The role of speech and language therapists (SLTs) in critical care can be unclear so this article sets out the scope of practice to increase awareness of the value of SLTs as part of the wider multidisciplinary team.
Speech and language therapists (SLT) are trained to deliver specialist clinical services to adults with a broad range of disorders, including hearing impairment, motor speech disorders (dysarthria, dyspraxia), acquired language disorders (dysphasia), voice problems (dysphonia), and swallowing problems (oropharyngeal dysphagia [OD]). These are often linked to an acquired neurological disorder, such as stroke, traumatic brain injury, Parkinson’s disease and dementia, although other acute conditions may affect these functions, especially when linked to respiratory dysfunction requiring tracheostomy and ventilation. The role of SLT within critical care has increased as patients with complex impairments are sedated less and experience communication and swallowing impairments. These functions are frequently seen as a return to normality during the process of recovery and a measure of quality of life (Segaran 2006; Carroll 2007; Karlsson et al. 2012; Engstrom et al. 2013). There are few UK-based studies evaluating SLT interventions in critical care; however, as the number of National Institute for Health Research fellowships for Allied Health Professionals (AHP) increases it is hoped that this will generate the research needed to provide evidenced-based therapy.
You might also like:ICU-related dysphagiaNormal swallowing and speech functions
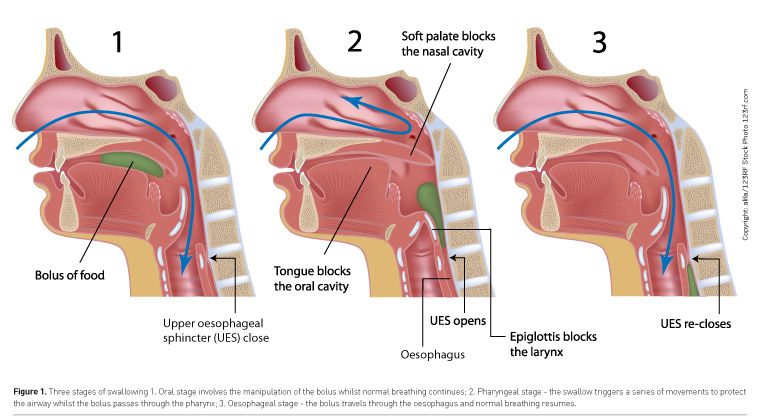
Swallowing employs the same range of muscles and nerves as those required for breathing and speaking, namely the pharynx, larynx, tongue and lips. The swallow is described in three phases (Groher and Crary 2015), although these are interlinked and dependent on each other to be effective (Figure 1). The oral phase requires volitional control of a bolus, either food or fluid, in preparation for swallowing; this includes chewing food using the full range of tongue and jaw movements and holding the food within the oral cavity using lip closure. The tongue then pushes this bolus posteriorly into the pharynx, triggering a series of reflexive movements to move the laryngeal structure vertically and anteriorly. These biomechanical movements are essential for a three-way closure of the glottis, to protect the airway from penetration—true and false vocal cords adduct and the epiglottis provides additional cover and redirection of the bolus towards the oesophageal entrance, which opens biomechanically with vertical and anterior laryngeal movement. It is for this reason that you may see an SLT assessing swallowing through midline palpation of the larynx to determine swallow timing and range of hyolaryngeal movement to indicate swallow completion. The adjacency of the airway next to the oesophageal entrance means that any mistiming or obstruction of the swallow movement can result in the entrance of food or fluid directly into the airway. The cough reflex is a strong airway-protective mechanism designed to expectorate foreign bodies. However, in patients who are intubated, these reflexes are often absent (Kallesen et al. 2016), increasing the risk of aspiration. If overt signs of coughing are absent, this is described as silent aspiration. To fully understand the breakdown and remediate the problem requires early recognition of risk factors and assessments sensitive to dysphagia. Bedside evaluation cannot always determine the effectiveness of pharyngeal clearance or airway protection.
