ICU Management & Practice, Volume 17 - Issue 4, 2017
Process indicator-based plan-do-act-check cycle in a single-centre quality improvement initiative
To assess whether intracranial pressure (ICP) management in severe traumatic brain injury patients is being applied according to our institutional protocol we developed a process indicator aimed at improving quality of care.
Background and rationale
Traumatic brain injury (TBI) is a leading cause of morbidity and mortality throughout the world. The initial management of primary brain injury may include prompt surgical decompression of intracranial haematomas. In the hours and days following the trauma, secondary brain injury can develop as a result of a cascade of molecular injury mechanisms. This cellular injury results in altered mitochondrial functioning, cell apoptosis and oedema. Oedema raises ICP, compromising cerebral perfusion pressure (CPP) and resulting in ongoing and additional brain injury. Given the importance of halting the vicious circle of secondary brain injury resulting in worse clinical outcome, prompt ICP management and maintenance of CPP according to a strict incremental management protocol are essential. Most international guidelines state that an ICP exceeding 20 mmHg should be treated (Bratton et al. 2007). The incremental management steps involve analgesia and sedatives first, followed by osmotic therapies to reduce cerebral oedema and ultimately the so-called second-tier therapies including barbiturates or decompressive craniectomy. Implementation of these guidelines in clinical practice can have a significant impact on outcomes following TBI (Fakhry 2004; Arabi 2010), and this effect may be augmented with better adherence and more aggressive management (Bulger 2002; Cnossen 2016).
You might also like:ICP monitoring lowers mortality in severe traumatic brain injury
The Erasmus Medical Center is an academic (neuro) trauma centre. Our TBI protocol is based on strict ICP management according to international guidelines. As part of a departmental quality improvement strategy in 2016, we decided to assess whether this protocol was optimally applied in our ICU. For this purpose we were greatly interested in a quantitative measurement tool that would adequately reflect ICP protocol adherence, but would not be too complex to calculate or interpret.
Over the last decade, numerous quality indicators have been developed as guides to monitor, evaluate and improve the quality of care. Quality indicators are measurement tools that focus on the availability of specific means (structure), the actual delivery of care (process) or the result of care (outcome).
Quantitative research usually reports outcome indicators, such as mortality or adverse events. Process indicators refer to the appropriateness of delivered care such as, in our case, protocol or guideline-recommendation adherence (Lingsma 2010). An intrinsic advantage of process measures is that they are generally more sensitive for or directly linked to changes in the quality of actual clinical care than outcome measures. Furthermore, only a valid process indicator can detect flaws in the practical management of, for instance, ICP treatment that would otherwise go unnoticed when looking at outcome measures alone. Improving the quality of care starts with the identification of such flaws and subsequently translating them into improvement measures. With the use of a process indicator for ICP treatment we aimed to identify frequent causes of non-adherence that we could subsequently address following a plan-do-check-act (PDCA) method (Taylor et al. 2014). As an additional benefit, process indicators are applicable to relatively small patient groups to provide information on the way care is actually delivered.
We therefore aimed to use ICP protocol adherence as a process indicator to reflect the quality of our ICP treatment in TBI patients. For this purpose, we performed a literature search on process indicators for ICP management to be used. However, this search yielded no applicable results, and we concluded that in the field of ICP treatment in TBI patients, a validated quality indicator such as protocol adherence had yet to be developed and evaluated.
Objective
To develop and apply a simple ICP process indicator and additionally construct a plan-docheck- act (PDCA) cycle describing key learning points and further iterative steps to improve quality of care in TBI patients.
Methods
This single-centre quality improvement project took place on the ICU of the Erasmus Medical Center in Rotterdam, the Netherlands. Institutional review board approval was obtained. It was judged that given the observational nature of the investigation and the fact that it was a quality improvement project informed consent was not a necessity.
First, we used focus group interviews to reach consensus on a suitable ICP indicator that was face-valid. The focus group consisted of a neurointensivist, neurosurgeon, ICU nurse with special expertise in neurocritical care, research nurse, quality manager and ICU manager. The result was an ICP indicator that was defined as:
the number of patients with ≥ 1 period of ICP > 20 mmHg for more than 30 minutes without appropriate treatment escalation (numerator) according to the protocol, as a percentage of the total number of patients with ICP monitoring (denominator). Ideally the indicator should be zero (%).
