HealthManagement, Volume 22 - Issue 4, 2022
The improvement in mortality outcomes of patients with sepsis is attributable to the early recognition of sepsis, better adherence to guidelines, and a prompt organisation's responsiveness. However, sepsis remains the leading cause of death in the intensive care units and long-term sepsis mortality is still a burden for our healthcare system. In fact, managing infections in a critical area are increasingly complex and today more than ever requires a shared multidisciplinary and structured approach. In order to further improve outcomes, it is vital to highlight the importance of defining uniquely and clearly what is sepsis, improving early recognition strategy, and increasing our understanding of innate and adaptive immune system derangements that facilitate the development of sepsis. A deeper understanding of complex interactions between the infectious insult and the host's immune response warrants interesting implications at diagnostic and therapeutic levels, allowing to tailor the treatment to patients’ characteristics and to certain phases of the disease.
The Importance of Definition
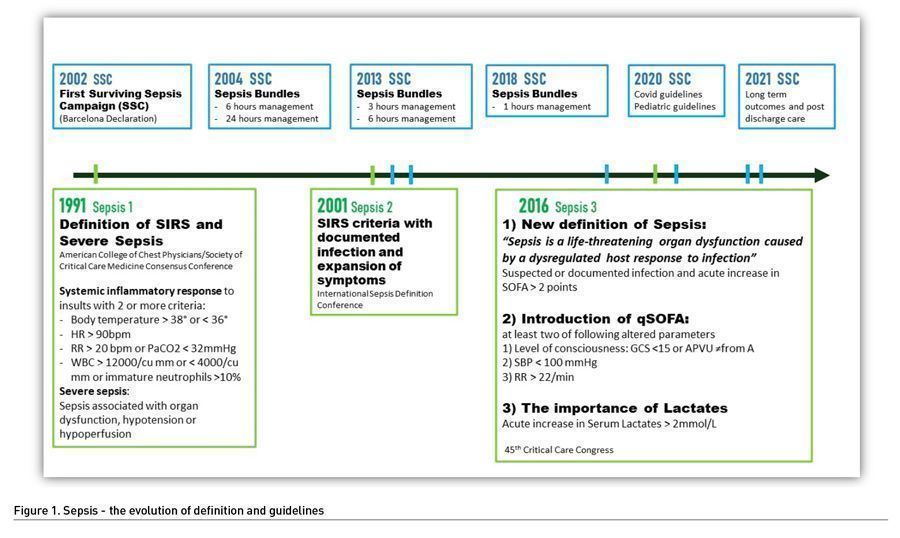
Sepsis is a "time dependent" disease, the clinical outcome of which depends on the speed of recognition and the effectiveness of clinical management starting from the first hour. The definition of sepsis has constantly changed over the time. This is due to the importance of defining uniquely and clearly what is sepsis for rapid recognition and treatment, in a standardised way.
In 1991, the initial definition of sepsis and septic shock was proposed during the American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference (Bone et al. 1992). Sepsis was defined as a host systemic inflammatory response syndrome (SIRS) to infection. SIRS was defined as a syndrome characterised by at least two of the following parameters: temperature >38° C or <36° C, heart rate > 90 beats per minute, respiratory rate >20 breaths per minute or CO2 partial pressure <32mmHg, white blood cell count >12,000/ml or <4,000/ml or >10% immature forms (band). Severe sepsis was sepsis complicated by organ dysfunction and septic shock was defined as sepsis characterised by persistent hypotension despite adequate fluid resuscitation.
In 2001, a task force of 29 participants from Europe and North America gathered during the International Sepsis Definition Conference with the aim of revisiting sepsis and septic shock definition. Due to the lack of strong evidence, the expert panel concluded not to change the definition of sepsis and septic shock; however, they expanded the list of signs and symptoms of septic panel (Ley et al. 2003). The use of the SIRS concept for the definition of sepsis is burdened by the problem of poor specificity, as multiple non-infectious clinical conditions can be associated with a picture of SIRS. In the emergency room, many patients have criteria compatible with SIRS, but only some have an associated infection, while on the other hand an infection does not always lead to a systemic inflammatory response. Furthermore, the criteria developed for the II definition presented the problem of being difficult to apply in the real world, both due to the complexity deriving from the number of parameters considered, and to the scarce specificity of the criteria.
In an attempt to overcome these problems and to identify more agile tools to be used in clinical practice, in 2016, during the 45th Critical Care Congress, the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) was proposed. Sepsis was defined as “a life-threatening organ dysfunction caused by a dysregulated host response to infection”. This new definition revolutionises the previous criteria, overcoming the concept of SIRS and clearly highlighting the importance of the interaction between the host and pathogen with consequent host response (local or systemic). The term “dysregulated” can be conceptualised as an anomalous response of the host that, independently of the infection, might induce organ damage. The immune imbalance resulting from this altered response is characterised by multiple concomitant proinflammatory and immunosuppressive aberrations involving both the innate and the adaptive immune response systems (Delano and Ward 2016). The loss of the normal homeostasis between the pro and anti-inflammatory system results in uncontrolled activation of the inflammatory response leading to the development of progressive physiologic dysfunction in two or more organs (Chen et al. 2018).
The other fundamental change concerned the introduction of the quick SOFA (qSOFA). The qSOFA is based on the use of three objective parameters: altered level of consciousness (GSC <15 or AVPU ≠ from A), PAS <100 mmHg, FR >22/min. In the presence of at least two of these parameters altered in the presence of infection, the suspicion of sepsis arises; in these patients the risk of death is high, and it is correct to implement the appropriate management protocols. Another important aspect introduced in 2016 was the concept of the importance of lactate. An increased level of serum lactate is a clear sign of tissue hypoperfusion and septic shock and is useful for early diagnosis. The usual cut-off value for an abnormal lactate level is >2 mmol/L.