Dysphagia management
Oropharyngeal dysphagia is often the primary reason for SLT referral in ICU, with its recognised link to increased risk of aspiration pneumonia and subsequent mortality (Altman et al. 2010). At the acute stage this is mainly related to effective oral secretion management, as patients who swallow infrequently or ineffectively risk aspirating saliva with a high bacterial load. This increases the risk of ventilator-associated pneumonia, so SLTs often deal with optimising oral hygiene and hydration in the early stages before fluid and food trials can be evaluated. Although early identification and management of dysphagia is recommended to reduce risk, access to SLT can be limited due to low staffing and lack of specialist skills. SLTs are often viewed as peripatetic services to ICU with referrals made by the medical team following a process of screening for swallowing difficulties by nursing staff (Cichero et al. 2009). A swallow screen should include an evaluation of oral motor functions, such as lip and tongue movements, voice, cough and swallow trials with varied consistencies to evaluate timing of swallowing and abnormal responses to food and drink. Currently there are no validated swallow screening tools for ICU patients; existing tools have been developed for use with stroke patients (Trapl et al. 2007; Martino et al. 2009; Edmiaston et al. 2010) and may be unreliable.
For patients requiring tracheostomy and ventilation both motor and sensory laryngeal function can be altered following intubation (Skoretz et al. 2010; Macht et al. 2011; Moraes et al. 2013), and disruption to normal breath-swallow synchrony can affect timing of swallowing (Martin-Harris et al. 2005; Terzi et al. 2007; Nishino 2012), leading to an increased risk of silent aspiration. A number of studies have reported OD as a feature of non-neurological patient subgroups, such as sepsis (Zielske et al. 2014), acute lung injury (Brodsky et al. 2014), ARDS (Brodsky et al. 2016), cardiac surgery (Daly et al. 2016), critical illness polyneuropathy (Ponfick et al. 2015) and spinal cord injury (Shem et al. 2012), suggesting a respiratory link. These impairments cannot be easily detected by screening tools so instrumental assessments of swallowing are recommended, namely videofluoroscopy (VFS) and fibreoptic endoscopic evaluation of swallowing (FEES). These are employed by SLTs with specialist competency training (Royal College of Speech and Language Therapists 2013; 2015) and provide different information on swallowing functions.
The VFS examination takes place in a radiology department and requires transfer of the medically stable patient. During the examination, a patient is sat upright and required to eat and drink a range of food textures coated with barium sulphate contrast that are video x-rayed to identify timing, efficiency and safety of swallowing across all phases (Logemann 1993). Aspiration of this material can cause harm, so this examination may need to be modified for patients requiring ventilation and is not suitable for those who need to remain supine or are haemodynamically unstable. FEES is a more accessible assessment (Langmore 2001) that can be undertaken at the bedside within the ICU using a video-nasendoscope. It provides a direct view of the pharynx and larynx only and helps to identify structural impairments and physiological responses to secretions and food trials, in terms of swallow timing, pharyngeal clearance and airway protection. Although aspiration is the key indicator of swallow safety, the role of the SLT is to identify the cause of breakdown and to trial strategies aimed at reducing risk of aspiration in order to facilitate safe oral intake. A number of studies have employed FEES to identify OD in post-extubation patients (Leder et al. 1998; Hafner et al. 2008; Scheel et al. 2015). For scenarios where swallow safety is at high risk and airway protection is limited, a decision may be made to keep a patient nil by mouth and optimise nutrition through non-oral routes, after discussion with the wider team.
SLTs are trained to plan and deliver a number of therapeutic interventions to manage the impairments that cause OD (Martino and McCulloch 2016). These can target an identified impairment, such as reduced hyolaryngeal elevation, through strengthening exercises or strategies. Treatment approaches include exercises to increase range and strength of lip and tongue movements, including resistance exercises. Voice work can be employed to increase laryngeal lift and vocal cord adduction and a number of swallow strategies have been developed to target specific impairments (Logemann 2006), such as the Shaker, Masako and Mendelsohn manoeuvres, Effortful swallow and Facial Oral Tract Therapy (Hansen and Jakobsen 2010). Alternatively, the risk of aspiration can be reduced through compensatory approaches that do not remediate the swallow problem. This approach is suitable for patients with cognitive or language problems who are unable to comply with exercises. The use of thickened fluids can be employed for patients who are unable to control fluids in the mouth or have a delayed swallow initiation. These impairments have to be verified on instrumental assessment as inappropriate use of thickener can have negative consequences (Cichero 2013). Similarly, diet modifications, such as purée or soft mashed food have to be used in relation to the oral and pharyngeal swallow impairment and their use monitored.
