ICU Management & Practice, Volume 21 - Issue 3, 2021
Introduction
Airway management including tracheal intubation (TI) is a common and integral part of the care provided to critically ill patients. Critically ill patients have minimal physiological reserves, leading to a physiologically difficult airway and predisposing them to an increased risk of complications. Furthermore, anatomic and logistical difficulties with airway management are also very common in these situations. In a recent prospective observational study evaluating intubation practices in critically ill patients across 29 countries, adverse events occurred after intubation in 45.2% of patients, including cardiovascular instability in 42.6%, severe hypoxaemia in 9.3%, and cardiac arrest in 3.1% (Russotto et al. 2021). This baseline physiologic risk is often further exaggerated when more than one attempt at tracheal intubation is required (De Jong et al. 2020a). Therefore, the goal of tracheal intubation in the critically ill patients is to have a first pass success without adverse events. A recent prospective observational study in 1513 emergency tracheal intubations showed that first pass success without adverse events was reduced to a similar extent in patients with anatomically and physiologically difficult airways, highlighting the importance of physiological optimisation along with the use of strategies and tools to overcome anatomical difficulty (Pacheco et al. 2021).
Challenges Associated with Airway Management in Critically Ill Patients
Anatomic challenges
The incidence of an anatomically difficult airway in ICU patients has been estimated to be around 10% (Griesdale et al. 2008; Jaber et al. 2006; Schwartz et al. 1995). Patients who might have been intubated with ease in the operating rooms (ORs) tend to be harder to intubate in the ICU with antecedent complications (Taboada et al. 2018). Furthermore, airway assessment in such scenarios can be sub-optimal; there may not be enough time to perform an airway assessment, or the patient may not cooperate for an airway exam.
Physiologic challenges
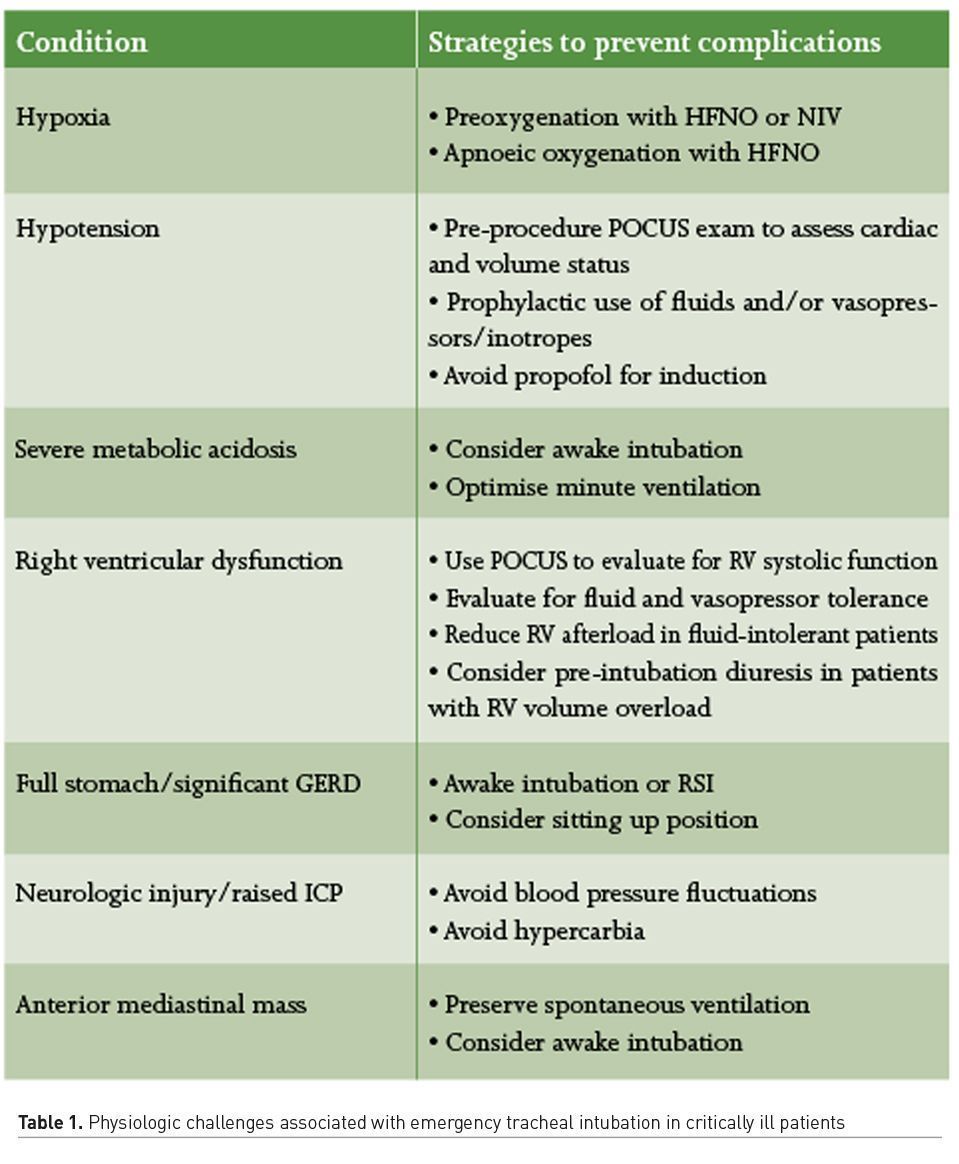
An airway is considered physiologically difficult when the physiologic derangements place the patient at a higher risk of cardiovascular collapse with intubation and conversion to positive pressure ventilation (Mosier et al. 2015). Critically ill patients often have conditions such as hypoxaemia, hypotension, severe metabolic acidosis and right ventricular dysfunction, that predispose them to a physiologically difficult airway. Table 1 lists some of the causes of physiologic challenges that providers might face in these circumstances as well as strategies to prevent associated complications.
