Poor ICU nutrition delivery remains a challenge worldwide. Objective malnutrition diagnosis and personalisation of nutrition delivery may be one way of addressing this problem.
Modern, and increasingly expensive ICU care now allows prolonged survival from illness and injury by providing life-sustaining support for extended periods of time, making previously nonsurvivable ICU insults, survivable. In fact, innovations in ICU care have resulted in yearly reductions in hospital mortality from sepsis (Kaukonen et al. 2014). However, these same data reveal many ICU “survivors” are not returning home to functional lives post-ICU; but instead to rehabilitation settings where it is unclear if they ever return to a meaningful quality of life (QoL) (Kress and Hall 2014). ICU acquired weakness (ICU-AW) is a common complication of critical illness. A hallmark of ICU-AW is reduced muscle mass and strength, which both are independent predictors of ICU survival (Weijs et al. 2014).
Muscle mass and quality derived by CT scans in mechanically ventilated patients, is associated with a 6-month higher mortality (Weijs et al. 2014; Looijaard et al. 2016; Looijaard et al. 2018). Tragically, an increasing number of patients who survive ICU suffer from severe, prolonged functional disabilities (Kress and Hall 2014; Hopkins et al. 2017; McNelly et al. 2016; da Silveira et al. 2018). Recent data shows ICU patients (mean age: 55) are likely to be discharged to post-acute care facilities and incur substantial costs of ~$3.5 million/functioning survivor (Herridge et al. 2016). Unfortunately, post-ICU functional disability is most common and most severe in survivors requiring time on a ventilator for respiratory failure, where recent data shows 2 out of 3 ICU survivors (65%) suffer significant functional limitations (Kress and Hall 2014). Thus, it must be asked in modern ICU care, “are we creating survivors…or victims?” This is a defining challenge for modern critical care all major ICU societies have recommended giving priority to research addressing post-ICU QoL outcomes in these survivors (Needham et al. 2017).
To improve functional and QoL outcomes in acute renal failure (ARF) survivors, one obvious low-cost therapeutic strategy that can be rapidly implemented is objective, data guided personalised nutrition delivery to attempt to maintain and allow recovery of muscle mass/function. This is particularly essential in patients with pre-existing and subsequent iatrogenic malnutrition that commonly occurs in ICU patients. In fact, despite increasing obesity rates in many countries, preexisting malnutrition is highly prevalent in ICU patients- with as many as 1 in 2 (30-50%) patients being malnourished at ICU admission(Normal et al. 2008). Unfortunately, unrecognised malnutrition in the hospital and ICU may be among the most pressing “silent epidemics” facing hospitalised patients in the world today. Although it is well known that greater then 1 in every 3 hospitalised patients is malnourished at hospital admission (Barker et al. 2011), limited older data estimates only 3% of malnourished U.S. hospitalised patients are being recognised and diagnosed (Corkins et al. 2014). Thus, only 1 in 10 malnourished patients are ever diagnosed and even fewer are treated. This silent epidemic of “the skeleton in the hospital closet” has been described for >40 years (Butterworth 2005), but this data shows it has yet to be addressed. This is tragic as mortality is 5 times greater (11.7% versus 2.4%) overall for hospitalised patients diagnosed with malnutrition versus well-nourished (Corkins et al. 2014). Further, the outcomes of ICU patients with pre-existing malnutrition and sarcopenia are further complicated by the fact that critical illness is characterised by an acute catabolic response leading to rapid loss of lean body mass (LBM) contributing to muscle wasting, weakness, and loss of function (Dinglas et al. 2017; Wischmeyer 2016; Wischmeyer 2018).
Poor ICU nutrition delivery remains a challenge worldwide. Review of current practice demonstrates the actual amount of nutrition delivered primarily via enteral nutrition (EN) in ICU patients is <50% of the prescribed goal even in our most malnourished patients (Cahill et al. 2010). In an era of heightened concern about patient safety and medical error, we and others have consistently documented that critically ill patients receive, on average only 40-50% of their prescribed goal nutritional requirements for prolonged periods (>1 week) after ICU admission (Barr et al. 2004; Binnekade et al. 2005; De Jonghe et al. 2001; Heyland et al. 2003; Krishnan et al. 2003; Rubinson et al. 2004). This is particularly concerning as the average protein delivery (thought essential for muscle/functional recovery) for the first 12 days of an ICU stay is only 0.6 g/kg/d (Cahill et al. 2010), which is one-third of the guideline recommendations of 1.5-2.0 g/kg/d in ICU (Taylor et al. 2016). This is an urgent patient safety crisis that must be addressed. One of the major drivers of lack of emphasis on improved nutrition delivery in ICU and post-ICU patients is lack of objective data to guide “personalised ICU nutrition.” Specifically, lack of objective tools to: 1) Diagnose nutrition risk objectively, 2) Determine accurate bedside energy requirement data which is known to change throughout the course of illness, and 3) Lack of quantitative assessment tools to evaluate effect of nutrition on patient. As ICU physicians would not deliver vasopressors without a continuous blood pressure measure; we believe the ICU community has not embraced a focus on nutrition delivery due to lack of objective data to guide personalised nutrition care.