Communication
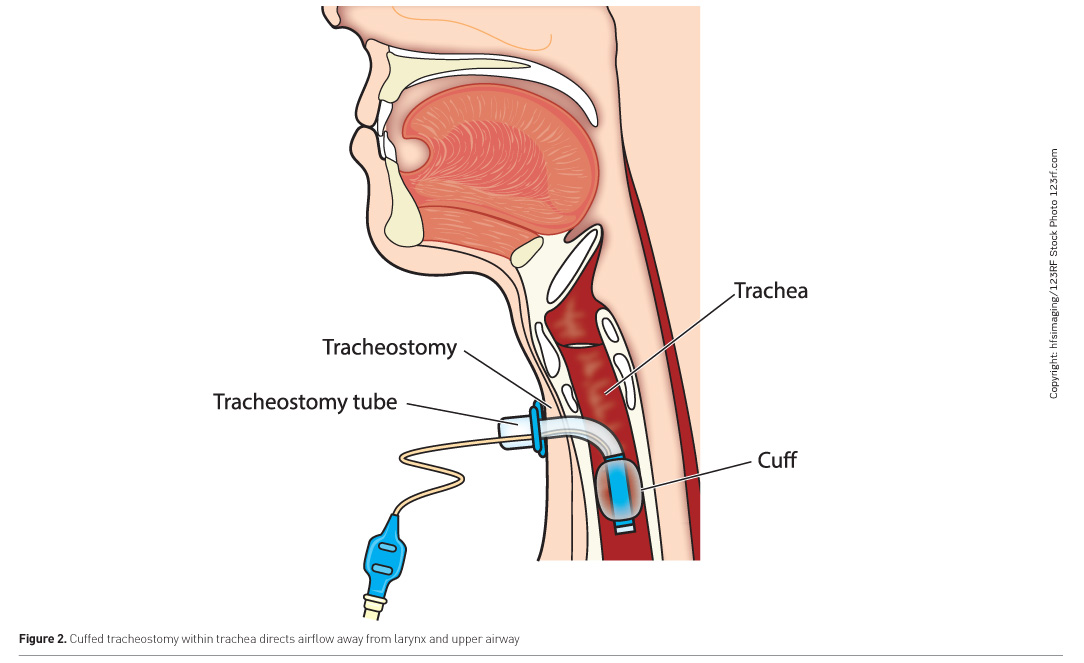
The ability to communicate and interact with their environment is a key concern for many patients in critical care, especially during intubation and ventilation whilst awake (Karlsson et al. 2012; Guttormson et al. 2015). This poses challenges for staff interaction and the verification of capacity (Wojnicki-Johansson 2001). A number of alternative methods are available, which range from low-technology aids, such as picture or alphabet charts (Radtke et al. 2011) to high-technology systems such as eye-gaze systems, although individual assessment is required as one device may not be suitable for all patients (ten Hoorn et al. 2016). The most effective method of communication is speech, and for those being ventilated via a tracheostomy this can only be achieved when the cuff is deflated (Figure 2). This can be done gradually as part of a ventilator weaning process termed laryngeal weaning, using collaborative team-working to include SLTs and respiratory physiotherapists to evaluate respiratory and laryngeal functions. This differs from respiratory weaning whereby the cuff remains inflated whilst the patient is trained to self-ventilate. A short-term alternative is a method termed above-cuff vocalisation (ACV) (McGrath et al. 2015), which uses an external air source through a subglottic suction port to achieve phonation whilst the tracheostomy cuff remains inflated. This has also been found to benefit laryngeal functions for swallowing (McGrath et al. 2018).