Logistical challenges
Along with the anatomic and physiologic challenges, it is also important to take into account the situational or logistical challenges that accompany airway management in the ICU. These situationally difficult airways can arise due to infrastructure, personnel and/or equipment related concerns. Space and lighting limitations and poor access to the patient’s head are some of the infrastructure related issues that exist. Personnel related limitations pose significant challenges as the airway team may be working at the bedside with members that are inexperienced and often times, expert help may not be readily available. Similarly, appropriate airway equipment and drugs may not be available in all situations and circumstances.
Planning and Preparation
Considering the high rates of complications associated with tracheal intubation in critically ill patients, adequate preparation during the peri-intubation period is essential. After assessing for potential anatomic, physiologic, or situational challenges that may be present, preparation and optimisation of the patient as well as the team is important. Ensuring availability of necessary equipment and personnel, maximising preoxygenation, and haemodynamic optimisation can prevent complications. Whenever feasible, a thorough discussion with the patient (or their surrogate) on the acceptance of intubation and mechanical ventilation should be held.
Clinical history and examination
A thorough review of the patient’s medical record for clinical history, relevant laboratory investigations and imaging reports along with a focused physical exam is vital in tailoring the approach to airway management including the choice of drugs, to avoid associated complications. Bedside point of care ultrasound (POCUS) exam may be very helpful, not only to help assess the haemodynamic status of the patient, but also to assess for increased aspiration risk. The protocolised use of a handheld POCUS device can improve the accuracy of diagnosis and outcomes in patients with acute respiratory or circulatory failure (Zieleskiewicz et al. 2021), and its utility in ICU airway management needs further evaluation.