Role of Muscle Mass, Body Composition, and Indirect Calorimetry Analysis in Malnutrition Diagnosis and Nutrition Delivery
The use of a quick non-invasive technique to evaluate skeletal muscle quantity and quality in ICU patients could have profound prognostic implications for how malnutrition is diagnosed. As stated in new Global Leadership Initiative on Malnutrition (GLIM) Guidelines (Cederholm et al. 2019), muscle mass is a new and innovative marker of malnutrition. A number of techniques are now available to assess muscle mass, lean body mass (LBM), or Fat-Free Mass (FFM) in ICU patients at bedside. Further, new and easy-to-use bedside indirect calorimetry devices have also recently been developed. Key new available techniques for ICU nutrition and metabolic analysis for the delivery of personalised nutrition are summarised in Table 1.
Muscle Ultrasound
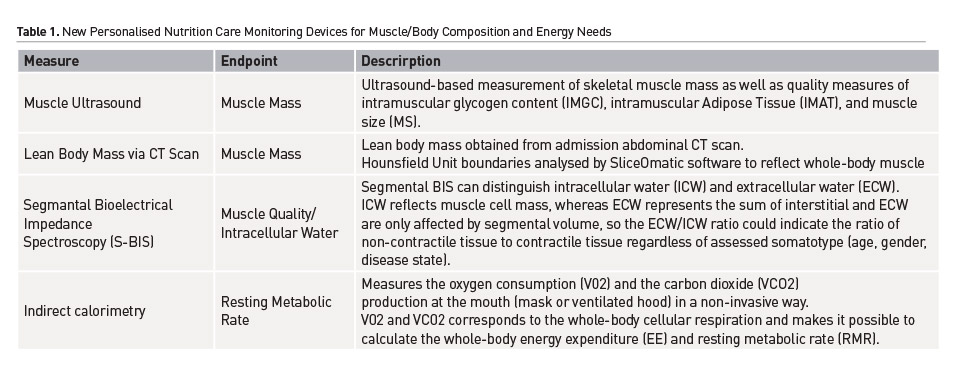
Recently, we have assisted with development of a muscle-specific U/S-based technique to enable non-invasive measurement of skeletal muscle mass as well as quality measures of intramuscular glycogen content (IMGC), intramuscular Adipose Tissue (IMAT), and muscle size (MS) (Wischmeyer et al. 2017; Wischmeyer and San Millan 2015). Validation of muscle mass from U/S with gold standard techniques (such as MRI/CT scan) has been previously published (Arbeille et al. 2009). Specific to ICU, a very recent trial showed good inter-/intra-rater reliability for muscle mass U/S in acutely ill patients (Pardo et al. 2018). Further, recent data has shown increased U/S-muscle mass is correlated to improved functional handgrip strength following an a targeted ICU nutrition intervention (Ferrie et al. 2016). Thus, muscle U/S measures correlates with improved patient function and this is modifiable by improved nutrition delivery. The decline in muscle function both during and after critical illness is the result of not only a change/reduction of skeletal muscle mass but also as a result of changes in muscle quality such as muscle composition, histology and morphology (Looijaard et al. 2016; Correa-de-Araujo et al. 2017; Fragala et al. 2015). Skeletal muscle quality is already recognised as a marker of function in healthy individuals (Watanabe et al. 2013) and critically ill patients (Wischmeyer et al. 2017; Parry et al. 2015; Puthucheary et al. 2013; Bear et al. 2017) and has been emerging as a means to describe the changes associated with altered muscle functioning (Fragala et al. 2015; Watanabe et al. 2013; Sieber 2017; Kelley and Kelley 2017). Assessing muscle mass and quality in clinical populations at the bedside is of key importance due to the emerging associations between low muscle quality with low muscle mass and poor functional status (da Silveira et al. 2018). This allows for an increased understanding of the relationship between skeletal muscle quantity and quality, and malnutrition/outcome risk (McNelly et al. 2016; da Silveira et al. 2018). Our group has initial validation data for muscle quality from U/S versus gold-standard CT scan muscle quality and have an R2 value of 0.989 (unpublished data). Finally, Puthucheary et al reported an increase in IMAT observed in muscle biopsies during ICU stay (Puthucheary et al. 2018). They described the existence of a compromised muscle bioenergetic status as a result of a dysregulated lipid oxidation (Puthucheary et al. 2013; Puthucheary et al. 2018).