Supporting tracheostomy weaning
Guidance set out by the Royal College of Speech and Language Therapists (2014) identified SLTs as being integral to the multidisciplinary environment of critical care and involved in the weaning process for tracheostomy and ventilator-dependent patients in addition to the rehabilitation of swallowing and communication difficulties. These needs were highlighted in the National Institute for Health and Care Excellence (NICE) guidance for rehabilitation after critical illness (2009), although a national audit of tracheostomy care (National Confidential Enquiry into Patient Outcomes and Death 2014) found delays to SLT referral for tracheostomy patients and a lack of access to FEES procedures to support clinical decision-making.
Conclusion
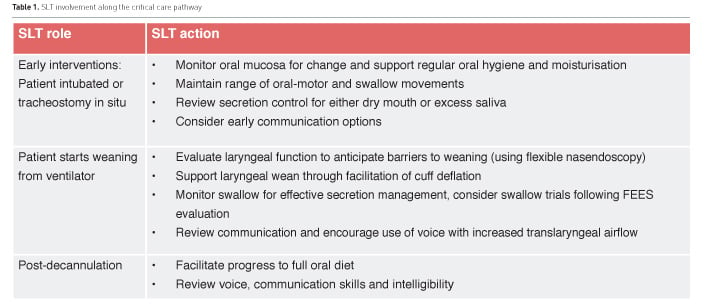
SLTs are considered a key member of many teams, linking up with physiotherapy, occupational therapy, nursing care, pharmacy and dietetics to deliver early therapeutic interventions for speech and swallowing. Early interventions will translate to prevention of complications and potential reduction in length of stay (Table 1). SLTs also add professional value by contributing to team decision-making and rehabilitation goals throughout the patient’s pathway, in line with Quality Standard 158 (National Institute for Health and Care Excellence 2017), especially as they often work across wards and may follow the patient through their rehabilitation and into the community. A current challenge is the lack of directly-funded SLT services in ICU despite support from the guidelines for the provision of intensive care services (GPICS) (Faculty of Intensive Care Medicine and Intensive Care Society 2015). This limits both clinical involvement and professional development within teams that contribute to changing culture and practice.
There is currently no post-registration training programme for critical care skills so SLTs require clinical experience alongside other team members to develop their competencies, with a minimum banding level of band 7 with senior support or band 8a if sole clinician (
nhsemployers.org/your-workforce/pay-and-reward/agenda-for-change/pay-scales). As this is a small professional group an online forum has been set up to provide support, share practice and problem-solve about situations specific to critical care. Education is provided through Clinical Excellence Networks (CEN) for tracheostomy, dysphagia and FEES/VFS. A new Twitter group,
@CriticalCareSLT links SLTs to the wider critical care world with responsive interactions, strategic discussions and an opportunity to share examples of practice which raise awareness of our role.
Conflict of interest
Jackie McRae declares that she has no conflict of interest.
Abbreviations
FEES fibreoptic endoscopic evaluation of swallowing
ICU intensive care unit
OD oropharyngeal dysphagia
SLT speech and language therapists
VFS videofluoroscopy
Altman
KW, Yu GP, Schaefer SD (2010) Consequence of dysphagia in the hospitalized
patient: impact on prognosis and hospital resources. Arch Otolaryngol Head Neck
Surg, 136(8): 784-9.
Brodsky MB, Gellar JE, Dinglas VD et al. (2014)
Duration of oral endotracheal intubation is associated with dysphagia symptoms
in acute lung injury patients. J Crit Care, 29(4): 574-9.
Brodsky MB, Huang M, Shanholtz C et al. (2016)
Recovery of dysphagia symptoms after oral endotracheal intubation in ARDS
survivors: a 5-year longitudinal study. Ann Am Thorac Soc, 14(3): 376-83.
Carroll SM (2007) Silent, slow lifeworld: the
communication experience of nonvocal ventilated patients. Qual Health Res,
17(9): 1165-77.
Cichero JAY (2013) Thickening agents used for
dysphagia management: effect on bioavailability of water, medication and
feelings of satiety. Nutr J, 12: 54.