In parallel, Surviving Sepsis Campaign (SSC) was launched for the first time by the Society of Critical Care Medicine (SCCM), ESICM, and the International Sepsis Forum during the ESICM annual meeting in Barcelona in 2002. In 2003, representatives of several international societies assembled and started to develop the guidelines for the management of sepsis and septic shock. The first and second edition of the SSC guidelines was published in Critical Care Medicine and Intensive Care Medicine (Dellinger et al. 2004; Dellinger et al. 2008). Guidelines implementation was further realised in 2012 and 2016, along with revised bundles (Dellinger et al. 2013; Rhodes et al. 2017). Finally, in 2021, the Surviving Sepsis Campaign updated the international guideline for management of sepsis and septic shock. The latest version put a special light on the evaluation of post discharge care, long-term outcome, and rehabilitation issues involving patients and their families (Evans et al. 2021). Despite progress in disease definition, there are still opportunities for improvement especially in light of the new understanding of the central role of proinflammatory and immunosuppressive aberrations and possible new diagnostic tools (i.e., immunomonitoring) as described below.
An overview of the evolution of sepsis definition and guidelines is provided in Figure 1.
The Importance of Preventing Sepsis and Early Identification of Patients at High Risk
In 2017, a Resolution of the World Health Assembly invited member states to act "for the improvement of the prevention, diagnosis and clinical management of sepsis"(Kilpatrick et al. 2018). The resolution highlighted the need for the prevention of healthcare-related infections, the correct use of antibiotics, and the necessity of training operators on the risk of progression from infection to sepsis. In fact, the response to sepsis can be carried out thanks to an integrated path that expresses the various functions in relation to the different settings. Managing infections in a critical area are increasingly complex and today more than ever requires a shared multidisciplinary and structured approach (Silal 2021). Preventing sepsis means preventing infection, through the management of risk factors that make it possible to frame the infection in the context (healthcare-related infections and lifestyles) and in the response of the host. It is, therefore, necessary to consider both the risk factors (i.e., frailty, comorbidities) and the patient's clinical history (i.e., recent surgery, immune response).
An important component of the sepsis and septic shock management concerns the identification of infectious risk and choosing the appropriate diagnostic and therapeutic pathways (Leong et al. 2021). The identification of infectious risk situations and the severity of sepsis is based on the use of three types of tools: the risk factors of infection, the severity score and the bioscore.
The identification of risk factors aims to identify the presence of predisposing factors. The reconstruction of the recent medical history (i.e., recent hospitalisation- the risk of colonisation) and the evaluation of the environment in which the patient lives (e.g., family infection cluster, local recent bacteria outbreak) aim to search for all those events that may increase the risk of sepsis and septic shock. The severity scores detect the deterioration of vital signs and are calculated at the time of contact between healthcare professionals and the patient. The severity scores (i.e., NEWS, MEOWS, qSOFA, SOFA) aim to systematically detect and quantify the deterioration of the vital signs of the patient and, therefore, they help the healthcare professional to assess how severe the health status of the patient is compromised (Sricharoen et al. 2022; Latten et al. 2021). The severity scores are intended to frame a state of instability and to trigger the appropriate treatment responses. Finally, the bioscore combines biomarkers (i.e., PCT, lactate), prognostic and infectious risk scores in a multiparametric way (Jekari et al. 2019; Song et al. 2019). This tool intends to direct the patient to the most appropriate path, based on the processing of all the information collected. The integration of the information collected allow to stratify the risk and to initiate sustainable and consistent diagnostic, antibiotic and sepsis stewardship paths.
The early identification of patients with sepsis and the evaluation of individual risk factors (i.e., immunodepression, long hospitalisation), are also extremely important for the best choice of microbiological test and for evaluating long-term outcome. Being as early as possible in formulating a diagnosis and establishing a rapid and adequate therapy is, undoubtedly, one of the most impacting aspects of the outcome of our patients. It is the clinician's task to identify which patients will benefit the most from new diagnostic methods (i.e., fast microbiology), mainly through multiparametric pathways of clinical reasoning, including, among other things, the advanced use of biomarkers (i.e., procalcitonin) (Mangioni et al. 2020). Fast microbiology allows the evaluation of antibiotic resistance with a faster prescription of the appropriate antibiotic treatment, and prompt de-escalation strategy (Muzzi et al. 2022).
Even more, it is worth remembering the long-term outcome and the rate of hospital readmission in patients who experienced septic shock (Prescott and Costa 2018). After sepsis, patients experience several common persisting complaints such as mental health problems (e.g., anxiety, depression, post-traumatic stress disorder, difficulty in thinking clearly, sleep disorders), muscle weakness, asthenia, difficulty swallowing, greater risk of contracting further infections, heart or kidney failure (Shankar-Hari et al. 2019; Sekino et al. 2022). Subsequent hospitalisation of patients who experienced sepsis is high and it is mainly due to the fact that sepsis is responsible for impairing immune system and accelerating the progression of pre-existing chronic conditions (Schmidt et al. 2020). Consequently, it is extremely important to evaluate the medical history of the patient experiencing sepsis and to individuate fragility patients especially, at risk of the long-term consequences of sepsis in order to create and support specific post-discharge rehabilitation treatment from hospitalisation for sepsis (e.g., physiotherapy), to identify new physical/cognitive problems and initiate appropriate treatment and to review and adjust long-term drug treatments.