The ease of adoption of muscle U/S at the ICU bedside has been markedly improved by the availability of a muscle specific U/S device (Musclesound Inc, Colorado, USA) This handheld U/S device is easy to carry and can be connected to a portable tablet device. The device is focused on allowing rapid, accurate measures of LBM at the bedside, with built-in guidance to ensure reproducible measurements. This new device is a significant improvement in LBM U/S technology. Unique measures of muscle glycogen and muscle quality can now be ascertained at the bedside in study subjects using the Musclesound U/S. As described. muscle quality has recently been correlated to muscle strength (Akazawa et al. 2018). Muscle glycogen U/S measures have been validated via muscle biopsy (Hill and Millan 2014) and we have shown ICU patients have significant muscle glycogen deficits (Wischmeyer et al. 2017; Wischmeyer and San Millan 2015). Muscle glycogen is known to change daily based on adequacy of nutrition intake, muscle uptake of substrate and “physical stress.” Thus, it could prove useful in monitoring of nutrition delivery and utilisation in ICU patients. See Figure 1 for example comparisons of muscle measures via MuscleSound and CT Scan.
Segmental Bioelectrical
Segmental bioelectrical impedance spectroscopy (SBIS) is another non-invasive approach of muscle quality assessment via bioelectrical impedance (BIA). SBIS or BIA equipment does not measure muscle mass directly, but instead derives an estimate of muscle mass based on whole-body electrical conductivity (Cruz-Jentoft et al. 2019; Kaido et al. 2012). Skeletal muscle has a large amount of water, and SBIS can separately assess intracellular water (ICW) and extracellular water (ECW), which are divided by the muscle cell membrane. SBIS can distinguish ICW and ECW from the total water in a particular segment. Segmental BIS is advantageous for assessments of a localised (left/right) region (arm, leg and trunk) instead of only the whole-body ICW reflects muscle cell mass, whereas ECW represents the sum of interstitial fluid and blood plasma in extracellular space. The calculations of ICW and ECW are only affected by segmental volume, so the ECW/ICW ratio could indicate the ratio of non-contractile tissue to contractile tissue regardless of assessed somatotype (age, gender, disease state). The phase angle and an estimation of energy expenditure are also reported in the SBIS measurement in modern devices.
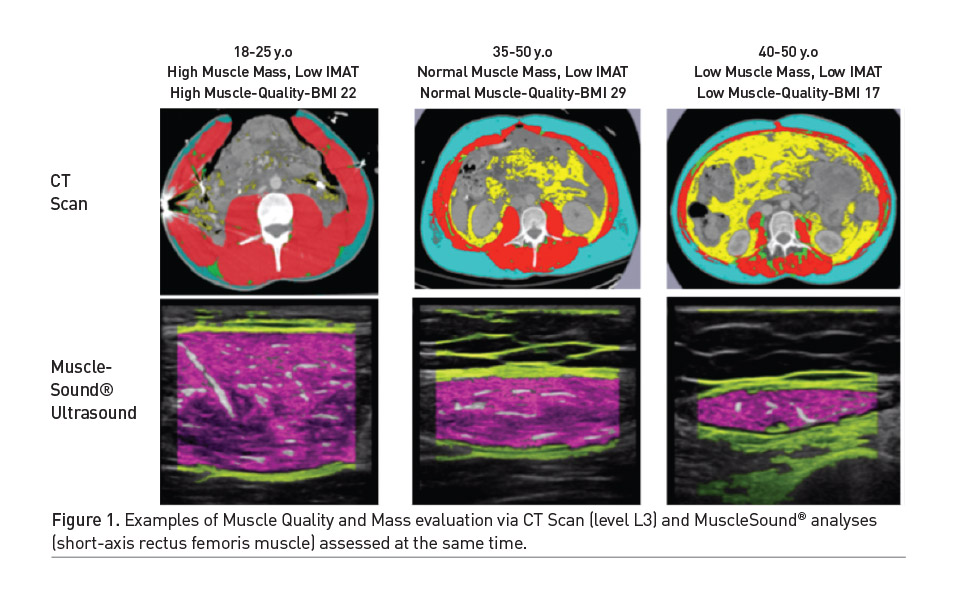
One limitation of SBIS and BIA measurements has been the concern for the effect of hydration status on measurements in the ICU (Looijaard et al. 2018). Recent research in critical illness has focused on the prognostic values of SBIS measurements such as the phase angle which are directly measured, and are not as sensitive to changes in hydration status. A multinational trial in a diverse population of 931 critically ill patients demonstrated a low phase angle at admission (day 1) was associated with increased 28-day mortality (Thibault