SMB's Objective: To reach out to all possible customers and service providers in developed as well as developing countries. SMB's Mission: To serve mankind by providing high quality products with innovative technologies and expertise ensuring safety of service providers & users. Company Profile: Dedicating 4 decades in manufacturing high quality products in the sphere of health care, accepted in more than 100 countries.
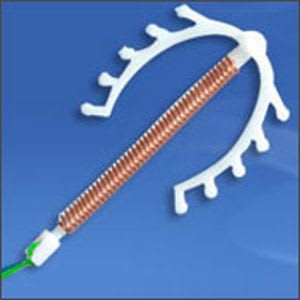
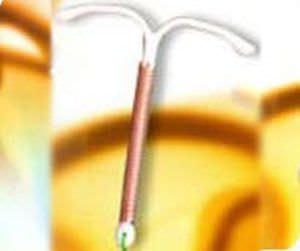
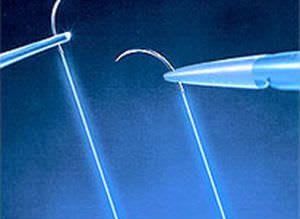
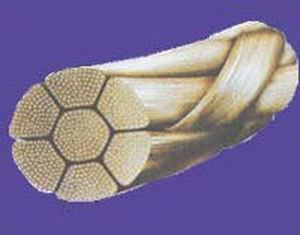
SMB a Star house exporter manufacturing versatile range of Medical Devices covering Intra-Uterine Devices (IUDs) IUD Thread Retriever Loading Device Hysterometer Non-Absorbable Surgical Sutures Tubal Rings Umbilical Cotton Tape Cytokit / Pap Smear Kit in world class manufacturing facility, under class 10,000 Clean Room Conditions right from manufacturing process of moulding to assembling. SMB facilities are Certified with ISO 9001:2008 / ISO 13485:2003 & WHO GMP.
All SMB products are CE Marked as per the latest directive of European Commission Medical Devices Guidance document MEDDEV 2.1/3 REV .3. Certifications: CE issued by Det Norske Veritas, Oslo for Intra Uterine Devices and Non-Absorbable Surgical Sutures. MEB (Medicines Evaluation Board), Netherlands Approval for IUDs ISO 9001:2008 / ISO 13485 : 2003 issued by Det Norske Veritas, Oslo. WHO GMP issued by FDA, Mumbai