Airway assessment
Identifying a potentially difficult airway is crucial; however, current bedside screening tests are limited by their poor sensitivity and specificity (Shiga et al. 2005). A history of difficulty with airway management described by the patient or documented in the patients’ medical record is important. Similarly, the time of last oral intake, contraindications to use succinylcholine or other drugs, drug allergies, history of sleep apnoea, presence of dentures, etc. is important to assess. Amongst the bedside airway assessment tests, the upper lip bite test has the highest sensitivity (Roth et al. 2018), and the combination of mallampati (MP) score and thyromental distance (TMD) provides the most accuracy at predicting difficult intubation (Shiga et al. 2005). Many of these methods are often not feasible in this setting where the patient may be uncooperative, sedated, agitated, unstable or delirious (Levitan et al. 2004). Automated face-analysis approach to predict a difficult airway may help overcome some of these limitations, though further studies are needed to validate this approach before it becomes part of routine assessment (Cuendet et al. 2016).
While recognising the anatomically difficult airway is one part of the pre-intubation assessment, the anticipation of physiologic challenges prior to airway management is equally important. The MACOCHA score (Mallampati score III or IV, sleep Apnoea syndrome, decreased Cervical mobility, mouth Opening <3cm, Coma defined by a Glasgow score <8, severe Hypoxaemia, and if the practitioner is not an Anaesthetist) that combines anatomic, physiologic, and operator characteristics is simple to perform, may be more suitable for use in critically ill patients and was recently validated in a multicentre study (De Jong et al. 2013).
Equipment and personnel
Considering the unpredictability associated with these situations, we suggest preparing for airway management in a way that assumes that every step might fail. The patient should be appropriately monitored and continuous EtCO2 monitoring should be utilised. It is reasonable to make the monitors, especially pulse-oximetry loud and audible to everyone in the room. Next, it is important to ensure that necessary equipment is on hand and functional. A cart with all of the necessary supplies to facilitate tracheal intubation, rescue oxygenation and haemodynamic support is essential to avoid the need for securing essential equipment at the last minute. If deemed necessary and time permitting, a flexible intubation scope should be brought to the bedside to assist in intubation. Considering that blood and vomitus in the airway are common in this situation and may predict a difficult airway (Gaither et al. 2014), availability of functioning high-efficiency suction devices should be ensured.
Tracheal intubation is an aerosol generating procedure and can impact the transmission of respiratory virus illnesses such as COVID-19 (Brown et al. 2020). Hence, as recommended by national organisations, providers involved with TI in patients with or suspected to have a respiratory viral illness should adhere to full contact and airborne personal protective equipment (Cook et al. 2020). Also, in such circumstances, intubation should be performed in an airborne infection isolation room by an experienced provider, with additional help available outside the room.
Elucidating the skill level of available staff and establishing clear roles and responsibilities of the team members before proceeding for TI is essential. Closed-loop and clear communication among the team members about the airway concerns, airway plan, backup plan etc. is essential in such stressful situations and can help prevent medical errors, as well as increase speed and efficiency. The presence of two airway operators, with at least one being experienced has shown to reduce complications during tracheal intubation (Jaber et al. 2010).
Checklists
Instituting a checklist for non-OR airways may help ensure the necessary preparations and precautions have been taken. Pre-intubation checklist is especially effective in less experienced hands, and it was found that the implementation of an intubation bundle can reduce life-threatening complications associated with emergency airway management (Jaber et al. 2010). The key is to keep the checklist simple and succinct, thus improving compliance and acceptance. A randomised trial evaluating the use of a written checklist prior to tracheal intubation in ICU compared with usual care found no difference in lowest oxygen saturation and lowest systolic blood pressure (SBP) from induction up to two minutes after TI between the groups (Janz et al. 2018). However, the checklist did not include interventions aimed at physiological optimisation [e.g. non-invasive ventilation (NIV), fluid load, early use of vasopressors], thus explaining why the selected outcomes did not improve with the use of checklist. A pre-intubation checklist, which includes interventions to enhance oxygenation and haemodynamic optimisation, may be effective in less experienced hands, as observed by reduction in peri-intubation complications following the implementation of an ICU intubation bundle (Jaber et al. 2010).