et al. 2016). This was further validated in a recent study of 196 heterogenous ICU patients. This study showed a low phase angle at ICU admission was associated with increased 90-day mortality (Stapel et al. 2018). See Figure 2 for example comparisons of body composition measures via SBIS (Inbody S10, Inbody Inc, California, USA) and muscle-specific U/S (Musclesound). The newly published GLIM criteria describes specific (Cederholm et al. 2019) Appendicular Skeletal Muscle Index (ASMI, kg/m2) and Fat Free Mass thresholds that may be obtained from BIA (or SBIS) for low muscle mass to objectively diagnose malnutrition.
A New Era of Indirect Calorimetry Devices for Measurement of Personalised Energy Expenditure
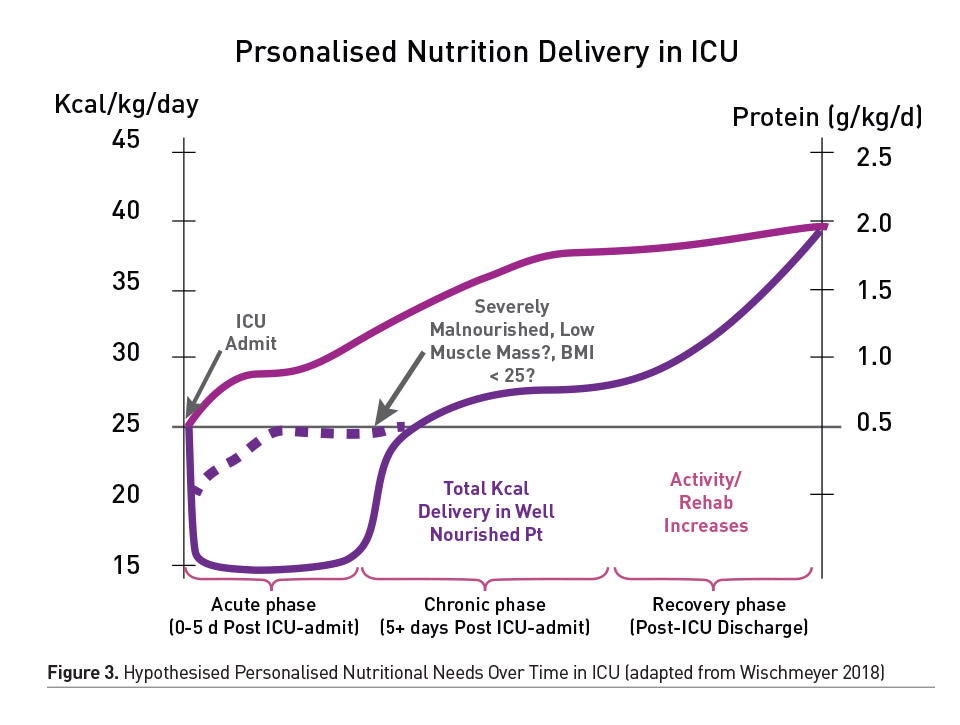
As shown in Figure 3, our research group has hypothesised that energy needs change throughout the course of illness and recovery (Wischmeyer 2018). However, this has not been validated with actual longitudinal resting energy expenditure (REE) measures, as is now possible with new metabolic cart devices (such as the Q-NRG, COSMED, Rome, Italy). As described, energy expenditure (EE) in ICU patients has been hypothesised to be highly variable based on a range of features including initial injury/illness, severity of illness (i.e. sepsis can dramatically decrease EE), nutritional status and other treatments (Wischmeyer 2018). It is also clear that clinicians ideally need to measure EE by indirect calorimetry (IC) to optimise nutritional support for better clinical outcome and to prevent over-/underfeeding (Heidegger et al. 2013; McClave et al. 2014). Difficulties in conduct, handling and interpretation of results often limit the use of IC in ICU patients. IC is the method utilised to measure EE in patients both during mechanical ventilation (MV) and can also now be routinely used in patients not requiring MV. The need for accurate determination of EE is increasing due to the rising prevalence of patients with clinical conditions making traditional equation-based estimation of EE unpredictable and plagued with variability. For example, elderly subjects with reduced lean body mass and increased fat mass have a reduced EE, not easily predicted with traditional equations for caloric need. In contrast, young patients, those with severe trauma, acute infection or significant obesity can have increased EE that is also difficult to estimate without IC (Heidegger et al. 2013; McClave et al. 2014). A number of studies have shown that predictive formula developed to calculate EE of such patients are not consistently clinically relevant (Fraipont and Preiser 2013; Guttormsen and Pichard 2014). Indeed, clinicians need to measure their patients’ EE in order to optimise the prescription of nutritional support and the clinical outcome (Heidegger et al. 2013; McClave et al. 2014), and IC is considered to be the gold standard for determining EE in the ICU patients. This is now becoming a reality with the development and ongoing evaluation of new, easy-to-use indirect calorimeter technology.
