ICU Management & Practice, Volume 21 - Issue 3, 2021
Introduction
Severe acute respiratory syndrome coronavirus CoV-2 is the highly contagious viral agent responsible for the ongoing COVID-19 pandemic. COVID-19 is the name of the syndrome caused by SARS-CoV-2 infection. Common signs of infection include upper respiratory tract symptoms as well as more severe symptoms: thromboembolic events, pneumonia, ARDS and respiratory failure eventually leading to multi-organ failure (Allado et al. 2021). Acute hypoxic respiratory failure (AHRF) is the most dreaded COVID-19 complication commonly resulting in ICU admission. During the pandemic, the abrupt and increasingly high demand for intensive care was unmatched by struggling healthcare systems throughout the world. Fortunately, several patients with AHRF could also be treated with great results using non-invasive ventilation and conventional oxygen therapy in general wards or specifically created wards. Currently, thanks to massive worldwide vaccination campaigns, infections rates are decreasing, reducing pressure on ICUs and hospitals.
Clinical & Pathological Findings
Usual diagnostic criteria for severe SARS-CoV-2 infection include positive molecular swab, compatible symptoms, and imaging studies. CT scans of COVID-19 induced lung injury are mostly described as ground glass opacities and variable degree of honeycombing with areas of lung consolidation that do not always correlate with gas exchanges variations (Ball et al. 2021). Pathology reports describe congested lungs with focal fibrosis and interstitial oedema along with capillary congestion and thrombi in small arterial vessels plus dilated alveolar ducts spaces filled with hyaline membranes (Carsana et al. 2020). In advanced stages of the disease, fibrosis progresses resulting in airspace obliteration, lung fibrosis and remodelling (Grillo et al. 2021). Clinically, this translates to AHRF with patients experiencing tachypnoea, dyspnoea and eventually increased work of breathing (WOB) often requiring conventional oxygen therapy or more advanced forms of ventilatory support.
Monitoring: Understanding FiO2 SpO2 SaO2 PaO2
Supplemental oxygen is typically started for hypoxic patients with oxygen saturation <92% as measured by pulse-oximetry. However, SpO2 does not always correlate well with SaO2 nor with PaO2 values. In fact, given the sigmoid shape of the oxygen dissociation curve, SpO2 higher than 90% may correspond to a large range of PaO2 values (Tobin 2020). The mean difference and limits of agreement between SpO2 and true SaO2 were 25.8 + 16% with SpO2 readings in critically ill patients being less reliable, and reproducible than in healthy volunteers (Ottestad et al. 2018; Tobin et al. 2020). Arterial blood samples offer reliable measurements of gas exchanges that should be used to set a suitable course of treatment. Another factor that contributes to change in the oxygen dissociation curve is represented by an increase in body temperature that generates a right shift in the curve: for a given PaO2 value, SaO2 will be lower (Tobin et al. 2020). Although these changes produce significant variation in SaO2 and PaO2, values remain constant as tissues in the body respond only to PaO2 and not SaO2 (Tobin et al. 2020).
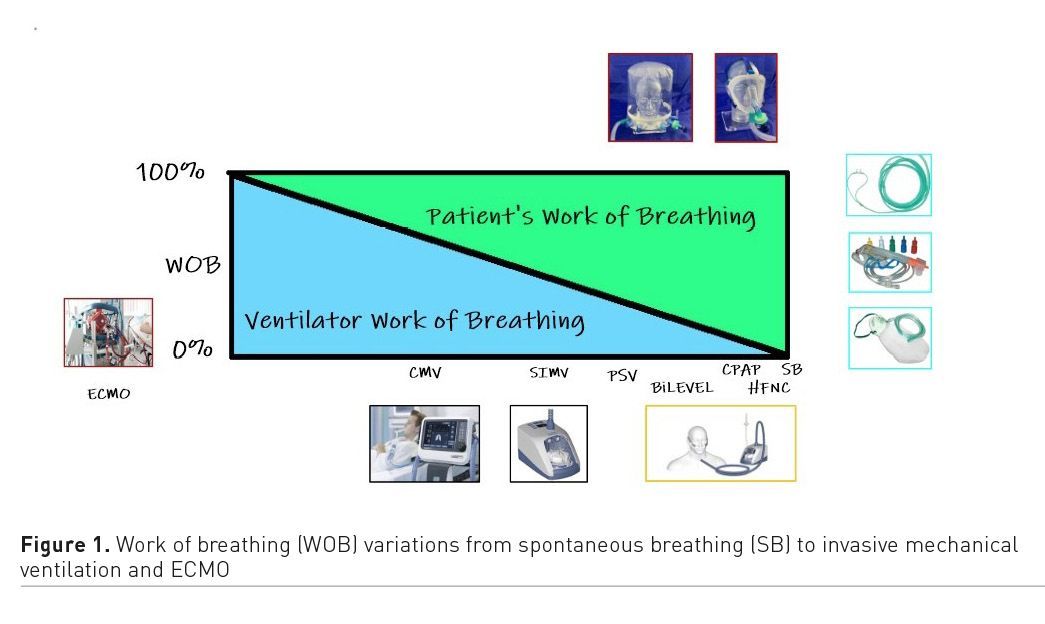 Respiratory Management and Considerations
Respiratory Management and Considerations
When the pandemic started, knowledge of the disease was scarce and indications for non-invasive ventilation (NIV) limited, resulting in relatively high intubation rates (Grasselli et al. 2020). As knowledge of the disease progressed and treatment options improved, we became aware that tachypnoea alone is not a justification for intubation because increased respiratory rate in COVID-19 patients represents a physiological response to lung inflammation (Tobin 2020), whereas work of breathing (WOB) is determined by the magnitude of swings in pleural pressure and tidal volume (Grieco et al. 2019). Evaluation of pulmonary infiltrates as a criterion for intubation is only valid when severely abnormal gas exchanges are present (Alhazzani et al. 2021). Furthermore, physiological response to values of PaO2 of around 60 mmHg is minimal dyspnoea and values higher than 40 mmHg have difficultly been associated with organ damage (Tobin 2020). Thus, hypoxaemia without dyspnoea or respiratory distress is not an indication for intubation without a prior attempt of non-invasive forms of oxygen therapy (Tobin 2020).