Tracheal Intubation Procedure
Patient positioning
Optimal patient positioning during TI should strive to optimise both anatomic and physiological parameters and is essential to increase the success of intubation and avoid complications. The ramped position reduces the risk of pulmonary aspiration of gastric contents and of desaturation by maintaining the patient’s functional residual capacity. The debate about whether the sniffing or ramped position may be more appropriate still persists. Pulmonary fellows experienced increased intubation difficulty with intubations performed in the ramped position compared with the sniffing position (Semier et al. 2017), whereas patients intubated by emergency department residents showed improved first-attempt success with ramping compared with supine (Turner et al. 2017). Also while an obese patient would significantly benefit from a ramped position, a frail patient with limited neck range of motion may not. Thus, in the absence of clear cut evidence favouring one versus the other, it is suggested that positioning should be individually tailored to patient characteristics, as well as the skillset of the intubating provider (Myatra 2019).
Preoxygenation and apnoeic oxygenation
For majority of patients that require airway management in the ICU, life-threatening hypoxaemia during the procedure is a major concern. Adequate preoxygenation is essential to better prepare such patients for intubation and should be an integral component of all emergent airway interventions. It is important that oxygen therapy be initiated immediately on arrival while preparations are underway to maximise the duration of preoxygenation. Oxygen delivery can be achieved using a simple face mask, standard or high flow nasal oxygen (HFNO), NIV mask or a combination of these devices. NIV or HFNO should be considered over conventional oxygen therapy for preoxygenation for TI in critically ill patients (Bailard et al. 2006; Frat et al. 2019; Guitton et al. 2019). In patients with moderate to severe hypoxaemia, NIV may be superior to HFNO. Apnoeic oxygenation should be continued during attempts at TI and gentle mask ventilation should be considered during rapid sequence intubation (RSI) to prevent or treat hypoxaemia (De Jong et al. 2020b).
Rapid sequence intubation
Critically ill patients may have gastroparesis associated with critical illness or may not be fasted at the time of TI and hence, an RSI technique is often used in this setting. The main objective of the technique is to minimise the time interval between loss of protective airway reflexes and TI. Despite the technique's widespread use, there is still no agreement on how it should best be performed especially with regards to manual ventilation and application of cricoid pressure (CP) or Sellick’s manoeuvre. Avoidance of manual ventilation before TI was traditionally recommended to avoid gastric insufflation, but recent evidence suggests that bag mask ventilation between induction and endotracheal tube placement may be well tolerated (Casey et al. 2019). In patients with physiologically difficult airway, mask ventilation may be lifesaving, and providers should balance the perceived risk of aspiration versus life threatening complications related to desaturation. The Sellick’s manoeuvre has been shown to have a questionable benefit. While there is evidence that gastric insufflation can be prevented by this manoeuvre (Rice et al. 2009), there are concerns that application of CP can result in an increased risk of aspiration by decreasing the lower oesophageal sphincter tone (Tournadre et al. 1997) and may impair the laryngeal view and thereby delay intubation and increase the potential for aspiration (Haslam et al. 2005).
Neuromuscular blockade versus maintenance of spontaneous ventilation
Neuromuscular blockade can improve mask ventilation, improve intubating conditions, abolish upper airway muscle tone including laryngospasm, optimise chest wall compliance and overall improve the first-attempt success with tracheal intubation in the ICU (Mosier et al. 2015). Also, the avoidance of paralytics during TI in the ICU has been found to be associated with difficult face mask ventilation and severe oxygen desaturation (Heuer et al. 2012). As a result, the use of paralytics as part of TI is recommended in critically ill patients by various national guidelines (Higgs et al. 2018; Quintard et al. 2019). However, apnoea after institution of muscle paralysis, may result in rapid desaturation, thus emphasising the importance of preoxygenation and apnoeic oxygenation. The fear of inability to mask ventilate after giving neuromuscular blockade is also one of the reasons providers are reluctant in using these agents.