Conclusion: An Exciting New Era for Objective Malnutrition Diagnosis and Personalisation of Nutrition Delivery
The advent of a range of novel and innovative technologies to allow objective diagnosis of malnutrition, accurate determination of nutrition needs, and evaluation of nutrition utilisation is a major advance in the nutritional and metabolic care of critically patients. We believe it is essential that some or all of these devices are utilised in all future critical care nutrition trials to assess and guide malnutrition diagnosis and nutrition therapy. All of these devices continue to require further research to better evaluate their optimal application in ICU outcomes and care. Many of these trials are planned or underway. It is our dream that one day the muscle-specific ultrasound, the SBIS, and the new generation of metabolic carts will become to the ICU dietitian what the pulse oximeter, blood pressure cuff, and stethoscope are to the ICU nurse. We believe this will finally usher in an era of truly personalised nutrition care.
Key points
- ICU acquired weakness (ICU-AW) is a common complication of critical illness.
- An increasing number of patients who survive ICU suffer from severe, prolonged functional disabilities.
- Priority must be given to research addressing post-ICU QoL outcomes in survivors.
- One low-cost therapeutic strategy that can be rapidly implemented is objective, data guided personalised nutrition delivery to attempt to maintain and allow recovery of muscle mass/function.
- The use of a quick non-invasive technique to evaluate skeletal muscle quantity and quality in ICU patients could have profound prognostic implications for how malnutrition is diagnosed.
Abbreviations
ARF Acute Renal Failure
ASMI Appendicular Skeletal Muscle Index
BIA Bioelectrical Impedance
ECW Extracellular Water
EE Energy Expenditure
FFM Fat-Free Mass
GLIM Global Leadership Initiative on Malnutrition
IC Indirect Calorimetry
ICU Intensive Care Unit
ICU-AW ICU Acquired Weakness
ICW Intracellular Water
IMAT Intramuscular Adipose Tissue
IMGC Intramuscular Glycogen Content
LBM Lean Body Mass
MS Muscle Size
MV Mechanical Ventilation
REE Resting Energy Expenditure
SBIS Segmental Bioelectrical Impedence Spectroscopy
U/S Ultrasound
QoL Quality of Life
Conflict of Interest
Paul Wischmeyer reported receiving grant funding related to this work from National Institutes of Health, Canadian Institutes of Health Research, Nutricia, Abbott, Baxter, Fresenius, and Takeda. Dr. Wischmeyer has served as a consultant to Abbott, Fresenius, Baxter, Cardinal Health, Nutricia, and Takeda for research related to this work. Dr. Wischmeyer has received unrestricted gift donation for nutrition research from Musclesound and Cosmed. Dr. Wischmeyer has received honoraria or travel expenses for CME lectures on improving nutrition care from Abbott, Baxter and Nutricia.
Jeroen Molinger has received grant funding related to this work from Nutricia and Baxter. He serves as consultant for Musclesound Inc. and Nutricia.