The Importance of Understanding the Central Role of Immune Dysfunction
Given that the centrality of the complex interactions between the infectious insult and the host's immune response is now well established, the investigators' attention cannot help but turn to the mechanisms involved in the dysregulation of the innate and adaptive immune response. Consequently, immune modulation strategies appear as promising adjuvant therapies in septic patients (Evans et al. 2021; Shoji and Opal 2021; Hennessy et al. 2010).
It is not only the pro-inflammatory responses that determine the outcome of an infection, but the final result of the process depends on the homeostasis that is established between pro-inflammatory and anti-inflammatory mechanisms, leading either to the resolution of the infection or to its evolution with organ damage and possible secondary infections. An exaggerated and uncontrolled activation of the inflammatory response, resulting from an alteration of the balance between pro- and anti-inflammatory stimuli that is necessary for proper healing, can result in the inability to confine the flogosis to the site of the primary insult, thus causing diffuse tissue damage and multiple organ failure (MOF). The cellular pathophysiology behind the transition from a circumscribed infection to a systemic syndrome with deep organ derangement still remains largely to be elucidated. It is now clear that the individual genomic profile, through the upregulation or downregulation of genes involved in the inflammatory response, is partly responsible for its inter-individual variability (Sapan et al. 2017). Certain allelic polymorphisms have been shown to be strongly correlated with the development of secondary complications and an increased likelihood of MOF (Qiu et al. 2019; Lu et al. 2019). Epigenetic modifications are also able to influence the extent of intercellular crosstalk, contributing to the spread of inflammation (Namath and Patterson 2011).
Among the families of genes that influence the individual response to an insult is that of pattern recognition receptors (PRRs), which are expressed by many cells of the innate immune system rendering them capable of recognising highly conserved motifs recurring in microbe structure, also known as pathogen-associated molecular pattern (PAMPs), and danger-associated molecular pattern molecules (DAMPs), which are molecules released by injured host cells that can start a non-infectious inflammatory response (Rajaee et al. 2018; Mogensen 2009; Grazioli and Pugin 2018). Among the PRRs, toll-like receptors (TLRs) and nucleotide-binding domain leucine-rich repeat containing (NLR) receptors, are the most represented (Ye and Ting 2008). The interaction between PRRs and one of their ligands triggers the activation of a complex transcriptional programme, including the nuclear transcription factor NF-kB, with consequent mediates the expression of genes that code for cytokines, chemokines, nitric oxide and other mediators. A total of ten TLRs are described and differ from each other in their localisation and for their ability to recognise microbial antigens. The possibility for some TLRs to bind different microbial antigens and the presence of multiple antigens on the same germ broaden the range of possible inflammatory cascades that can be activated in response to an infection, further accentuating the interindividual variability (Latz et al. 2004; Wong et al. 2016).
Furthermore, the vascular endothelium is an important effector of the non-specific response, as it is reached by multiple activation signals that cause vasodilation and which are responsible for a series of complex interactions between endothelial cells and immune cells that facilitate the adhesion and migration of the latter through the endothelium to reach the site of interest. The price to pay is endothelial damage and consequent dysfunction due to the inflammatory response that is triggered by immune cells through the release of mediators such as nitric oxide and reactive oxygen species (ROS). Endothelial dysfunction is one of the cornerstones of the pathophysiology of MOF syndrome, paving the way for further tissue damage and organ failure (Ince et al. 2016). The infection is able to trigger also the complement cascade that in turn contributes to amplify the phenomena described so far (Ward 2010; Markiewski et al. 2008). In addition, endothelial activation with its consequences and the formation of the fibrin plug hinder the ability of germs to disseminate into the tissues through the systemic circulation (Levi and van der Poll 2010). Furthermore, the counter-regulatory pathways that inhibit coagulation are suppressed and fibrinolysis is inhibited overall (Levi and van der Poll 2017); the cascade of inflammation and coagulation stimulate and amplify each other, aggravating endothelial dysfunction and thus playing a crucial role in the onset of MOF (Levi 2010).
However, as already mentioned, in the pathophysiology of sepsis not only pro-inflammatory stimuli contribute, but also the anti-inflammatory forces have their own weight in the overall balance. In some patients, for reasons not yet fully understood which certainly depend on multiple factors, anti-inflammatory factors predominate overall, determining a condition of immunoparalysis, equally deleterious even if in different ways (Hamers et al. 2015; Boomer et al. 2011; Nierhaus et al. 2003). In both cases, however, it is evident that the in-depth knowledge of the complex interactions that govern the immune response could give some explanations on how to possibly intervene to regulate it, conditioning the evolution of sepsis towards its resolution.
Potential Future Role of Immune Monitoring
The increasing knowledge of the complexity of the pathophysiology of MOF and the growing awareness of the need for targeted therapy based on the individual patient's immune status is leading to a growing interest in immunomonitoring. In fact, the possibility of identifying clinical and laboratory markers that allow portraying the immune status of a patient, defining its degree of activation or suppression, could guide therapeutic choices with a possible impact also on the clinical outcome.
