Describes the epidemiology and outcomes for very old patients as known in 2018, along with a short introduction to the most relevant “geriatric syndromes” important also for intensivists, and discusses where we should increase our body of knowledge to make a more precise triage in this patient group.
The very old ICU patient is a term often used for those patients aged ≥ 80 years. This group of patients constitutes 10-15% of today’s ICU patients in Europe, and is expected to rise in absolute and relative terms in parallel with the increase of life span in our countries. If we continue to have the same policy towards treatment of the critically ill very old for the coming 25 years, we may well see a doubling of this patient group. Even today there is an ongoing discussion about who to treat, when and where to treat these patients, and arguments span from not to consider age at all, to question the admittance of the very old in general. Most researchers and clinicians argue there must be a middle way, allowing for active treatment in selected patients, while on the other hand reducing the treatment intensity, maybe to comfort and care, in others.
The problem is obvious: how to select those that will profit from intensive care from those that most certainly will not? This is of course a generic challenge intensive care has had for decades, and we hoped the traditional severity scoring systems could offer us help. However, these systems have not been found accurate enough to guide decisions at the individual patient level, although they may perform well on a group level. They have also been found to perform less well in ICU-subpopulations, like in the very old (Minne et al. 2011).
Epidemiology
We are not sure if the absolute increase of patients ≥ 80 years we observe in many ICUs really is parallel with the increase in the elderly population per se. We know the elderly population is increasing in European countries as well as in the rest of the world, but very few studies adjust the increase in elderly ICU admission to the increase in the general population. This was recently done in Scotland where they in fact found the admission rates among the elderly (≥80) to decrease over time from around 37/105 in 2005 to 29/105 in 2009, a reduction of 22%! (Docherty et al. 2016) They speculate whether this reduction can be rationing based on chronological age, selecting less morbid elderly patients, but no data was revealed to support this assumption.
Outcomes
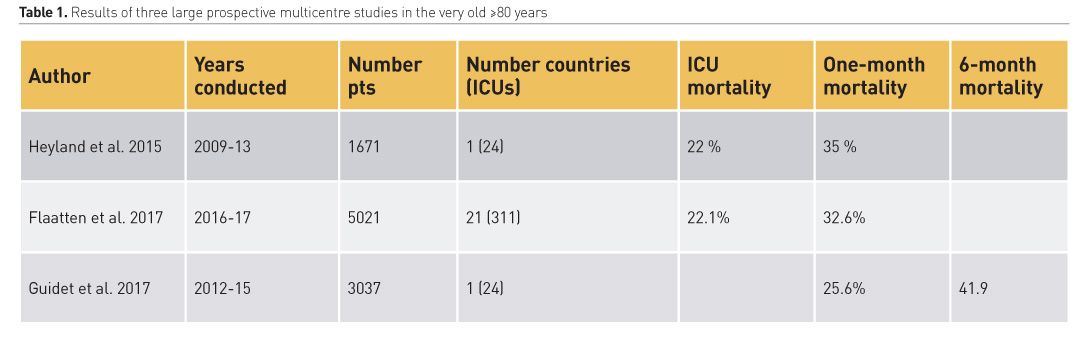
Several studies about short- and long-term mortality in very old ICU patients have been published over the last 15 years. Lately also some large prospective multicentre studies have been performed, mainly to determine survival, but also quality of life (QOL). Table 1 summarises results from three such recent studies showing that 25-35% of the very old die within a month. In the Canadian study, also QOL of one-year survivors was studied, indicating that approximately 50% of the patients that survived to one year (≈50%) had significantly reduced QOL.
Geriatric syndromes
Geriatric medicine has for decades used a “battery” of age-related assessments found to be very relevant with advancing age. For some time, many of these assessments also have found use outside traditional geriatric medicine, in particular for surgical procedures. Lately some of these syndromes, particularly frailty, have also been used to describe ICU cohorts.
Frailty
With frailty, we understand the gradual decline in various body functions that occurs with age, and manifests itself as diminished reserves during stress. It is important to underline two important facts with frailty:
- Frailty does not always parallel chronological age; hence we can find frailty in ICU patients not considered to be aged, and we can find patients with a high chronological age without being frail.
- Frailty is not a disease, and must be separated from such, although the border between frailty and disease is sometimes difficult to define.
Typical frailty symptoms may include (but are not restricted to):
- Slow walking speed
- Reduced activity level
- Exhaustion
- Decreased muscle mass and strength
- Unintentional weight loss
There are several methods to identify frailty. Such assessment is usually based on one of two different assessment methods: Fried criteria and Rockwood methods. The former is often called frailty phenotype, and the latter frailty index. The phenotype is based on a pre-defined set of criteria (categorical variables), while the frailty index is a result of a clinical evaluation using continuous variables and a more unspecified set of criteria (Cessari et al. 2014). Since both methods have clinical disadvantages, particularly in the acute care setting, a new clinical frailty scale (CFS) was developed in Canada in the second part of the large Canadian study of health and ageing (Rockwood et al. 2005). The last version of CFS includes a 9-scale partly visual and partly descriptive scale and has gained popularity in situations where patients are unable to participate. Several large prospective studies have been conducted in recent years using the CFS, showing very good correlation between frailty and outcomes in the very old population of critically ill patients. In the large Very Old Intensive Care Patient: A Multinational Prospective Observation Study (VIP1) study of more than 5000 elderly ICU patients in Europe, frailty was found to be an important and independent factor for 30-day mortality, with a near linear relation between increasing frailty and mortality (Flaatten et al. 2017).
