ICU Management & Practice, Volume 24 - Issue 3, 2024
Physiological monitoring has a rich 200-year history. In this article, the authors look into the components of an ideal monitoring system and highlight how advancements in modern technology could enable the development of an effective continuous monitoring and response system.
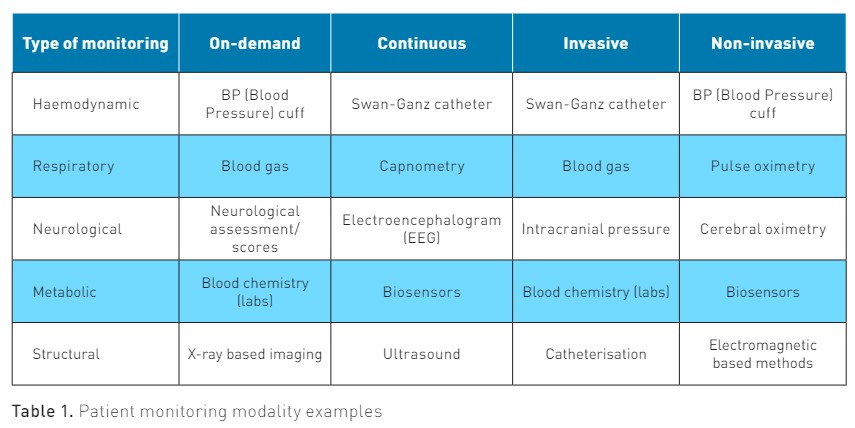
The continuous monitoring of patient vital signs is a required standard in acute care and procedural settings. In these settings, monitoring provides data that are an integral part of diagnostic and treatment decision-making. As the cost and complexity of this technology come down, the question of dissemination to other areas inside and outside the hospital is inevitable. After all, isn’t closer monitoring better? In this article, we will discuss the progress of continuous monitoring towards hospital-wide dissemination and why there is reason to hope that parallel advances in real-time analytics may allow us to overcome some of the traditional barriers to their widespread use as a valuable aid to clinical reasoning.
200 Years to Measure Blood Pressure – Monitoring Technology Takes Time to Refine
Heart rate, respiratory rate, and body temperature were three vital signs that could be measured without sophisticated instrumentation. During the first public demonstration of general anaesthesia by Dr Morton using ether in 1846 at Massachusetts General Hospital (MGH), Dr Heywood measured a patient’s heart rate as an indicator of the depth of anaesthesia. Blood pressure was thought to be a better marker, but at the time, only invasive techniques for measurement were known. One hundred years previously, Reverend Stephen Hales measured a horse's blood pressure by inserting a glass tube into the carotid artery, an invasive method that was considered too risky for routine use in patients. It wasn’t until 1855 when Von Vierordt of Tübingen introduced the first sphygmomanometer, that non-invasive blood pressure monitoring was possible. It took another 40 years for this technology to be refined (Riva-Rocci 1896) to the point where it was considered a sufficiently accurate method to measure systolic blood pressure and another ten years for Nikolai Korotkoff to establish a methodology to measure diastolic blood pressure with the aid of a stethoscope proximal to the Riva-Rocci cuff to identify the sounds associated with resumption of blood flow back into the previously occluded artery.
In the 1960s, Dr Maynard Ramsey III invented an automated oscillometric blood pressure monitor while he was a medical student at Duke University and created the company Applied Medical Research (AMR) to bring it to market. In 1976, AMR introduced the world's first microprocessor-based vital signs monitor, the "Device for Indirect Non-invasive Mean Arterial Pressure" (Dinamap 825). Thanks in part to the invention of intravascular catheters during the 1960s, continuous invasive arterial blood pressure monitoring became possible, and this remains the gold standard in continuous mean arterial blood pressure monitoring. From the 2000s to the present, there has been substantial investment in developing continuous non-invasive monitors of vital signs such as blood pressure. In general, these modern monitors rely on combinations of data elements such as heart rate (HR) and modified normalised pulse volume (mNPV), or pulse wave velocity (PWV), which involves a combination of an electrocardiogram (ECG) and a photoplethysmogram (PPG) to create their measurement. This approach has proven inaccurate and error-prone in non-ideal conditions, and at the time of writing, no FDA-certified medical-grade cuffless blood pressure monitors are available. This serves as a reminder of how difficult it can be to solve the engineering challenges associated with continuous physiologic monitoring.
