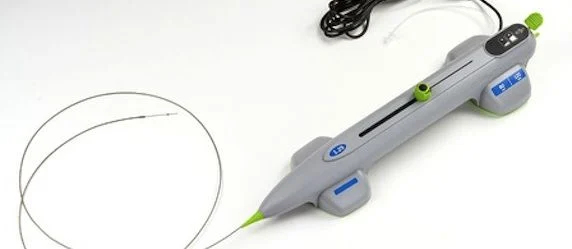
The Mount Sinai Hospital’s leading interventional cardiologists are the premiering the use of a newly FDA approved device to treat gravely calcified coronary arteries before placing a cardiac stent on a blocked artery.
The Diamondback 360® Coronary Orbital Atherectomy System is being introduced at The Mount Sinai Hospital’s Cardiac Catheterization Laboratory. It features a spinning, electrically powered 1.25 mm diamond-coated crown situated on a thin cardiac catheterization guide wire, and requires only seconds to complete the reduction of hard calcium buildup in a coronary artery. Small calcium particles are sanded from the artery's wall in this process, which are then naturally discarded from the heart and the body.
Manufactured by Cardiovascular Systems, Inc. this is the company’s first new coronary atherectomy system in more than 20 years.
Samin S. Sharma, MD, Director of Clinical and Interventional Cardiology at The Mount Sinai Hospital and the Zena, and Michael A. Wiener, Professor of Medicine at Icahn School of Medicine at Mount Sinai, are excited to be the first in the world to offer their patients this technological innovation which, they believe, will allow them to drastically decrease their patients' heart blockage percentage for more successful cardiac stent placement.
“We hope its use will facilitate improved outcomes for these patients with severely calcified blockages who are traditionally more challenging to treat", they conclude.
About a quarter of coronary artery disease patients needing percutaneous coronary intervention (PCI) treatment for a heart blockage have severe levels of arterial calcium lining their arteries. Up to now however, there have only been limited treatment options available for the arterial calcium removal. The increase in calcium can raise the risk of poor outcomes in these patients, including major adverse cardiac events (MACE) or increased mortality.
Dr. Sharma explains that when the Diamondback system was subjected to previous clinical trials such as the ORBIT II, results have shown the device to be effective and safe in treating severely calcified lesions. Furthermore, most patients were free from MACE following the trail procedure and Dr. Sharma now looks forward to offering greater safety to this complex patient population at Mount Sinai.
Dr. Sharma is the national Principal Investigator of the MACE study for the newly FDA approved device which will prospectively evaluate the economic outcomes of treating varying degrees of calcified blockages in the coronary arteries.
Source: Science Daily
29 October 2013
Latest Articles
Cardiology, PCI, Heart, arterial calcium
The Mount Sinai Hospital’s leading interventional cardiologists are the premiering the use of a newly FDA approved device to treat gravely calcified coro...