Device selection
Intubation in critically ill patients is technically more challenging than elective intubation performed in the OR with worse glottic visualisation, increase in moderate or difficult intubation, lower first-attempt success, and a higher rate of complications (Taboada et al. 2018). Video laryngoscopy (VL) improves visualisation of the glottic opening; however, difficulty with navigating the endotracheal tube to and beyond the larynx is a concern and requires training. A recent meta-analysis that included nine randomised controlled trials with over 2000 critically ill patients, found that the use of a video-laryngoscope (VL) did not improve first-pass success, even when evaluating the studies according to the experience of the operator (Cabrini et al. 2018). Thus, the routine use of a VL for tracheal intubation in ICU remains controversial, but it clearly improves glottic visualisation as compared with direct laryngoscopy making it an important tool for difficult airway management (Jaber et al. 2019). Future trials will better define the role of a VL in ICU. Nonetheless, a video-laryngoscope should always be available as a backup tool to rescue difficult intubation and/or unsuccessful first attempt at DL in all ICU TIs.
Supraglottic airways (SGAs) can be utilised for establishing an airway in critically ill patients and are usually used after failed intubation as a rescue device in these situations (Shavit et al. 2018). They are relatively easy to insert and provide some protection against aspiration. If successful oxygenation can be achieved with a SGA, then this can be maintained until expert help arrives. An SGA may be used as a primary airway device and an intubation conduit in situations where an anatomically difficult airway is predicted. The endotracheal tube introducer (Bougie) is also a useful tool when the epiglottis is visible but vocal cords cannot be seen with a significantly higher first attempt intubation success reported in emergency situations (Driver et al. 2018). Hence, a bougie may be used for TI in the ICU by providers who have experience with its use.
Haemodynamic optimisation and pharmacologic management
Hypotension or cardiovascular collapse is common during and following TI in critically ill patients. In fact, cardiovascular instability was observed in 42.6% of all patients undergoing emergency intubation in a large multinational observational study and was found to be associated with significant morbidity and mortality (Russotto et al. 2021). This haemodynamic instability possibly results from a combination of pharmacologically induced sympatholytic action, conversion from negative-pressure to positive-pressure ventilation as well as the amelioration of the hypoxia and hypercarbia associated sympathetic drive. There is limited prospective data on optimal strategies for haemodynamic support during emergency airway management. Administration of a fluid bolus prior to intubation has been shown to be of minimal benefit (Smischney et al. 2020) and may actually cause harm and lead to postintubation hypoxaemia in non-volume responsive patients (Janz et al. 2019). It could be helpful in patients that are hypovolaemic and assessment of fluid responsiveness by a quick passive leg raising test or bedside point of care ultrasound (POCUS) exam may help identify the suitable candidates for a fluid bolus. Another widely used intervention to avoid peri-intubation hypotension is the use of bolus or push-dose vasopressors and/or continuous infusion of vasopressor agents either during or immediately after intubation (Jaber et al. 2010; Weingart 2015). This prophylactic use of vasopressors may be an alternative and needs further investigation.
Amongst the induction agents, ketamine preserves the patient’s respiratory drive, and can be used for delayed sequence intubation (optimising airway preparation and preoxygenation in an otherwise uncooperative or agitated patient). Caution should be exercised with its use in patients in whom excessive sympathetic stimulation could be detrimental, such as those with acute myocardial infarction or acute cerebrovascular event. The combination of ketamine and propofol (ketofol) may offer an acceptable haemodynamic profile when used for intubation in critically ill patients (Smischney et al. 2019). Etomidate is frequently used in emergent intubations in critically ill patients, as it tends not to cause hypotension on induction. However, it is a selective adrenocortical 11 beta-hydroxylase inhibitor and causes transient adrenal insufficiency, the clinical implications of which are debatable. No difference in morbidity has been observed with the use of ketamine or etomidate for emergency endotracheal intubation of critically ill patients (Jabre et al. 2009). Propofol can have a profound effect on the haemodynamics, especially in patients with hypovolaemia and/or impaired cardiac function and is rarely used for TI in critically ill patients. Titrating the dose, using smaller doses, pre-emptive or concomitant administration of vasopressor agents and judicious fluid administration may avoid significant haemodynamic perturbations. With a lack of consensus on an ideal induction agent, either etomidate or ketamine could be used as the first choice induction agent based on specific scenarios. The use of propofol should be limited to situations where TI is required for airway protection in the absence of cardiopulmonary compromise.