Akazawa N et al. (2018) Muscle mass and intramuscular fat of the quadriceps are related to muscle strength in non-ambulatory chronic stroke survivors: A cross-sectional study. PLoS One, 13(8):e0201789.
Arbeille P et al. (2009) Quantification of muscle volume by echography: comparison with MRI data on subjects in long-term bed rest. Ultrasound Med Biol, 35(7):1092-7.
Barker LA, Gout BS, Crowe TC (2011) Hospital malnutrition: prevalence, identification and impact on patients and the healthcare system. Int J Environ Res Public Health, 8(2):514-27.
Barr J et al. (2004) Outcomes in critically ill patients before and after the implementation of an evidence-based nutritional management protocol. Chest, 125(4):1446-57.
Bear DE, Parry SM, Puthucheary ZA (2018) Can the critically ill patient generate sufficient energy to facilitate exercise in the ICU? Current Opinion in Clinical Nutrition and Metabolic Care, 21(2):110-115.
Binnekade JM et al. (2005) Daily enteral feeding practice on the ICU: attainment of goals and interfering factors. Crit Care, 9(3):R218-25.
Butterworth CE Jr (2005) [The skeleton in the hospital closet. 1974]. Nutr Hosp, 20(4)302-7, 301; discussion 297-300.
Cahill NE et al. (2010) Nutrition therapy in the critical care setting: what is "best achievable" practice? An international multicenter observational study. Critical Care Medicine, 38(2):395-401.
Cederholm T et al. (2019) GLIM criteria for the diagnosis of malnutrition - A consensus report from the global clinical nutrition community. Clin Nutr, 38(1):1-9.
Corkins MR et al. (2014) Malnutrition diagnoses in hospitalized patients: United States, 2010. JPEN J Parenter Enteral Nutr, 38(2):186-95.
Correa-de-Araujo R et al. (2017) The Need for Standardized Assessment of Muscle Quality in Skeletal Muscle Function Deficit and Other Aging-Related Muscle Dysfunctions: A Symposium Report. Front Physiol, 8:87.
Cruz-Jentoft AJ et al. (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing, 48(1):16-31.
da Silveira LTY et al. (2018) Decline in functional status after intensive care unit discharge is associated with ICU readmission: a prospective cohort study. Physiotherapy.
De Jonghe B et al. (2001) A prospective survey of nutritional support practices in intensive care unit patients: what is prescribed? What is delivered? Crit Care Med, 29(1):8-12.
Dinglas VD et al. (2017) Muscle Weakness and 5-Year Survival in Acute Respiratory Distress Syndrome Survivors. Crit Care Med, 45(3):446-453.
Ferrie S et al. (2016) Protein Requirements in the Critically Ill: A Randomized Controlled Trial Using Parenteral Nutrition. JPEN J Parenter Enteral Nutr, 40(6):795-805.
Fragala MS, Kenny AM, Kuchel GA (2015)
Muscle quality in aging: a multi-dimensional approach to muscle functioning
with applications for treatment. Sports Med, 45(5)641-58.
Fraipont V, Preiser JC (2013) Energy
estimation and measurement in critically ill patients. JPEN J Parenter Enteral
Nutr, 37(6):705-13.
Guttormsen
AB, Pichard C (2014) Determining
energy requirements in the ICU. Curr Opin Clin Nutr Metab Care, 17(2):171-6.
Heidegger CP et al. (2013) Optimisation of
energy provision with supplemental parenteral nutrition in critically ill
patients: a randomised controlled clinical trial. Lancet, 381(9864):385-93.
Herridge MS et al. (2016) Recovery and
outcomes after the acute respiratory distress syndrome (ARDS) in patients and
their family caregivers. Intensive Care Med, 42(5):725-738.
Heyland DK et al. (2003) Nutrition support
in the critical care setting: current practice in canadian ICUs--opportunities
for improvement? JPEN J Parenter Enteral Nutr, 27(1):74-83.
Hill JC, Millan IS (2014) Validation of
musculoskeletal ultrasound to assess and quantify muscle glycogen content. A
novel approach. Phys Sportsmed, 42(3):45-52.
Hopkins RO et al. (2017) Instrumental
Activities of Daily Living after Critical Illness: A Systematic Review. Ann Am
Thorac Soc, 14(8):1332-1343.
Kaido T et al. (2012) Pre- and
perioperative factors affecting infection after living donor liver
transplantation. Nutrition, 28(11-12):1104-8.