Oxygen therapy is suggested for values of SpO2 < 92% and is recommended if SpO2 <90%, but should be maintained no higher than 96% (Alhazzani et al. 2021). These targets can be reached by increasing oxygen delivery, the product of arterial oxygen content and cardiac output (Tobin 2020). Lower oxygen delivery is initially counterbalanced by higher oxygen extraction and normal consumption until reaching a critical threshold – decrease to less than 25% normal value – then total oxygen consumption decreases and metabolism shifts to anaerobiosis leading to vital organ dysfunction (Tobin 2020).
Oxygen Delivery Systems
Venturi masks
Venturi masks (VM) are frequently used when nasal cannulas are not providing adequate FiO2 control in hypoxic patients. Venturi’s effect is used to provide supplemental oxygen, but this setup is limited to flow rates of 15 litres/minute, offering only a fraction of the needed flow in dyspnoeic patients. An increase in respiratory rate and/or tidal volume generates high minute volume ventilation and the FiO2 will be diluted in a larger volume of air decreasing the actual inspiratory FiO2. When 15 litres/minute flows are insufficient to satisfy respiratory needs, a step up is required to increase inspiratory flows
High flow nasal cannulas
High flow nasal cannulas (HFNC) is an oxygen delivery system capable of providing 30-60 litres/minute of heated and humidified gas meeting the demands of tachypnoeic patients providing high minute ventilation while ensuring constant FiO2 (Rochwerg et al. 2020). High flows have a double effect on upper airways: enhancing anatomical dead space washout and CO2 clearance thus reducing respiratory rate and effort, resulting in an increased lung homogeneity and end expiratory lung volume (Basile et al. 2020). Thus, given our experience in Pisa, the main indications for HFNC are represented by 1) patients that do not tolerate CPAP/NIV in the absence of increased work of breathing; 2) early stages of the disease to prevent therapeutic escalation - if PEEP is not required as inferred by chest CT and/or lung echo; 3) alternation to CPAP especially in weaning contexts. When compared to conventional oxygen therapy HFNC decreased the need for intubation as well as respiratory support escalation though there is no clear evidence on its effects on mortality and hospital or ICU stay (Rochwerg et al. 2020).
Continuous positive airway pressure
Continuous positive airway pressure (CPAP) and non-invasive ventilation are first line treatments for respiratory failure with concomitant clinical signs of increased WOB or fatigue, inside and outside the ICU (Gattinoni et al. 2020). Advantages include possible application in early pre-hospital and hospital settings resulting in lower intubation and infection rates, decreasing morbidity and mortality and providing more favourable outcomes (Brusasco et al. 2015). CPAP system consists of an interface delivering an inspiratory flow with an adjustable FiO2 and constant end-expiratory positive pressure (PEEP) at the patients mouth adjustable via a PEEP valve (Grieco et al. 2021). CPAP interfaces are either a helmet or a facial mask with the latter generally being less tolerated by patients if treated for long periods of time. During respiratory distress, patients can generate extremely high inspiratory flows - in some cases exceeding 60 litres/minute - hence, it is mandatory to use systems capable of providing high flows that match patients' peak inspiratory flow in order to be able to revert increased work of breathing and to prevent respiratory muscle exhaustion (Brusasco et al. 2015; Grieco et al.