Succinylcholine, which is a depolarising neuromuscular blocker, provides a rapid onset and short duration of action. Its major side effects include malignant hyperthermia and acute hyperkalaemia in susceptible patients. Rocuronium does not carry the same risk of malignant hyperthermia or acute hyperkalaemia, but the onset and duration of action is highly dependent on the doses used for intubation. While meta-analyses have reported that the intubating conditions are better with succinylcholine (Perry et al. 2003; Tran et al. 2017), a recent study showed that clinician grading of intubating conditions was similar with both these drugs, and intubation-related complications occurred more often in the succinylcholine group (Guihard et al. 2019).
Future Directions
Future research is required to find interventions to reduce the high incidence of cardiorespiratory complications associated with TI in critically ill patients. These interventions should be aimed at improving first-pass tracheal intubation success while optimising the patient’s cardiorespiratory status. Although significant work has been done to enhance the actual process of TI in critically ill patients, there is still limited knowledge on appropriate management of a physiologically difficult airway. The use of POCUS prior to TI can help assess the patient’s cardiovascular status, facilitate rapid screening for difficult laryngoscopy, and assess for increased aspiration risk. Its routine use and its impact on outcomes needs validation. Studies investigating patient positioning, techniques for preoxygenation and the role of RSI/Sellick’s manoeuvre based on patients’ physiologic derangements would be beneficial. Future work should also address device development, including different blade shapes, angles, lengths and cameras as well as stylet modifications. The optimum choice of drugs for induction of anaesthesia as well as muscle relaxation based on patients’ physiological alterations also needs further evaluation.
Conflict of Interest
AKK is funded with a Clinical and Translational Science Institute (CTSI) NIH/NCTAS KL2 TR001421 award for a trial on continuous postoperative haemodynamic and saturation monitoring.
References:
Baillard C, Fosse JP, Sebbane M et al. (2006) Noninvasive ventilation improves preoxygenation before intubation of hypoxic patients. Am J Respir Crit Care Med, 174:171-7.
Brown J, Gregson FKA, Shrimpton A et al. (2020) A quantitative evaluation of aerosol generation during tracheal intubation and extubation. Anaesthesia
Casey JD, Janz DR, Russell DW et al. (2019) Bag-Mask Ventilation during Tracheal Intubation of Critically Ill Adults. N Engl J Med, 380:811-21.
Cabrini L, Landoni G, Baiardo MR et al. (2018) Tracheal intubation in critically ill patients: a comprehensive systematic review of randomized trials. Crit Care, 22:6.
Cook TM, El-Boghdadly K, McGuire B et al. (2020) Consensus guidelines for managing the airway in patients with COVID-19: Guidelines from the Difficult Airway Society, the Association of Anaesthetists the Intensive Care Society, the Faculty of Intensive Care Medicine and the Royal College of Anaesthetists. Anaesthesia, 75:785-99.
Cuendet GL, Schoettker P, Yuce A et al. (2016) Facial Image Analysis for Fully Automatic Prediction of Difficult Endotracheal Intubation. IEEE Trans Biomed Eng, 63:328-39.
De Jong A, Molinari N, Terzi N et al. (2013) Early identification of patients at risk for difficult intubation in the intensive care unit: development and validation of the MACOCHA score in a multicenter cohort study. Am J Respir Crit Care Med, 187:832-9.
De Jong A, Rolle A, Pensier J et al. (2020a) First-attempt success is associated with fewer complications related to intubation in the intensive care unit. Intensive Care Med, 46:1278-80.
De Jong A, Casey JD, Myatra SN (2020b) Focus on noninvasive respiratory support before and after mechanical ventilation in patients with acute respiratory failure. Intensive Care Med, 46:1460-3.