Cichero JAY, Heaton S, Bassett L (2009) Triaging
dysphagia: nurse screening for dysphagia in an acute hospital. J Clin Nurs,
18(11): 1649-59.
Daly E, Miles A, Scott S et al. (2016) Finding the red
flags: swallowing difficulties after cardiac surgery in patients with prolonged
intubation. J Crit Care. 31(1): 119-24.
Edmiaston J, Connor LT, Loehr L et al. (2010)
Validation of a dysphagia screening tool in acute stroke patients. Am J Crit
Care. 19(4): 357-64.
Engstrom A, Nystrom N, Sundelin G et al. (2013)
People's experiences of being mechanically ventilated in an ICU: a qualitative
study. Intensive Crit Care Nurs, 29(2): 88-95.
Faculty of Intensive Care Medicine and Intensive Care
Society (2015) Guidelines for provision of intensive care services. London:
FICM & ICS. [Accessed: 21 April 2015] Available from ficm.ac.uk/standards-and-guidelines/gpics
Groher ME, Crary MA (2015) Dysphagia: clinical
management in adults and children. 2nd ed. St Louis: Mosby.
Guttormson JL, Bremer KL, Jones RM (2015) “Not being
able to talk was horrid”: a descriptive, correlational study of communication
during mechanical ventilation. Intensive Crit Care Nursing, 31(3): 179-86.
Hafner G, Neuhuber A, Hirtenfelder S et al. (2008)
Fiberoptic endoscopic evaluation of swallowing in intensive care unit patients.
Eur Arch Otorhinolaryngol, 265(4): 441-6.
Hansen TS, Jakobsen D (2010) A decision-algorithm defining
the rehabilitation approach: 'Facial oral tract therapy'. Disabil Rehabil,
32(17): 1447-60.
Kallesen M, Psirides A, Huckabee ML (2016) Comparison
of cough reflex testing with videoendoscopy in recently extubated intensive
care unit patients. J Crit Care, 33: 90-4.
Karlsson V, Bergbom I, Forsberg A. (2012) The
lived experience of adult intensive care patients who were conscious during
mechanical ventilation: a phenomenological-hermeneutic study. Intensive Crit
Care Nurs, 28: 6-15.
Langmore S (2001) Endoscopic evaluation and treatment
of swallowing disorders. New York: Thieme.
Leder SB, Cohn SM, Moller BA (1998) Fiberoptic
endoscopic documentation of the high incidence of aspiration following
extubation in critically ill trauma patients. Dysphagia, 13: 208–12.
Logemann J (2006) Medical and rehabilitative therapy
of oral, pharyngeal motor disorders. GI Motility Online, 16 May. doi:10.1038/gimo50.
Logemann JA (1993) Manual for the videofluorographic
study of swallowing. Austin, TX: Pro-Ed.
Macht M, Wimbish T, Clark BJ et al. (2011)
Postextubation dysphagia is persistent and associated with poor outcomes in
survivors of critical illness. Crit Care, 15(5): R231.
Martin-Harris B, Brodsky MB, Michel Y et al. (2005)
Breathing and swallowing dynamics across the adult lifespan. Arch Otolaryngol
Head Neck Surg, 131(9): 762-70.
Martino R, McCulloch T (2016) Therapeutic intervention
in oropharyngeal dysphagia. Nat Rev Gastroenterol Hepatol, 13: 665-79.
Martino R, Silver F, Teasell R et al. (2009) The
Toronto Bedside Swallowing Screening Test (TOR-BSST): development and
validation of a dysphagia screening tool for patients with stroke. Stroke, 40:
555-61.
McGrath B, Lynch J, Wilson M et al. (2015) Above cuff
vocalisation: a novel technique for communication in the ventilator-dependent
tracheostomy patient. J Intensive Care Soc, 17(1): 19-26.
McGrath BA, Wallace S, Wilson M et al. (2018) Safety
and feasibility of above cuff vocalisation for ventilator-dependant patients
with tracheostomies. J Intensive Care Soc, 28 Mar. doi: 10.1177/1751143718767055.