Much attention has been paid to the search for a correlation between the plasma levels of some key cytokines and clinical outcomes (Qiao et al. 2018; Kraft et al. 2015) (i.e., IL10/TNFα ratio). Data from trauma patient studies showed that the IL-6/IL-10 ratio is able to discriminate between more inflammatory response and one that tends toward immunoparalysis, showing in both cases correlation with MOF (Sapan et al. 2017). Finally, in burn patients, high levels of IL-8 correlated with an increased risk of MOF, sepsis, and mortality (Kraft et al. 2015). Nevertheless, none of these are routinely used to stage and monitor septic patients (Bozza et al. 2007). Even more, cytofluorometry allows the measurement of the level of expression of markers on the cell surface, emerging as a method to evaluate immune cell activation and immunophenotypic changes of cells during sepsis (Daix et al. 2018). It is now possible to measure the expression of CD64 on neutrophils, the expression of HLA-DR on monocytes and the percentage of circulating regulatory T lymphocytes. The HLA system molecules are involved in the presentation of antigen to the CD4 + T lymphocytes, leading to their activation and therefore triggering the adaptive immune response (Kessel et al. 2009). It is interesting to note that HLA-DR molecules exhibit a high level of conservation in those domains that are responsible for the interaction with conserved domains of T lymphocyte receptors, but at the same time show extensive polymorphisms in the domains responsible for binding to antigens and interacting with the variable regions of T-cell receptors (Jin and Wang 2003). This reflects the ability to enormously expand the number of antigens they are able to recognise, always preserving the ability to present them to the cells responsible for carrying out a specific response. Importantly, many studies show that reduced mHLA-DR expression is associated with adverse clinical outcomes, including increased rates of secondary infections and increased mortality, following various types of insults (e.g., trauma, burns, major surgery, and sepsis) (Hotchkiss et al. 2013; Monneret et al. 2006; Venet et al. 2013). Moreover, mHLA-DR levels can be used to assess the status of immunosuppression: patients with reduced levels of mHLA-DR should be considered immunosuppressed and therefore could benefit from targeted immune-enhancing therapies (Boomer et al. 2011; Boomer et al. 2014; Peters et al. 2018).
However, despite the extensive insights from the literature, it should always be kept in mind that measuring the concentration or expression of a single molecule provides only a partial picture of the patient's clinical situation, whereas results are capable of having an impact on the formulation of therapeutic strategies could derive from panels of markers which, unlike the single parameter, could provide greater details about the current immunity profile of the patient.
Potential Future Role of Immune Modulation Treatment
The possibility of systematically evaluating, in a certain phase of the disease, the state of the immune system of a patient opens up interesting implications also at therapeutic level, allowing to personalise the interventions and to evaluate their accuracy and effectiveness during therapy (Venet et al. 2013).
Despite the large arsenal of potential immune-modulating therapies available for the septic patient (Figure 2), the only one for which the international guidelines of the Surviving Sepsis Campaign 2021 maintain a recommendation in favour of its use are glucocorticoids (weak recommendation) (Evans et al. 2021). As for vitamin C, no recommendation could be made about its use in the septic patient: in fact, the large trials that have evaluated the effectiveness of administering high-dose vitamin C alone or in combination with thiamine and hydrocortisone did not show significant differences in mortality (Putzu et al. 2019; Fowler et al. 2019; Coloretti et al. 2020; Marik et al. 2017). Similarly, the international guidelines for the management of sepsis maintain the recommendation against the use of IVIGs in this clinical context (Rhodes et al. 2017; Evans et al. 2021). Given that low endogenous immunoglobulin levels in the septic patient correlate with a worse outcome (Venet et al. 2011), the use of immunoglobulins as an adjuvant therapy appeared promising right from the start. It seems that exogenous immunoglobulins not only act as agents capable of enhancing the proinflammatory response, but they act rather as modulators of the immune response as a whole (Lux et al. 2010), proving potentially useful not only in the early stages of sepsis but also in the late ones, characterised by profound immunoparalysis (Lux et al. 2010; Schmidt et al. 2021; Bermejo-Martin and Giamarellos-Bourboulis 2015). However, the overall balance of the desired and potentially harmful effects remains to be defined, given the non-negligible number of adverse reactions.
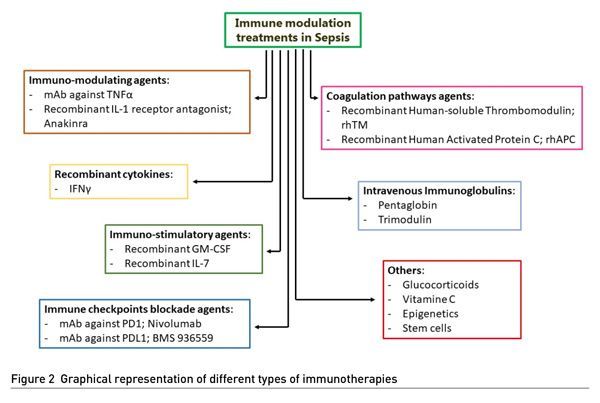
Potential new immune-modulating therapies are constantly increasing with controversial and contrasting results in vitro and vivo studies (Figure 2). Recombinant cytokines, pharmacological analogues of endogenous cytokines and of factors stimulating the growth of colonies, may potentially increase the immune response in the course of immune paralysis induced by sepsis. Studies conducted on animal models have shown that a defect in the production of IFN-γ increases secondary infections and mortality (Wang et al. 1994). In humans, IFN-γ levels have been found to be reduced in dying septic patients compared to surviving ones (Boomer et al. 2011). However, despite the encouraging data in animal studies, it is still unclear what the role of IFN-γ adjuvant treatment in clinical practice may be, as there is a lack of large clinical trials capable of demonstrating its real benefits (Jarvis et al. 2012; Turrel-Davin et al. 2011). Likewise, granulocyte-macrophage colony-stimulating factor (GM-CSF), a myelopoietic growth factor, has attracted great interest due to its immunomodulatory properties active on the innate system. Despite the promising results of preliminary studies involving the administration of rhGM-CSF, at the moment it is not possible to recommend the use of rhGM-CSF in clinical practice (Mathias et al. 2015; Bernstein et al. 2002; Bo et al. 2011). Given the key role played by some molecules in the cytokine storm during sepsis, interest has grown in monoclonal antibodies capable of selectively targeting them, so as to reduce their levels and somehow mitigate the deleterious inflammatory response. Anakinra represents an interesting example of monoclonal antibody against TNF-alpha and a recombinant antagonist of the IL-1 receptor (Qui et al. 2013; Abraham et al. 2001; Reinhart et al. 2001; Gallagher et al. 2001; Shakoory et al. 2016).