Driver BE, Prekker ME, Klein LR et al. (2018) Effect of Use of a Bougie vs Endotracheal Tube and Stylet on First-Attempt Intubation Success Among Patients With Difficult Airways Undergoing Emergency Intubation: A Randomized Clinical Trial. JAMA, 319:2179-89.
Frat JP, Ricard JD, Quenot JP et al. (2019) Non-invasive ventilation versus high-flow nasal cannula oxygen therapy with apnoeic oxygenation for preoxygenation before intubation of patients with acute hypoxaemic respiratory failure: a randomised, multicentre, open-label trial. Lancet Respir Med, 7:303-12.
Gaither JB, Spaite DW, Stolz U et al. (2014) Prevalence of difficult airway predictors in cases of failed prehospital endotracheal intubation. J Emerg Med, 47:294-300.
Griesdale DE, Bosma TL, Kurth T et al. (2008) IComplications of endotracheal intubation in the critically ill. Intensive Care Med, 34:1835-42.
Guihard B, Chollet-Xemard C, Lakhnati P et al. (2019) Effect of Rocuronium vs Succinylcholine on Endotracheal Intubation Success Rate Among Patients Undergoing Out-of-Hospital Rapid Sequence Intubation: A Randomized Clinical Trial. JAMA, 322:2303-12.
Guitton C, Ehrmann S, Volteau C et al. (2019) Nasal high-flow preoxygenation for endotracheal intubation in the critically ill patient: a randomized clinical trial. Intensive Care Med, 45:447-58.
Haslam N, Parker L, Duggan JE (2005) Effect of cricoid pressure on the view at laryngoscopy. Anaesthesia, 60:41-7.
Heuer JF, Barwing TA, Barwing J et al. (2012) Incidence of difficult intubation in intensive care patients: analysis of contributing factors. Anaesth Intensive Care, 40:120-7.
Higgs A, McGrath BA, Goddard C et al. (2018) Guidelines for the management of tracheal intubation in critically ill adults. Br J Anaesth, 120:323-52.
Jaber S, Amraoui J, Lefrant JY et al. (2006) Clinical practice and risk factors for immediate complications of endotracheal intubation in the intensive care unit: a prospective, multiple-center study. Crit Care Med, 34:2355-61.
Jaber S, Jung B, Corne P et al. (2010) An intervention to decrease complications related to endotracheal intubation in the intensive care unit: a prospective, multiple-center study. Intensive Care Med, 36:248-55.
Jaber S, De Jong A, Pelosi P (2019) Cabrini L, Reignier J, Lascarrou JB. Videolaryngoscopy in critically ill patients. Crit Care, 23:221.
Jabre P, Combes X, Lapostolle F et al. (2009) Etomidate versus ketamine for rapid sequence intubation in acutely ill patients: a multicentre randomised controlled trial. Lancet, 374:293-300.
Janz DR, Semler MW, Joffe AM et al. (2018) A Multicenter Randomized Trial of a Checklist for Endotracheal Intubation of Critically Ill Adults. Chest, 153:816-24.
Janz DR, Casey JD, Semler MW et al. (2019) Effect of a fluid bolus on cardiovascular collapse among critically ill adults undergoing tracheal intubation (PrePARE): a randomised controlled trial. Lancet Respir Med, 7:1039-47.
Levitan RM, Everett WW, Ochroch EA (2004) Limitations of difficult airway prediction in patients intubated in the emergency department. Ann Emerg Med, 44:307-13.
Mosier JM, Joshi R, Hypes C et al. (2015) The Physiologically Difficult Airway. West J Emerg Med, 16:1109-17.
Mosier JM, Sakles JC, Stolz U et al. (2015) Neuromuscular blockade improves first-attempt success for intubation in the intensive care unit. A propensity matched analysis. Ann Am Thorac Soc, 12:734-41.
Myatra SN (2019) Optimal position for laryngoscopy - Time for individualization? J Anaesthesiol Clin Pharmacol, 35:289-91.