This indicator was then evaluated for its relevance, reliability, validity, feasibility and usability using the Appraisal of Indicators through Research and Evaluation (AIRE) 20-points checklist (Dimick, 2010). It met all the requirements of the AIRE 20-points checklist and was therefore considered to be a suitable process indicator. Finally, the ICP indicator was tested for feasibility in all the adult TBI patients receiving ICP monitoring and treatment at our ICU, admitted between April and September 2016.
As stated by our protocol, indications for ICP monitoring are defined as a Glasgow Coma Score (GCS) ≤ 8 and an abnormal CT scan showing evidence of mass effect from lesions such as haematomas, contusions or swelling, or a GCS ≤ 8 with a normal CT scan but two or more of the following items: age above 40 years, an M2 or M3 on the GCS scale or a systolic blood pressure below 90 mmHg. Exclusion criteria are patients that did receive an ICP monitoring device at ICU admission, but were considered to have a moribund prognosis (based on the interpretation of the CT scan and ICP at the start of treatment by the attending neurosurgeon and intensivist). We excluded this last category of patients from analysis, because they were not expected to represent patients in whom strict adherence to the ICP treatment protocol would confer benefit. Data on ICP levels and applied treatments were collected from our patient data management system (PDMS) that automatically records ICP levels with a ten-minute interval.
Reasons for protocol non-adherence (defined as ICP > 20 mmHg for ≥ 30 minutes but no escalation of the management) were first assessed by a research nurse and subsequently interpreted by an ICU fellow and neurointensivist. Lessons learned regarding details of non-adherence were incorporated in a predefined PDCA cycle construct. A PDCA cycle is a four-step management method for continuous improvement of processes. The fundamental principle of this method is iteration: once a hypothesis or target is obtained or not obtained, executing the cycle and repeating it will extend knowledge and will bring one closer to perfection of the process. Ultimately a process indicator and PDCA cycle well applied will ideally result in the indicator becoming redundant, since no further improvement for that specific indicator can be obtained.
Results
We analysed 43 patients, of whom 5 had a moribund prognosis, resulting in 38 included patients (Table 1). Adherence to treatment protocol was inadequate in 3 cases (8%). In 1 patient, there was an adequate intervention (osmotic therapy was started according to protocol), but not within the set time, while in 2 patients the therapy was inadequate (failure to control fever resulting in several temperature spikes and the start of osmotic therapy whilst sedation was still inadequate). With these data we constructed a PDCA cycle (Figure 1).

Figure 1. PDCA cycle
The first step (Plan) is to establish an objective, in our case to improve quality of care by improving protocol adherence. The second step (Do) is to implement a measure to reach your objective. Ours was the development and implementation of the ICP indicator. After implementation, results of the ICP indicator were studied in the third step (Check). In the final step (Act), we aim to implement improvement measures based on the results of step three. In practice this meant that we aimed to provide, additional education of the complete medical and nursing staff on our ICP management protocol, and specifically provided feedback on the indicator (Figure 2) to the medical and nursing staff. For this purpose, we used our department’s website, individual emails and newsletters and explained the potential for improvements.
Since
the PDCA cycle is an iterative process, we plan to continue reporting the
process indicator and thereby assess the outcome of this indicator on protocol
adherence every six months after the implementation of extra education of the
staff. We are currently in this process and first results of further indicator measurements
are shown in Figure 2.
