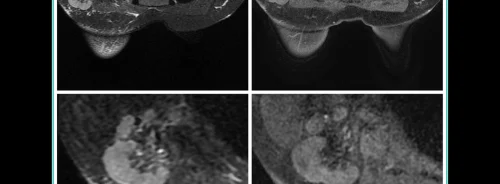
Researchers, Yashar Mashayekhi and Amirmohammad Heidari, from the University of Buckingham Medical School (UK) recently presented results from a study “A critical evaluation of Immunotherapeutic Agents for the Treatment of Triple Negative breast cancer.” In the United Kingdom, breast cancer is the most prevalent type in women; the subtype Triple Negative Breast Cancer (TNBC) is the most aggressive. This overview considers the most recent clinical trials involving immunotherapeutic targeting agents, their effectiveness and specific areas that require further research.
TCBN, a subtype of breast cancer that does not project oestrogen, progesterone or HER2 Receptors, accounts for 15% of all breast cancer cases (Bareche et al. 2020). Compared to other cancers, this type often shows a poorer prognosis and patients with metastatic TNBC experience an early onset of the disease. As TNBC is diverse in nature, treatment is limited to chemotherapy with a TAC regiment. Consequently, the treatment is short-lived and the overall survival period is 16 months on average.
In about 30% of TNBC tumours, the presence of tumour infiltrating lymphocytes combined with PD-L1 expression has shown improved immune check point inhibitors (ICI). Furthermore, the success of using immunotherapy agents to deal with other cancers that present similar tumour characteristics, for example, renal cell carcinoma and lung cancer, has paved the way to investigate immunotherapy in treating TNBC. The researchers thus focused on critically evaluating the advances in developing immunotherapeutic agents for TNBC.

Poly- ADP ribose polymerase (PARP inhibitors, Iniparib, veliparib)
A number of cancers, for instance pancreatic carcinoma, seem to be dependent on PARP pathways in comparison to regular healthy cells. For this reason, targeting therapy is a good choice. Currently, there are few trials testing PARP inhibitors on TNBC. However, a clinical trial in 2015 showed that three groups of patients either received only paclitaxel (PTX), paclitaxel combined with the parp inhibitor twice per week (PTI) or paclitaxel plus iniparib once per week (PWI). They found that the responses in all three groups were marginally similar, there was no significant change in anti-tumour activity or any induced toxicity.
Tyrosine kinase inhibitors (Cabozanitib)
It seems that MET, tyrosine kinase receptor leads to induced proliferation and invasion of cancer cells. it is not a good prognostic indicator in breast cancer patients either. Cabazonitib is a tyrosine kinase inhibitor (TKI) that inhibits MET and vascular endothelial growth factor receptor 2 (VEGFR2). Studies showed that cabozanitib helps to activate the immune system and prevents angiogenesis.
Drug Conjugates (Sacituzumab govitecan)
Sacituzumab govitecan (Trodelvy) is a conjugate of trop-2-directed antibody and topoisomerase inhibitor. It targets the trop-2 receptor that is linked to cancer cell proliferation and to topoisomerase inhibitor, which induces toxicity to cancer cells.
A successful phase I/II study in 2019 led to the FDA approving the use of this drug (Seligson et al., 2021). However, 32% of patients showed side-effects such as anaemia, hair loss and low white blood cell count. Despite these, conjugate drugs seem to offer a viable treatment of TNBC patients.
Clinical trials such as Impassion phase 3 and KEYNOTE-522 studies showed promising results. However, the efficacy of immunotherapeutic agent in treating TNBC is still a matter of concern. Although the combined use of ICIs such as PD-L1 Inhibitors with chemotherapy agents offer some hope, it still seems that the combination of surgery, radiotherapy and chemotherapy remain the most reliable treatment for TNBC patients. The researchers recommend that the emphasis should be on developing biomarkers for TNBC, which could lead to subdivision and categorisation of tumours which can aid in selecting more specific targeting agents and improving treatment.
Source: The British Institute of Radiology
Image credit: iStock
References:
Bareche Y, Buisseret L, Gruosso T, Girard E, Venet D, Dupont F, Desmedt C, Larsimont D, Park M, Rothé F, Stagg J, Sotiriou C. Unraveling Triple-Negative Breast Cancer Tumor Microenvironment Heterogeneity: Towards an Optimized Treatment Approach. J Natl Cancer Inst. 2020 Jul 1;112(7):708-719. doi: 10.1093/jnci/djz208. PMID: 31665482
Seligson ND, Knepper TC, Ragg S, Walko CM. Developing Drugs for Tissue-Agnostic Indications: A Paradigm Shift in Leveraging Cancer Biology for Precision Medicine. Clin Pharmacol Ther. 2021 Feb;109(2):334-342. doi: 10.1002/cpt.1946.