2021). Its use in acute lung injury decreases arteriovenous shunting improving oxygenation and dyspnoea while decreasing WOB (L’Her et al. 2005). Efficient CPAP must not only provide appropriate inspiratory airflow but also guarantee minimal CO2 rebreathing (Patroniti et al. 2003). A simple way to ensure proper inspiratory flow to patients during CPAP is to match their inspiratory demands. This can be achieved by measuring the temperature inside the helmet: an early sign of CO2 rebreathing and expression of unmatched inspiratory airflow demands. Concerning PEEP titration, CT studies have shown that PEEP induced alveolar recruitment using PEEP of 8-16 cmH2O resulted in minimal recruitment of lung parenchyma independently of respiratory system mechanics and compliance as well as modifications in gas exchange (Ball et al. 2021). This is easily explained by autopsy findings which reported injury in the alveolar epithelial cells, hyaline membrane formation, hyperplasia of type II pneumocytes, diffuse alveolar damage with consolidation due to fibroblastic proliferation with extracellular matrix and fibrin forming clusters in air-spaces and capillary vessel finally leading to alveolar spaces substituted by fibrosis (Ball et al. 2021; Grillo et al. 2021). Thus, high PEEP levels are not routinely recommended but rather PEEP titration should be individualised case by case.
Pressure support ventilation
Physiological effects of pressure support ventilation (PSV) during acute lung injury include increments in tidal volume (L’Her et al. 2005), but we have to be aware that increased tidal volumes inferred by the absolute changes in transpulmonary pressure (Pl) are considered to be associated with dynamic lung stress and NIV failure for de novo AHRF (Carteaux et al. 2016). Only one study reports physiological measurements during NIV (L’Her et al. 2005). It showed that non-invasive pressure support of 10-15 cmH2O above a PEEP of 5-10 cm H2O was the best combination to reduce inspiratory effort, dyspnoea and ameliorate oxygenation but inspiratory effort and WOB, estimated by Pes pressure-time product, are highly inter-individually variable among patients. Hence, this could imply that not all the patients are exposed to the same risk of self-induced lung injury. Moreover, ventilator settings during PSV have been shown to influence patient-ventilator synchrony and asynchronies which are associated with prolonged mechanical ventilation (Chao et al. 1997) and mortality (Blanch et al. 2015).
Helmet-NIV in pressure support mode, compared to HFNC, has shown lower incidence of intubation though there was not a significant difference of in-hospital mortality (Grieco et al. 2021). Despite that, the reported overall in-hospital mortality for Helmet-PSV was quite high (24%) if compared with another retrospective study on Helmet-CPAP in-hospital mortality which was 14% (Brusasco et al. 2021) and was additionally confirmed by other reports (Oranger et al. 2020; Tobin et al. 2021). This difference in in-hospital mortality can be explained by the fact that during PSV, but not during CPAP nor during HFNC, oesophageal pressure (Pes) swings, sum up with positive airway pressure to determine transpulmonary pressure (Pl) possibly leading to SILI if the treatment is not individualised by assessing early inspiratory efforts through oesophageal manometry (Tonelli et al. 2020).
Treatment Failure and Indications for Invasive Mechanical Ventilation
HFNC and CPAP/NIV should be the first line of treatment in acute hypoxic respiratory failure and a transition to invasive ventilation should be made when they are no longer effective as described by an increased WOB at risk of SILI (Cabrini et al. 2020). Non-invasive treatment failure is more likely in patients with more severe clinical conditions: higher SAPSII and lower PaO2/FiO2 ratio (Carteaux et al. 2016). Predicted treatment success and failure of HFNC therapy can be calculated using ROX index (SpO2/FiO2 to respiratory rate): values >4.88 are likely to avoid intubation whereas lower values are at high risk of treatment failure (Roca et al. 2016). Detection of CPAP/NIV failure can be estimated by using the HACOR score (HR, pH, GCS, PaO2/FiO2, RR) after one hour of treatment: scores >5 represent high risk for NIV failure (Duan et al. 2017). Also expired tidal volumes greater than 9.5 ml/kg of predicted body weight during NIV for moderate-severe hypoxaemia in AHRF are independently associated with non-invasive ventilation failure (Carteaux et al. 2016). Diaphragmatic ultrasonography has been suggested as a potential useful tool to predict adverse outcomes (Corradi, et al. 2021) and to predict response to CPAP in hospitalised patients for COVID-19 pneumonia (Corradi et al. 2021). Respiratory muscle ultrasonography, represents a quick method to evaluate other factors indicative of treatment failure such as respiratory muscle dysfunction during dyspnoea, increased WOB and patient-ventilator asynchronies (Tuinman et al. 2020).
