): 1996-2005.
Shaefi S, Mittel AM, Hyam JA et al. (2016) Hypothermia for severe traumatic brain injury in adults: Recent lessons from randomized controlled trials. Surg Neurol Int, 7: 103.
Singer M (2006) The key advance in the treatment of sepsis in the last 10 years...doing less. Crit Care, 10(1): 122.
Soares M, Silva UV, Homena WS Jr et al; ORCHESTRA (ORganizational CHaractEeriSTics in cRitcal cAre) Study Investigators (2017) Family care, visiting policies, ICU performance, and efficiency in resource use: insights from the ORCHESTRA study. Intensive Care Med, 43(4): 590-1.
Tamma PD, Avdic E, Li DX et al. (2017) Association of adverse events with antibiotic use in hospitalized patients. JAMA Intern Med, 177(9): 1308-15.
Van Regenmortel N, Verbrugghe W, Roelant E et al. (2018) Maintenance fluid therapy and fluid creep impose more significant fluid, sodium, and chloride burdens than resuscitation fluids in critically ill patients: a retrospective study in a tertiary mixed ICU population. Intensive Care Med, 44(4): 409-17.
Writing Group for the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART) Investigators, Cavalcanti AB, Suzumura ÉA et al. (2017) Effect of lung recruitment and titrated positive end-expiratory pressure (PEEP) vs low PEEP on mortality in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA, 318(14): 1335-45.
Zimmerman JE, Seneff MG, Sun X et al. (1997) Evaluating laboratory usage in the intensive care unit: patient and institutional characteristics that influence frequency of blood sampling. Crit Care Med, 25(5): 737-48.