Considering the massive activation of the coagulation cascade during sepsis, interesting therapeutic implications seemed to derive from the use of recombinant human thrombomodulin (rhTM) and recombinant activated protein C (rhAPC), given the involvement of the corresponding endogenous molecules in the modulation of coagulation in an anticoagulant sense. RhTM seems to exert a protective effect on the development of damage to various organs during MOF (Yoshihiro et al. 2019; Yamakawa et al. 2015). RhAPC (drotrecogin alpha), on the other hand, exerts a set of pleiotropic effects by modulating not only the coagulation cascade, but also the inflammatory one, with a protective effect also on the endothelium (Levi and van der Poll 2007; Bernard et al. 2001).
Certain pathways involving immune checkpoints also play a role in sepsis-induced immunosuppression, therefore, the possibility of inhibiting them allows to enhance the immune response (Hotchkiss et al. 2019a). In sepsis-induced immunosuppression, the pathway involving programmed cell death protein-1 (PD-1) and its programmed cell death-ligand 1 (PD-L1) is upregulated (Hotchkiss et al. 2019b; Chang et al. 2013). The administration of nivolumab, a monoclonal antibody directed against PD-1 already used in the therapy of various tumours, in combination with the antibiotic meropenem can improve the clearance of the pathogen in septic patients compared to the administration of the antibiotic alone (Hotchkiss et al. 2019a; Gillis et al. 2021; Watanabe et al. 2020).
Another possible weapon to be exploited in the treatment of organ damage induced by sepsis derives from the field of regenerative medicine and consists of the use of mesenchymal stem cells. Mesenchymal stem cells not only act through the direct mechanism of replacing and soliciting cells in damaged tissues, but also exert potentially therapeutic effects through mechanisms that involve the release of cellular mediators, capable of modulating the inflammatory state and consequently the damage of organs (Lee et al. 2009; Keane et al. 2017). In particular, the release of microvesicles containing cellular components including mitochondria appears to play a complex role in the dialogue between cells, also allowing the exchange of material (Zhu et al. 2014). Some preclinical studies have produced evidence of the efficacy of MSCs in reducing organ damage induced by sepsis affecting the lung, heart and liver (Wang et al. 2015; Nong et al. 2016); however, the possibility of using MSCs in the therapy of the septic patient remains a very distant prospect (Keane et al. 2017).
Finally, the interaction between pathogen and host can induce important epigenetic changes, capable of influencing both the acute phase of sepsis and having an impact on its long-term sequelae including a prolonged inflammatory or immunoparalysis state (Falcão-Holanda et al. 2021; Cross et al. 2019); it can also determine a functional reprogramming of the transcriptional pathways of innate immunity cells which takes the name of "trained immunity" (Netea et al. 2020; Saeed et al. 2014; Netea et al. 2016; van der Heijden et al. 2018), thus favouring the memory of the infection (Moorlag et al. 2020). Given the attractive prospects opened by the possibility of regulating epigenetics, preclinical studies are underway which are evaluating various histone deacetylase inhibitors (i.e., HDACi), molecules already in use in cancer patients (i.e., Vorinostat, Givinostat) (Kim and Bae 2011; Mann et al. 2007).
Conclusions
The management of infection is increasingly complex and requires a common multidisciplinary approach. Central aspects of fighting sepsis are represented by the need for the prevention of healthcare-related infections, the correct use of antibiotics, and the necessity of training operators on the risk of progression from infection to sepsis.
The new definition revolutionises the previous criteria, overcoming the concept of SIRS and clearly highlighting the importance of the interaction between the host and pathogen. Consequently, the new understanding of the central role of proinflammatory and immunosuppressive aberrations opens up interesting diagnostic and therapeutic implications, theoretically leading to a tailored medicine. It is, therefore, necessary to consider both the risk factors and the patient's clinical history and to choose the appropriate diagnostic and therapeutic pathways.
Conflict of Interest
None.
References:
Abraham E, Laterre PF, Garbino J et al. (2001) Lenercept (p55 tumor necrosis factor receptor fusion protein) in severe sepsis and early septic shock: a randomized, double-blind, placebo-controlled, multicenter phase III trial with 1,342 patients. Crit Care Med. 29(3):503-10.
Bermejo-Martin JF, Giamarellos-Bourboulis EJ (2015) Endogenous immunoglobulins and sepsis: New perspectives for guiding replacement therapies. Int J Antimicrob Agents. 46 Suppl 1:S25-8.
Bernard GR, Vincent JL, Laterre PF et al. (2001) Efficacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med.344(10):699-709.