Assuming engineers have done their job, and the device is accurately measuring physiologic data continuously and accurately, what’s next? How do you integrate the device into the workflow of the clinical decision makers? How do you avoid overburdening them with data that is not helpful? The challenges of dissemination and implementation are considerable for a new device coming to market. Who puts the device on the patient? What happens if there is missing data? Who interprets the data? What actions are expected?
What would be the composition of an ideal monitoring system?
- True representation of anatomical and physiological function of all human body organs and systems
- Direct measurement or accurate indirect representation
- Non-invasive rather than invasive with the ultimate goal of non-contact
- Automated interpretation and presentation in a convenient package within the workflow of clinical decision-makers
- Prediction of future changes
- Real-time coupling with a response system that can evaluate the patient and initiate a diagnostic workup or treatment plan as needed.
New Sensors
Most current physiological sensors are based on established principles of physics, chemistry and materials science. Developing a new generation of non-invasive, continuous sensors requires breakthroughs in basic science as well as innovation in our approach to data integration. Examples of these sensors include electrochemical stickers (Wang et al. 2022), field-effect transistor (FET) sensors (Panahi and Ghafar-Zadeh 2023), interstitial fluid (ISF) sensors (Wu et al. 2024), magneto-inductive sensors (Zhu et al. 2024), graphene-based sensors (Huang et al. 2019), and other modalities that are currently in active development. The practical implementation of such sensors can have a profound impact on our ability to monitor physiological parameters non-invasively and continuously.
Rise of Artificial Intelligence
Recent advances in machine learning and artificial intelligence (AI) create opportunities to move closer to an ideal monitoring system. Recall that it was a combination of two technologies and observable data elements that led to the breakthrough in non-invasive blood pressure monitoring. How can AI help us progress towards the ideal physiological monitoring solution?
- Dealing with indirect measurements. Many currently available or promising technologies do not directly measure parameters of interest. Machine learning can correlate these measurements with a gold standard and use them in production systems.
- Dealing with data resolution and missing data. Novel techniques can use less data in established models.
- Enabling predictive ability of monitoring system. This is a well-known and advertised ability of ML/AI. To create such a system with accuracy (95+%), multimodal data and a complex system should be created. Future biological modalities, like genetic, cell-level data, etc., could potentially be used.
- Automatic interpretation and ambient data representation. This area is advancing particularly rapidly today. AI can interpret complex or data-dense sensor outputs, such as the automatic interpretation of electrocardiogram (ECG) waveforms. AI can also handle massive amounts of data and package it in a convenient way.
Development of Integrative Monitoring Modality With Actionable Prediction
With the current state of sensor technologies, data availability, and ML methods, we have as many opportunities as limitations to advance patient monitoring to the next level. One such modality would be the prediction of patient deterioration and alerting for potential actionable problems that could prevent the escalation of care. The current gold standard in hospitals is the Rapid Response Team (RRT), which has well-established criteria for activation. There is evidence that vital signs could predict RRT activation within twelve hours of emergency department admission (Walston et al. 2016). Still, the development and production of an integrated real-time system has its own challenges:
- Routine collection of vital signs on the hospital floor and in locations outside of the ICU (Intensive Care Unit)
- High throughput computer network abilities allow for the screening of hundreds and thousands of patients in real-time.
High accuracy of prediction models enables acceptance by clinicians without increasing cognitive load.