In light of high mortality rates, up to 90% in COVID-19 patients submitted to invasive mechanical ventilation. The appropriate time for tracheal intubation is when signs and symptoms of significant respiratory distress or tissue hypoxia are present despite maximal non-invasive support (Pisano et al. 2021; Tobin et al. 2021). In those cases progression to invasive mechanical ventilation should be carried out without delay, especially in case of persisting dyspnoea and/or vigorous respiratory efforts despite 4-6 hours of CPAP (COVID-19 Lombardy ICU Network et al. 2020; Gattinoni et al. 2020). However, no significant difference of in-hospital mortality rate, between early versus late intubation policy has been described (Lee et al. 2020) and there is no prospective study that evaluates whether a comprehensive strategy of intubation, according to predefined criteria, could affect outcomes.
Conclusions
Oxygen therapy and non-invasive ventilation in COVID-19 should be based on pathophysiological changes and a step-by-step approach should be adopted in choosing the right therapeutic strategy for each patient. CPAP has been demonstrated to be an effective alternative to invasive mechanical ventilation even for severe acute hypoxic respiratory failure in COVID-19 patients. Hence, the choice to treat a patient with non-invasive supports should not be viewed as a second choice due to lack of ICU availability, but rather as the best early therapeutic approach in the initial phase of the disease (Pisano et al. 2021).
Conflict of Interest
None.
References:
Alhazzani W, Evans L, Alshamsi F et al. (2021) Surviving Sepsis Campaign Guidelines on the Management of Adults With Coronavirus Disease 2019 (COVID-19) in the ICU: First Update. Critical Care Medicine, 49(3):e219.
Allado E, Poussel M, Valentin S et al. (2021) The Fundamentals of Respiratory Physiology to Manage the COVID-19 Pandemic: An Overview. Frontiers in Physiology, 11.
Ball L, Robba C, Maiello L et al. (2021) Computed tomography assessment of PEEP-induced alveolar recruitment in patients with severe COVID-19 pneumonia. Critical Care, 25(1):81.
Basile MC, Mauri T, Spinelli E et al. (2020) Nasal high flow higher than 60 L/min in patients with acute hypoxemic respiratory failure: A physiological study. Critical Care, 24(1):654.
Blanch L, Villagra A, Sales B et al. (2015) Asynchronies during mechanical ventilation are associated with mortality. Intensive Care Medicine, 41(4):633–641.
Brusasco C, Corradi F, Di Domenico A et al. (2021) Continuous positive airway pressure in COVID-19 patients with moderate-to-severe respiratory failure. European Respiratory Journal, 57(2):2002524.
Brusasco C, Corradi F, Ferrari AD et al. (2015) CPAP Devices for Emergency Prehospital Use: A Bench Study. Respiratory Care, 60(12), 1777–1785. https://doi.org/10.4187/respcare.04134
Cabrini L, Ghislanzoni L, Severgnini P et al. (2020) Early versus late tracheal intubation in COVID-19 patients: A pro-con debate also considering heart-lung interactions. Minerva Cardioangiologica.
Carsana L, Sonzogni A, Nasr A et al. (2020) Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: A two-centre descriptive study. The Lancet Infectious Diseases, 20(10):1135–1140.
Carteaux G, Millán-Guilarte T, De Prost N et al. (2016) Failure of Noninvasive Ventilation for De Novo Acute Hypoxemic Respiratory Failure: Role of Tidal Volume. Critical Care Medicine, 44(2):282–290.
Chao DC, Scheinhorn DJ, Stearn-Hassenpflug M (1997) Patient-ventilator trigger asynchrony in prolonged mechanical ventilation. Chest, 112(6):1592–1599.
Corradi F, Isirdi A, Malacarne P et al. (2021) Low diaphragm muscle mass predicts adverse outcome in patients hospitalized for COVID-19 pneumonia: An exploratory pilot study. Minerva Anestesiologica, 87(4):432–438.
Corradi F, Vetrugno L, Orso D (2021) Diaphragmatic thickening fraction as a potential predictor of response to continuous positive airway pressure ventilation in Covid-19 pneumonia: A single-center pilot study. Respiratory Physiology & Neurobiology, 284:103585.