Bernstein HM, Calhoun DA, Christensen RD (2002) Use of myeloid colony-stimulating factors in neonates with septicemia. CurrOpinPediatr. 14(1):91-4
Bo L, Wang F, Zhu J, Li J, Deng X (2011) Granulocyte-colony stimulating factor (G-CSF) and granulocyte-macrophage colony stimulating factor (GM-CSF) for sepsis: a meta-analysis. Crit Care. 15(1):R58.
Boomer JS, To K, Chang KC et al. (2011) Immunosuppression in patients who die of sepsis and multiple organ failure. Jama. 306(23):2594-605.
Boomer JS, Green JM, Hotchkiss RS (2014) The changing immune system in sepsis: Is individualized immuno-modulatory therapy the answer? Virulence. 5(1):45-56.
Bone RC, Balk RA, Cerra FB et al. (1992) Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 101(6):1644-55.
Bozza FA, Salluh JI, Japiassu AM et al. (2007) Cytokine profiles as markers of disease severity in sepsis: a multiplex analysis. Crit Care. 11(2):R49.
Chang KC, Burnham CA, Compton SM et al. (2013) Blockade ofthe negative co-stimulatory molecules PD-1 and CTLA-4 improves survival in primary and secondary fungal sepsis. Crit Care. 17(3):R85.
Chen L, Deng H, Cui H et al. (2018) Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 9(6):7204-18.
Coloretti I, Biagioni E, Venturelli S et al. (2020) Adjunctive therapy with vitamin c and thiamine in patients treated with steroids for refractory septic shock: A propensity matched before-after, case-control study. J Crit Care. 59:37-41.
Cross D, Drury R, Hill J, Pollard AJ (2019) Epigenetics in Sepsis: Understanding Its Role in Endothelial Dysfunction, Immunosuppression, and Potential Therapeutics. Front Immunol. 10:1363.
Daix T, Guerin E, Tavernier E et al. (2018) Multicentric Standardized Flow Cytometry Routine Assessment of Patients With Sepsis to Predict Clinical Worsening. Chest. 154(3):617-27.
Delano MJ, Ward PA (2016) The immune system's role in sepsis progression, resolution, and long-term outcome. Immunol Rev. 274(1):330-53.
Dellinger RP, Carlet JM, Masur H et al. (2004) Surviving Sepsis Campaign guidelines for management of severe sepsis and septic shock. Crit Care Med. 32(3):858-73.
Dellinger RP, Levy MM, Carlet JM et al. (2008) Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Crit Care Med. 36(1):296-327.
Dellinger RP, Levy MM, Rhodes A et al. (2013) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 41(2):580-637.
Evans L, Rhodes A, Alhazzani W et al. (2021) Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 47(11):1181-247.
Falcão-Holanda RB, Brunialti MKC, Jasiulionis MG, Salomão R (2021) Epigenetic Regulation in Sepsis, Role in Pathophysiology and Therapeutic Perspective. Front Med (Lausanne). 8:685333.
Fowler AA, Truwit JD, Hite RD et al. (2019) Effect of Vitamin C Infusion on Organ Failure and Biomarkers of Inflammation and Vascular Injury in Patients With Sepsis and Severe Acute Respiratory Failure: The CITRIS-ALI Randomized Clinical Trial. Jama. 322(13):1261-70.
Gallagher J, Fisher C, Sherman B et al. (2001) A multicenter, open-label, prospective, randomized, dose-ranging pharmacokinetic study of the anti-TNF-alpha antibody afelimomab in patients with sepsis syndrome. Intensive Care Med. 27(7):1169-78.
Gillis A, Ben Yaacov A, Agur Z (2021) A New Method for Optimizing Sepsis Therapy by Nivolumab and Meropenem Combination: Importance of Early Intervention and CTL Reinvigoration Rate as a Response Marker. Front Immunol. 12:616881.
Grazioli S, Pugin J (2018) Mitochondrial Damage-Associated Molecular Patterns: From Inflammatory Signaling to Human Diseases. Front Immunol. 9:832.
Hamers L, Kox M, Pickkers P (2015) Sepsis-induced immunoparalysis: mechanisms, markers, and treatment options. Minerva Anestesiol. 81(4):426-39.
Hennessy EJ, Parker AE, O'Neill LA (2010) Targeting Toll-like receptors: emerging therapeutics? Nat Rev Drug Discov. 9(4):293-307.
Hotchkiss RS, Monneret G, Payen D (2013) Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat Rev Immunol. 13(12):862-74.
Hotchkiss RS, Colston E, Yende S et al. (2019a) Immune Checkpoint Inhibition in Sepsis: A Phase 1b Randomized, Placebo-Controlled, Single Ascending Dose Study of Antiprogrammed Cell Death-Ligand 1 Antibody (BMS-936559). Crit Care Med. 47(5):632-42.
Hotchkiss RS, Colston E, Yende S et al. (2019b) Immune checkpoint inhibition in sepsis: a Phase 1b randomized study to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of nivolumab. Intensive Care Med. 45(10):1360-71.
Ince C, Mayeux PR, Nguyen T et al. (2016) The endothelium in sepsis. Shock. 45(3):259-70.
Jarvis JN, Meintjes G, Rebe K et al. (2012) Adjunctive interferon-γ immunotherapy for the treatment of HIV-associated cryptococcal meningitis: a randomized controlled trial. Aids. 26(9):1105-13.
Jekarl DW, Lee S, Kim M et al. (2019) Procalcitonin as a prognostic marker for sepsis based on SEPSIS-3. J Clin Lab Anal. 33(9):e22996.