The Path to Effective Continuous Monitoring and Response Systems
To be effective, continuous monitoring must be linked to an appropriate response system. It is the combination of awareness and response that yields health outcome benefits. It is important to acknowledge that the path to effective continuous monitoring has to address some significant potential failure points:
- Quality of device data (missing data, more noise than signal)
- Variable connectivity of devices (companies advance their own ecosystem)
- Quality of alerts (too sensitive, not specific)
- Variability in effector arm response (vague expectations, inadequate training, staffing)
- Resistance to change (stakeholders not engaged, lack of understanding)
- Poorly executed implementation (missing readiness to implement measures)
- Missing evaluation steps
- Lack of trust
Failure of any of these components will cause the entire enterprise to perform poorly. Indeed, the Society of Critical Care Medicine just published a white paper on "Recognizing and Responding to Clinical Deterioration Outside the ICU," in which they make "no recommendation regarding the use of continuous vital sign monitoring" for this purpose in non-ICU patients (Honarmand et al. 2024).
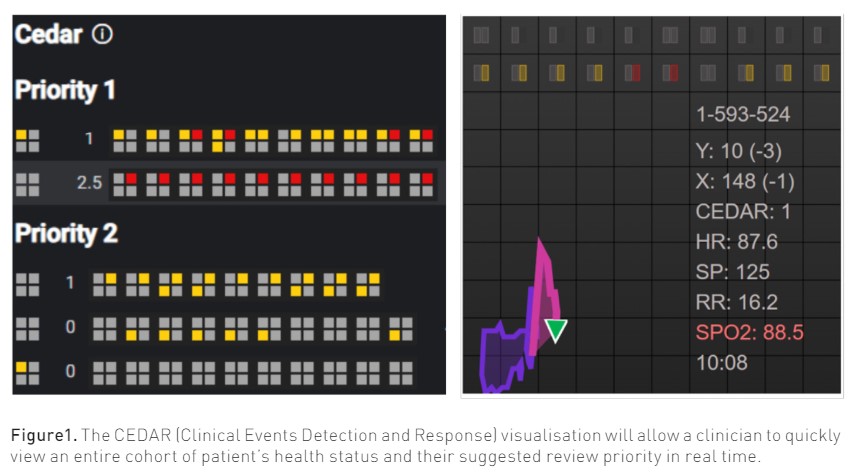
We are exploring one approach to solving some of these challenges in our own institution. With access to the longitudinal patient record, real-time streaming of continuous device data, and advanced analytics, we are developing the Clinical Emergent Events Detection and Response CEDAR (Clinical Events Detection and Response) system for detecting and responding to patients outside of the ICU setting with indicators of clinical deterioration. By combining electronic health record data with device data and a targeted clinical review, we intend to monitor hospitalised patients with the intention of assigning a priority for targeted clinical review.
Conclusion
We are at a point in the 200-year history of monitoring technology where we expect to see new sensing modalities and signal processing techniques combined with other data sources in an integrated detection and response system that brings us closer to the ideal paradigm of continuous non-invasive predictive human monitoring. Our hope is that it will be much faster these days.
Conflict of Interest
None.
References:
Honarmand K, Wax RS, Penoyer D et al. (2024) Society of Critical Care Medicine Guidelines on Recognizing and Responding to Clinical Deterioration Outside the ICU: 2023. Crit Care Med. 52(2):314-330.
Huang H, Su S, Wu N et al. (2019) Graphene-Based Sensors for Human Health Monitoring. Front Chem. 7:399.
Panahi A, Ghafar-Zadeh E (2023) Emerging Field-Effect Transistor Biosensors for Life Science Applications. Bioengineering (Basel). 10(7):793.
Walston JM, Cabrera D, Bellew SD et al. (2016) Vital Signs Predict Rapid-Response Team Activation Within Twelve Hours of Emergency Department Admission. West J Emerg Med. 17(3):324-30.
Wang M, Yang Y, Min J et al. (2022) A wearable electrochemical biosensor for the monitoring of metabolites and nutrients. Nat. Biomed. Eng. 6:1225-1235.
Wu Z, Qiao Z, Chen S et al. (2024) Interstitial fluid-based wearable biosensors for minimally invasive healthcare and biomedical applications. Commun Mater. 5:33.
Zhu Z, Estevez D, Feng T et al. (2024) A Novel Induction-Type Pressure Sensor based on Magneto-Stress Impedance and Magnetoelastic Coupling Effect for Monitoring Hand Rehabilitation. Small. e2400797.