Moraes DP, Sassi FC, Mangilli LD et al. (2013)
Clinical prognostic indicators of dysphagia following prolonged orotracheal
intubation in ICU patients. Crit Care, 17: R243.
National Confidential Enquiry into Patient Outcomes
and Death (2014) On the right trach? A review of the care received by patients
who underwent a tracheostomy. London:
NCEPOD. [Accessed: 20 June 2014] Available from ncepod.org.uk/2014tc.html
National Institute for Health and Care Excellence (2017)
Rehabilitation after critical illness in adults: quality standard [QS158]. [Accessed:
17th September 2017] Available from nice.org.uk/guidance/qs158
National Institute for Health and Clinical Excellence (2009)
Rehabilitation after critical illness. (NICE clinical guideline 83). [Accessed:
1 May 2014] Available from nice.org.uk/guidance/cg83/evidence/full-guideline-242292349
Nishino T (2012) The swallowing reflex and its
significance as an airway defensive reflex. Front Physiol, 3: 489.
Ponfick M, Linden R, Nowak DA (2015) Dysphagia—a
common, transient symptom in critical illness polyneuropathy: a fiberoptic
endoscopic evaluation of swallowing study. Crit Care Med, 43: 365–72.
Radtke JV, Baumann
BM, Garrett KL et al. (2011) Listening to the voiceless patient: case reports
in assisted communication in the intensive care unit. J Palliat Med, 14(6):
791-5.
Royal College of Speech and Language Therapists (2013)
Videofluoroscopic evaluation of oropharyngeal swallowing function (VFS): The
role of speech and language therapists. RCSLT Position Paper 2013. London:
RCSLT. [Accessed: 25 April 2018] Available from rcslt.org/members/publications/publications2/videofluoroscopic_position_paper2013
Royal College of Speech and Language Therapists (2014)
Position paper: speech and language therapy in adult critical care. London: RCSLT.
[Accessed: 1 December 2014] Available from rcslt.org/members/publications/publications2/criticalcare_positionpaper_060114
Royal College of Speech and Language Therapists (2015)
Position paper: fibreoptic endoscopic evaluation of swallowing (FEES): the role
of speech and language therapy. London: RCSLT. [Accessed: 25 April 2018]
Available from rcslt.org/members/publications/publications2/fees_position_paper_300315
Scheel R, Pisegna JM, McNally E et al. (2015)
Endoscopic assessment of swallowing after prolonged intubation in the ICU
setting. Ann Otol Rhinol Laryngol, 125(1):
43-52.
Segaran E (2006) Returning to normal: the role of
eating in recovery from a critical illness. BJNN, 2(3): 141-8.
Shem K, Castillo K, Wong SL et al. (2012) Dysphagia
and respiratory care in individuals with tetraplegia: incidence, associated
factors, and preventable complications. Top Spinal Cord Inj Rehabil, 18(1): 15-22.
Skoretz SA, Flowers HL, Martino R (2010) The incidence
of dysphagia following endotracheal intubation: a systematic review. Chest, 137:
665-73.
ten Hoorn S, Elbers PW, Girbes AR, Tuinman PR. (2016)
Communicating with conscious and mechanically ventilated critically ill
patients: a systematic review. Crit Care, 20(1): 333.
Terzi N, Orlikowski D, Aegerter P et al. (2007)
Breathing–swallowing interaction in neuromuscular patients: a physiological
evaluation. Am J Respir Crit Care Med, 175(3): 269-76.
Trapl M, Enderle P, Nowotny M et al. (2007) Dysphagia
bedside screening for acute-stroke patients: the Gugging Swallowing Screen.
Stroke, 38(11): 2948-52.
Wojnicki-Johansson G (2001) Communication between
nurse and patient during ventilator treatment: patient reports and RN
evaluations. Intensive Crit Care Nurs, 17(1): 29-39.
Zielske J, Bohne S,
Brunkhorst FM et al. (2014) Acute and long‐term dysphagia in critically ill patients with severe
sepsis: results of a prospective controlled observational study. Eur Arch
Otorhinolaryngol, 271: 3085–93.