Sarcopaenia
Sarcopaenia is the specific name for muscle wasting in the elderly. It is an important cause of functional decline and is interwoven with frailty. Occurrence of sarcopaenia is very common. It is found in 11-50% of elderly people > 80 years and is associated with a negative outcome in a variety of studies. There are multiple causes of sarcopaenia, and inactivity is probably the most important one, although malnutrition and inflammation also may play a role. Commonly it can be diagnosed using imaging techniques like MRI, CT and ultrasound, but more often simpler methods like gait speed and muscle strength in the arms are used as screening tools. The European Working Group on Sarcopenia in Older People has published clinical guidelines and consensus criteria for age-related sarcopaenia (Cruz-Jentoft et al. 2010). Here gait speed < 1 m/s in a 6-metre course and handgrip strength < 30 kg in men and < 20 in women may indicate sarcopaenia.
In intensive care these methods to document sarcopaenia are difficult, since both gait speed and handgrip strength require awake and cooperative patients. However, it may have a role at discharge and post-ICU follow-up. Since sarcopaenia at admission adds to ICU-acquired loss of muscle mass (inactivity and stress-mediated catabolism) the net result may be detrimental for the very old with regards to post-ICU rehabilitation. Hence methods to prevent further muscle mass in critically ill sarcopaenia patients are vital.
Recently, studies using ultrasound measurement to diagnose sarcopaenia have been published and may provide a fast, noninvasive method to document muscle mass at admission that can find its way into initial assessment of elderly ICU patients.
Cognitive decline
Age-related cognitive decline is understood as a normal (non-disease related) ageing of cognitive functions. It does not affect all elderly people, but mild cognitive impairment (MCI) is a frequent finding in the elderly and has been found in 15-20% of people aged 65 and above. It may affect primarily memory (amnestic MCI) or other thinking skills, known as “non-amnestic MCI”. A detailed mapping of cognitive function takes time and requires a cooperative patient. There are quick methods like mini-mental state examinations that are often used to screen for dementia, but these also require awake patients. However, a simple questionnaire designed to ask close relatives about their next-of-kin’s mental state is available and can be used also in emergency settings. It is called IQCODE: the informant questionnaire on cognitive decline in the elderly (Jorm et al. 1989). Here an informant who knows the patient well is asked a series of questions about mental state, and compares present status (before the illness) with 10 years ago. Although not as sensitive as direct examination of the patient, it is a quick screening tool and may have relevance for rehabilitation and cooperation with the patients while in the ICU.
Immunosenescence
Our immune system is affected by age. Immune cells are continuously renewed from stem cells. Both the proliferative capacity and the number of these immune cells are decreased due to progressive telomere shortening, resulting in an immune dysfunction over the years, which is called immunosenescence. This may explain the increased susceptibility in elderly people to acquire infections, clearly demonstrated with the markedly increased incidence of sepsis in the elderly population. Unfortunately, tests for immune function are at present not fully developed, and we lack a quick and reliable method to identify patients at risk.
Specific ICU care for the elderly
Studies have repeatedly shown that the elderly patient is given less active treatments compared with their younger counterparts. The reasons for this are not clear, but may imply therapeutic nihilism. This is of course unfortunate, since when admitted, all patients should be given appropriate care until a decision of limitation eventually is chosen. The potential to involve a geriatrician in the ICU team is also attractive. Elderly patients usually come with a lot of “baggage”: co-morbidity and associated drug therapies. A study from nursing homes (Barber et al. 2009) revealed that on average the residents used 8 different medications daily. Not all of them are necessary, and some may be potentially harmful in the ICU setting. To help sort this out a geriatrician can be of help. Geriatric competence may also be helpful in working out the best plan for rehabilitation in very old ICU survivors.
The response to ICU therapy is important and should be evaluated as soon as possible after admission. For this daily organ failure assessment is important, and responders should show improved function within some days. If this does not occur or failure increases, most would then consider further ICU treatment questionable. Withholding or withdrawal of care should be considered and discussed with caregivers or family. Many of the patients would then be offered comfort and care instead of intensive care.
Delirium is frequent in the very old, and is the rule more often than not. Hence the ICU should be prepared, and avoid known factors that increase delirium: heavy sedation, particularly use of benzodiazepines; ensuring sleep at night and that patients are awake and mobilised at daytime, even if still on a ventilator, are also important factors. Further muscle wasting and malnutrition should also be focused on.
What more do we need to know?
There are more questions than answers with regards to very old ICU patients. First and foremost, we need better prediction ability to identify elderly patients that most probably will profit from ICU admittance, and those that probably will not. We know the one-month mortality after intensive care is about 40% after acute admissions, and to identify most of them before ICU admittance should be given high priority. Not only is this important for our societies with an increasing shortage of ICU beds, but primarily for the patients and caregivers. An ICU admission is a burden for all, and should probably not be offered if most lights alert red. The search for prediction systems with a very high sensitivity and specificity may prove to be impossible. Still, we must continue to investigate this in depth, and in particular to include information from “geriatric” syndromes as specified above. The message from using frailty assessment at admission is promising. Again, in the VIP1 study we found frailty to be the best individual predictor of 30-day mortality, even better than the Sequential Organ Failure Assessment (SOFA) score at admission (Flaatten et al. 2017). It is possible that a combination of the geriatric syndromes alone or with other selected markers can give us a useful prognostic score, to be helpful in the pre-ICU triage process. The only way to find out is through large prospective studies testing the power of these new markers to predict outcomes.
Today we have simple and robust methods to map frailty, sarcopaenia and cognition before ICU admission. Hopefully also immunosenescence will be possible to assess in a simple way in the near future.
Conflict of interest
Hans Flaatten declares that he has no conflict of interest.