Kaukonen KM et al.(2014) Mortality related
to severe sepsis and septic shock among critically ill patients in Australia
and New Zealand, 2000-2012. JAMA, 311(13):1308-16.
Kelley GA, Kelley KS (2017) Is sarcopenia
associated with an increased risk of all-cause mortality and functional
disability? Experimental Gerontology, 96:100-103.
Kress JP, Hall JB (2014) ICU-acquired weakness
and recovery from critical illness. N Engl J Med, 370(17):1626-35.
Krishnan JA et al. (2003) Caloric intake in
medical ICU patients: consistency of care with guidelines and relationship to
clinical outcomes. Chest, 124(1):297-305.
Looijaard WG et al. (2016) Skeletal muscle
quality as assessed by CT-derived skeletal muscle density is associated with
6-month mortality in mechanically ventilated critically ill patients. Crit
Care, 20(1):386.
Looijaard W, Molinger J, Weijs PJM (2018)
Measuring and monitoring lean body mass in critical illness. Curr Opin Crit
Care, 24(4):241-247.
McClave SA et al. (2014) Feeding the
critically ill patient. Crit Care Med,42(12):2600-10.
McNelly AS et al. (2016) An Exploratory
Study of Long-Term Outcome Measures in Critical Illness Survivors: Construct
Validity of Physical Activity, Frailty, and Health-Related Quality of Life
Measures. Crit Care Med, 44(6):e362-9.
Needham DM et al. (2017) Core Outcome
Measures for Clinical Research in Acute Respiratory Failure Survivors. An
International Modified Delphi Consensus Study. Am J Respir Crit Care Med,
196(9):1122-1130.
Norman K et al. (2008) Prognostic impact of
disease-related malnutrition. Clin Nutr, 2(1):5-15.
Pardo E et al (2018) Reliability of
ultrasound measurements of quadriceps muscle thickness in critically ill
patients. BMC Anesthesiol, 18(1):205.
Parry SM et al. (2015) Ultrasonography in
the intensive care setting can be used to detect changes in the quality and
quantity of muscle and is related to muscle strength and function. J Crit Care,
30(5):1151 e9-14.
Puthucheary ZA et al. (2013) Acute skeletal
muscle wasting in critical illness. JAMA, 310(15):1591-600.
Puthucheary ZA et al. (2018) Metabolic
phenotype of skeletal muscle in early critical illness. Thorax.
Rubinson L et al. (2004) Low caloric intake
is associated with nosocomial bloodstream infections in patients in the medical
intensive care unit. Crit Care Med, 32(2):350-7.
Sieber CC (2017) Frailty - From concept to
clinical practice. Exp Gerontol, 87(Pt B):160-167.
Stapel SN et al. (2018) Bioelectrical
impedance analysis-derived phase angle at admission as a predictor of 90-day
mortality in intensive care patients. Eur J Clin Nutr, 72(7):1019-1025.
Taylor BE et al. (2016) Guidelines for the
Provision and Assessment of Nutrition Support Therapy in the Adult Critically
Ill Patient: Society of Critical Care Medicine (SCCM) and American Society for
Parenteral and Enteral Nutrition (A.S.P.E.N.). Crit Care Med, 44(2):390-438.
Thibault R et al. (2016) Fat-free mass at
admission predicts 28-day mortality in intensive care unit patients: the
international prospective observational study Phase Angle Project. Intensive
Care Med, 42(9):1445-53.
Watanabe Y et al. (2013) Echo intensity
obtained from ultrasonography images reflecting muscle strength in elderly men.
Clinical Interventions in Aging.
Weijs P et al. (2014) Low skeletal muscle
area is a risk factor for mortality in mechanically ventilated critically ill
patients-annotated. Crit Care,(18:R12).
Wischmeyer PE (2016) Are we creating
survivors...or victims in critical care? Delivering targeted nutrition to
improve outcomes. Curr Opin Crit Care, 22(4):279-84.
Wischmeyer PE (2018) Nutrition Therapy in
Sepsis. Crit Care Clin, 34(1):107-125.
Wischmeyer PE et al. (2017) Muscle mass and
physical recovery in ICU: innovations for targeting of nutrition and exercise.
Curr Opin Crit Care, 23(4):269-278.
Wischmeyer PE, San-Millan I (2015) Winning
the war against ICU-acquired weakness: new innovations in nutrition and
exercise physiology. Crit Care, 19(3):S6.