Jin P, Wang E (2003) Polymorphism in clinical immunology - From HLA typing to immunogenetic profiling. J Transl Med. 1(1):8.
Keane C, Jerkic M, Laffey JG (2017) Stem Cell-based Therapies for Sepsis. Anesthesiology. 127(6):1017-34.
Kessel A, Bamberger E, Masalha M, Toubi E (2009) The role of T regulatory cells in human sepsis. J Autoimmun. 32(3-4):211-5.
Kilpatrick C, Saito H, Allegranzi B, Pittet D (2018) Preventing sepsis in health care – It’s in your hands: A World Health Organization call to action. J Infect Prev. 19(3):104-6.
Kim HJ, Bae SC (2011) Histone deacetylase inhibitors: molecular mechanisms of action and clinical trials as anti-cancer drugs. Am J Transl Res. 3(2):166-79.
Kraft R, Herndon DN, Finnerty CC et al. (2015) Predictive Value of IL-8 for Sepsis and Severe Infections After Burn Injury: A Clinical Study. Shock. 43(3):222-7.
Latten GHP, Polak J, Merry AHH et al. (2021) Frequency of alterations in qSOFA, SIRS, MEWS and NEWS scores during the emergency department stay in infectious patients: a prospective study. Int J Emerg Med. 14(1):69.
Latz E, Schoenemeyer A, Visintin A et al. (2004) TLR9 signals after translocating from the ER to CpG DNA in the lysosome. Nat Immunol. 5(2):190-8.
Lee JW, Gupta N, Serikov V, Matthay MA (2009) Potential application of mesenchymal stem cells in acute lung injury. Expert OpinBiolTher. 9(10):1259-70.
Leong K, Gaglani B, Khanna AK, McCurdy MT (2021) Novel Diagnostics and Therapeutics in Sepsis. Biomedicines. 9(3).
Levi M, van der Poll T (2010) Inflammation and coagulation. Crit Care Med. 38(2 Suppl):S26-34.
Levi M, van der Poll T (2017) Coagulation and sepsis. Thromb Res. 149:38-44.
Levi M, van der Poll T (2007) Recombinant human activated protein C: current insights into its mechanism of action. Crit Care. 11(Suppl 5):S3.
Levi M (2010) The coagulant response in sepsis and inflammation. Hamostaseologie. 30(1):10-2, 4-6.
Levy MM, Fink MP, Marshall JC et al. (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 31(4):1250-6.
Lu H, Wen D, Wang X et al. (2019) Host genetic variants in sepsis risk: a field synopsis and meta-analysis. Crit Care. 23.
Lux A, Aschermann S, Biburger M, Nimmerjahn F (2010) The pro and anti-inflammatory activities of immunoglobulin G. Ann Rheum Dis. 69 Suppl 1:i92-6.
Mangioni D, Peri AM, Rossolini GM et al. (2020) Toward Rapid Sepsis Diagnosis and Patient Stratification: What's New From Microbiology and Omics Science. J Infect Dis. 221(7):1039-47.
Mann BS, Johnson JR, Cohen MH et al. (2007) FDA approval summary: vorinostat for treatment of advanced primary cutaneous T-cell lymphoma. Oncologist. 12(10):1247-52.
Marik PE, Khangoora V, Rivera R et al. (2017) Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Before-After Study. Chest. 151(6):1229-38.
Markiewski MM, DeAngelis RA, Lambris JD (2008) Complexity of complement activation in sepsis. J Cell Mol Med. 12(6a):2245-54.
Mathias B, Szpila BE, Moore FA et al. (2015) A Review of GM-CSF Therapy in Sepsis. Medicine (Baltimore). 94(50).
Mogensen TH (2009) Pathogen recognition and inflammatory signaling in innate immune defenses. Clin Microbiol Rev. 22(2):240-73, Table of Contents.
Monneret G, Lepape A, Voirin N et al. (2006) Persisting low monocyte human leukocyte antigen-DR expression predicts mortality in septic shock. Intensive Care Med. 32(8):1175-83.
Moorlag SJ, Khan N, Novakovic B et al. (2020) β-Glucan Induces Protective Trained Immunity against Mycobacterium tuberculosis Infection: A Key Role for IL-1. Cell Rep. 31(7).
Muzzi M, Viaggi B, Fabbri S et al. (2022) The Impact of Fast Microbiology in Intensive Care Units in the Era of Antibiotic Resistance: An Observational Retrospective Study. CurrMicrobiol. 79(3):79.
Namath A, Patterson AJ (2011) Genetic polymorphisms in sepsis. Crit Care Nurs Clin North Am. 23(1):181-202.
Netea MG, Domínguez-Andrés J, Barreiro LB et al. (2020) Defining trained immunity and its role in health and disease. Nat Rev Immunol. 20(6):375-88.
Netea MG, Joosten LA, Latz E et al. (2016). Trained immunity: a program of innate immune memory in health and disease. Science. 352(6284):aaf1098.
Nierhaus A, Montag B, Timmler N et al. (2003) Reversal of immunoparalysis by recombinant human granulocyte-macrophage colony-stimulating factor in patients with severe sepsis. Intensive Care Med. 29(4):646-51.
Nong K, Wang W, Niu X et al. (2016) Hepatoprotective effect of exosomes from human-induced pluripotent stem cell-derived mesenchymal stromal cells against hepatic ischemia-reperfusion injury in rats. Cytotherapy. 18(12):1548-59.