Pacheco GS, Hurst NB, Patanwala AE et al. (2021) First Pass Success Without Adverse Events Is Reduced Equally with Anatomically Difficult Airways and Physiologically Difficult Airways. West J Emerg Med, 22:360-8.
Perry J, Lee J, Wells G. (2003) Rocuronium versus succinylcholine for rapid sequence induction intubation. Cochrane Database Syst Rev, CD002788.
Quintard H, l'Her E, Pottecher J et al. (2019) Experts' guidelines of intubation and extubation of the ICU patient of French Society of Anaesthesia and Intensive Care Medicine (SFAR) and French-speaking Intensive Care Society (SRLF) : In collaboration with the pediatric Association of French-speaking Anaesthetists and Intensivists (ADARPEF), French-speaking Group of Intensive Care and Paediatric emergencies (GFRUP) and Intensive Care physiotherapy society (SKR). Ann Intensive Care, 9:13.
Rice MJ, Mancuso AA, Gibbs C (2009) Cricoid pressure results in compression of the postcricoid hypopharynx: the esophageal position is irrelevant. Anesth Analg, 109:1546-52.
Roth D, Pace NL, Lee A et al. (2018) Airway physical examination tests for detection of difficult airway management in apparently normal adult patients. Cochrane Database Syst Rev, 5:CD008874
Russotto V, Myatra SN, Laffey JG et al. (2021) Intubation Practices and Adverse Peri-intubation Events in Critically Ill Patients From 29 Countries. JAMA, 325:1164-72.
Schwartz DE, Matthay MA, Cohen NH (1995) Death and other complications of emergency airway management in critically ill adults. A prospective investigation of 297 tracheal intubations. Anesthesiology, 82:367-76.
Semler MW, Janz DR, Russell DW et al. (2017) A Multicenter, Randomized Trial of Ramped Position vs Sniffing Position During Endotracheal Intubation of Critically Ill Adults. Chest, 152:712-22.
Shavit I, Aviram E, Hoffmann Y (2018) Biton O, Glassberg E. Laryngeal mask airway as a rescue device for failed endotracheal intubation during scene-to-hospital air transport of combat casualties. Eur J Emerg Med, 25:368-71.
Shiga T, Wajima Z, Inoue T, Sakamoto A (2005) Predicting difficult intubation in apparently normal patients: a meta-analysis of bedside screening test performance. Anesthesiology, 103:429-37.
Smischney NJ, Khanna AK, Brauer E et al. (2020) Risk Factors for and Outcomes Associated With Peri-Intubation Hypoxemia: A Multicenter Prospective Cohort Study. J Intensive Care Med, 885066620962445.
Smischney NJ, Nicholson WT, Brown DR et al. (2019) Ketamine/propofol admixture vs etomidate for intubation in the critically ill: KEEP PACE Randomized clinical trial. J Trauma Acute Care Surg, 87:883-91.
Taboada M, Doldan P, Calvo A et al. (2018) Comparison of Tracheal Intubation Conditions in Operating Room and Intensive Care Unit: A Prospective, Observational Study. Anesthesiology, 129:321-8.
Tournadre JP, Chassard D, Berrada KR, Bouletreau P (1997) Cricoid cartilage pressure decreases lower esophageal sphincter tone. Anesthesiology, 86:7-9.
Tran DTT, Newton EK, Mount VAH et al. (2017) Rocuronium vs. succinylcholine for rapid sequence intubation: a Cochrane systematic review. Anaesthesia, 72:765-77.
Turner JS, Ellender TJ, Okonkwo ER et al. (2017) Feasibility of upright patient positioning and intubation success rates At two academic EDs. Am J Emerg Med, 35:986-92.
Weingart S (2015) Push-dose pressors for immediate blood pressure control. Clin Exp Emerg Med, 2:131-2.
Zieleskiewicz L, Lopez A, Hraiech S et al. (2021) Bedside POCUS during ward emergencies is associated with improved diagnosis and outcome: an observational, prospective, controlled study. Crit Care, 25:34.