COVID-19 Lombardy ICU Network, Foti G, Giannini A et al. (2020) Management of critically ill patients with COVID-19: Suggestions and instructions from the coordination of intensive care units of Lombardy. Minerva Anestesiologica, 86(11).
Duan J, Han X, Bai L et al. (2017) Assessment of heart rate, acidosis, consciousness, oxygenation, and respiratory rate to predict noninvasive ventilation failure in hypoxemic patients. Intensive Care Medicine, 43(2):192–199.
Gattinoni L, Chiumello D, Rossi S (2020) COVID-19 pneumonia: ARDS or not? Critical Care, 24(1):154, s13054-020-02880-z.
Grasselli G, Zangrillo A, Zanella A et al. (2020) Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA, 323(16):1574–1581.
Grieco DL, Menga LS., Eleuteri D, Antonelli M (2019) Patient self-inflicted lung injury: Implications for acute hypoxemic respiratory failure and ARDS patients on non-invasive support. Minerva Anestesiologica, 85(9):1014–1023.
Grieco DL, Menga LS, Cesarano M et al. (2021) Effect of Helmet Noninvasive Ventilation vs High-Flow Nasal Oxygen on Days Free of Respiratory Support in Patients With COVID-19 and Moderate to Severe Hypoxemic Respiratory Failure: The HENIVOT Randomized Clinical Trial. JAMA, 325(17):1731.
Grillo F, Barisione E, Ball L et al. (2021) Lung fibrosis: An undervalued finding in COVID-19 pathological series. The Lancet Infectious Diseases, 21(4):e72.
Lee YH, Choi KJ, Choi SH et al. (2020) Clinical Significance of Timing of Intubation in Critically Ill Patients with COVID-19: A Multi-Center Retrospective Study. Journal of Clinical Medicine, 9(9).
L’Her E, Deye N, Lellouche F et al. (2005) Physiologic Effects of Noninvasive Ventilation during Acute Lung Injury. American Journal of Respiratory and Critical Care Medicine, 172(9):1112–1118.
Oranger M, Gonzalez-Bermejo J, Dacosta-Noble P et al. (2020) Continuous positive airway pressure to avoid intubation in SARS-CoV-2 pneumonia: A two-period retrospective case-control study. European Respiratory Journal, 56(2):2001692.
Ottestad W, Kåsin JI, Høiseth LØ (2018) Arterial Oxygen Saturation, Pulse Oximetry, and Cerebral and Tissue Oximetry in Hypobaric Hypoxia. Aerospace Medicine and Human Performance, 89(12):1045–1049.
Patroniti N, Foti G, Manfio A et al. (2003) Head helmet versus face mask for non-invasive continuous positive airway pressure: A physiological study. Intensive Care Medicine, 29(10):1680–1687.
Pisano A, Yavorovskiy A, Verniero L, Landoni G (2021) Indications for Tracheal Intubation in Patients With Coronavirus Disease 2019 (COVID-19). Journal of Cardiothoracic and Vascular Anesthesia, 35(5):1276–1280.
Roca O, Messika J, Caralt B et al. (2016) Predicting success of high-flow nasal cannula in pneumonia patients with hypoxemic respiratory failure: The utility of the ROX index. Journal of Critical Care, 35:200–205.
Rochwerg B, Einav S, Chaudhuri D et al. (2020) The role for high flow nasal cannula as a respiratory support strategy in adults: A clinical practice guideline. Intensive Care Medicine, 46(12):2226–2237.
Tobin MJ (2020) Basing Respiratory Management of COVID-19 on Physiological Principles. American Journal of Respiratory and Critical Care Medicine, 201(11):1319–1320.
Tobin MJ, Jubran A, Laghi F (2021) Noninvasive strategies in COVID-19: Epistemology, randomised trials, guidelines, physiology. European Respiratory Journal, 57(2):2004247.
Tobin MJ, Laghi F, Jubran A (2020) Why COVID-19 Silent Hypoxemia Is Baffling to Physicians. American Journal of Respiratory and Critical Care Medicine, 202(3):356–360.
Tonelli R, Fantini R, Tabbì L et al. (2020) Early Inspiratory Effort Assessment by Esophageal Manometry Predicts Noninvasive Ventilation Outcome in De Novo Respiratory Failure. A Pilot Study. American Journal of Respiratory and Critical Care Medicine, 202(4):558–567.
Tuinman PR, Jonkman AH, Dres M et al. (2020) Respiratory muscle ultrasonography: Methodology, basic and advanced principles and clinical applications in ICU and ED patients - a narrative review. Intensive Care Medicine, 46(4):594–605.