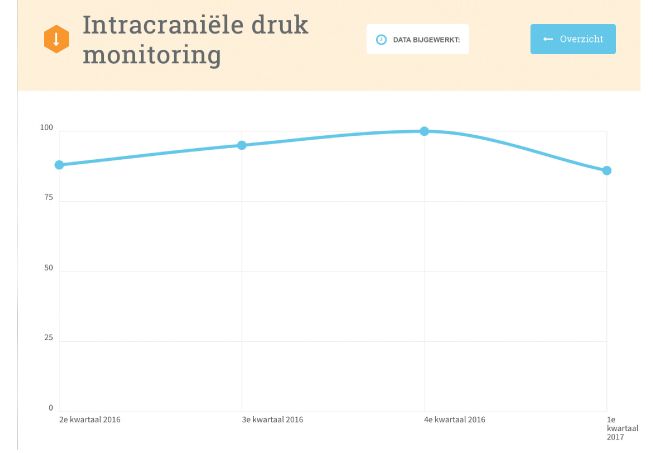
Figure 2. Snapshot from our institutional website with the ICP indicator showing guideline adherence quarterly. The fourth quarter of 2016 (third dot from the left) had the highest adherence (100%).
| Month | No. patients with ICP monitoring | No. patients with moribund diagnosis | No. patients with ICP ≥ 20 mmHg ≥ 30 mins | No. cases of protocol breach | Numerator | Denominator | Indicator (%) |
| April | 4 | 0 | 4 | 1 | 1 | 4 | 25 |
| May | 11 | 0 | 9 | 1 | 1 | 11 | 9 |
| June | 4 | 2 | 2 | 0 | 0 | 2 | 0 |
| July | 7 | 1 | 5 | 0 | 0 | 6 | 0 |
| August | 10 | 1 | 9 | 1 | 1 | 9 | 11 |
| September | 7 | 1 | 7 | 0 | 0 | 6 | 0 |
| Total | 43 | 5 | 38 | 3 | 3 | 38 | 8 |
Table 1. ICP indicator results
Conclusion
We showed the feasibility of our ICP process indicator, which proved to be useful for the construction of a simple PDCA cycle. Since the cycle is aimed to be iterative, either further improvement can be expected and/or development of a slightly adapted indicator may be expected depending on the user’s convenience and perceived usability, since not only indicator outcome but also indicator construct will be re-evaluated.
Future perspectives
The next step will be to try and translate this process improvement into a possible outcome benefit. For example, we could compare predicted outcome (mortality) of our TBI patients (using the CRASH score developed by IMPACT https://iii.hm/dr8) with actual outcome as a benchmark measure, and relate this information to the level of adherence to the process indicator. One might hypothesise that the ideal indicator is sensitive to processes of care, provides clear information on how to improve and is associated with a benchmark outcome. We hope to further develop this concept in our institution and publish results on its feasibility in the future when patient numbers included allow for more robust statistical analyses.
Conflict of interest
Joyce A.M. Heijneman declares that she has no conflict of interest. Jasper van Bommel declares that he has no conflict of interest. Mathieu van der Jagt declares that he has no conflict of interest.
Abbreviations
CPP cerebral perfusion pressure
ICP intracranial pressure
PDCA plan-do-check-act
TBI
traumatic brain injury
References:
Bratton SL, Chestnut RM, Ghajar J et al. (2007) Guidelines for the management of severe traumatic brain injury. VIII. Intracranial pressure thresholds. J Neurotrauma, 24 Suppl 1: S55-8.
Bulger EM, Nathens AB, Rivara FP et al. (2002) Management of severe head injury: institutional variations in care and effect on outcome. Crit Care Med, 30: 1870-6.
Cnossen MC, Scholten AC, Lingsma HF et al. (2016) Adherence to guidelines in adult patients with traumatic brain injury: a living systematic review. J Neurotrauma, 33: 1-14.
Dimick JB (2010) What makes a "good" quality indicator? Arch Surg: 145(3): 295.
Fakhry SM, Trask AL, Waller MA et al. (2004) Management of brain-injured patients by evidence-based medicine protocol improves outcomes and decreases hospital charges. J Trauma, 56: 492-9.
Lingsma HF (2010) Measuring quality of care, methods and applications to acute neurologic diseases. Thesis, Erasmus University, Rotterdam. https://repub.eur.nl/pub/21742
Taylor MJ, McNicholas C, Nicolay C, et al. (2014) Systematic review of the application of the plan–do–study–act method to improve quality in healthcare. BMJ Qual Saf, 23: 290-8.