Peters van Ton AM, Kox M et al. (2018) Precision Immunotherapy for Sepsis. Front Immunol. 9.
Prescott HC, Costa DK (2018) Improving Long-term Outcomes after Sepsis. Crit Care Clin. 34(1):175-88.
Putzu A, Daems AM, Lopez-Delgado JC et al. (2019) The Effect of Vitamin C on Clinical Outcome in Critically Ill Patients: A Systematic Review With Meta-Analysis of Randomized Controlled Trials. Crit Care Med. 47(6):774-83.
Qiao Z, Wang W, Yin L et al. (2018) Using IL-6 concentrations in the first 24 h following trauma to predict immunological complications and mortality in trauma patients: a meta-analysis. Eur J Trauma Emerg Surg. 44(5):679-87.
Qiu P, Wang L, Ni J, Zhang Y (2019) Associations between HMGB1 gene polymorphisms and susceptibility and clinical outcomes in Chinese Han sepsis patients. Gene. 687:23-9.
Qiu P, Cui X, Sun J, Welsh J et al. (2013) Anti-Tumor Necrosis Factor Therapy is Associated with Improved Survival in Clinical Sepsis Trials: A Meta-analysis. Crit Care Med. 41(10).
Rajaee A, Barnett R, Cheadle WG (2018) Pathogen- and Danger-Associated Molecular Patterns and the Cytokine Response in Sepsis. Surg Infect (Larchmt). 19(2):107-16.
Reinhart K, Menges T, Gardlund B et al. (2001) Randomized, placebo-controlled trial of the anti-tumor necrosis factor antibody fragment afelimomab in hyperinflammatory response during severe sepsis: The RAMSES Study. Crit Care Med. 29(4):765-9.
Rhodes A, Evans LE, Alhazzani W et al. (2017) Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 43(3):304-77.
Sapan HB, Paturusi I, Islam AA et al. (2017) Interleukin-6 and interleukin-10 plasma levels and mRNA expression in polytrauma patients. Chin J Traumatol. 20(6):318-22.
Saeed S, Quintin J, Kerstens HH et al. (2014) Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. Science. 345(6204):1251086.
Schmidt K, Gensichen J, Fleischmann-Struzek C et al. (2020) Long-Term Survival Following Sepsis: Results of a Single-Center Registry Study With 4-Year Follow-Up. DtschArztebl Int. 117(46):775-82.
Schmidt C, Weißmüller S, Bohländer F et al. (2021) The Dual Role of a Polyvalent IgM/IgA-Enriched Immunoglobulin Preparation in Activating and Inhibiting the Complement System. Biomedicines. 9(7).
Sekino N, Selim M, Shehadah A (2022) Sepsis-associated brain injury: underlying mechanisms and potential therapeutic strategies for acute and long-term cognitive impairments. J Neuroinflammation. 19.
Shakoory B, Carcillo JA, Chatham WW et al. (2016) Interleukin-1 Receptor Blockade Is Associated With Reduced Mortality in Sepsis Patients With Features of Macrophage Activation Syndrome: Reanalysis of a Prior Phase III Trial. Crit Care Med. 44(2):275-81.
Shankar-Hari M, Harrison DA, Ferrando-Vivas P et al. (2019) Risk Factors at Index Hospitalization Associated With Longer-term Mortality in Adult Sepsis Survivors. JAMA Netw Open. 2(5).
Shoji H, Opal SM (2021) Therapeutic Rationale for Endotoxin Removal with Polymyxin B Immobilized Fiber Column (PMX) for Septic Shock. Int J Mol Sci. 22(4).
Silal SP (2021) Operational research: A multidisciplinary approach for the management of infectious disease in a global context. Eur J Oper Res. 291(3):929-34.
Song J, Park DW, Moon S et al. (2019) Diagnostic and prognostic value of interleukin-6, pentraxin 3, and procalcitonin levels among sepsis and septic shock patients: a prospective controlled study according to the Sepsis-3 definitions. BMC Infect Dis. 19(1):968.
Sricharoen P, Chueluecha C, Yuksen C, Jenpanitpong C (2022) The Use of qSOFA, SOFA, and Ramathibodi Early Warning Score (REWS) to Predict Severe Complications in Hematologic Malignancy Patients. Open Access Emerg Med. 14:51-61.
Turrel-Davin F, Venet F, Monnin C et al. (2011) mRNA-based approach to monitor recombinant gamma-interferon restoration of LPS-induced endotoxin tolerance. Crit Care. 15(5):R252.
van der Heijden CD, Noz MP, Joosten LA et al. (2018) Epigenetics and Trained Immunity. Antioxid Redox Signal. 29(11):1023-40.
Venet F, Lukaszewicz AC, Payen D et al. (2013) Monitoring the immune response in sepsis: a rational approach to administration of immunoadjuvant therapies. CurrOpin Immunol. 25(4):477-83.
Venet F, Gebeile R, Bancel J et al. (2011) Assessment of plasmatic immunoglobulin G, A and M levels in septic shock patients. Int Immunopharmacol. 11(12):2086-90.
Wang T, Town T, Alexopoulou L et al. (2004) Toll-like receptor 3 mediates West Nile virus entry into the brain causing lethal encephalitis. Nat Med. 10:1366-73.
Wang ZE, Reiner SL, Zheng S et al. (1994) CD4+ effector cells default to the Th2 pathway in interferon gamma-deficient mice infected with Leishmania major. J Exp Med. 179(4):1367-71.
Wang X, Gu H, Qin D et al. (2015) Exosomal miR-223 Contributes to Mesenchymal Stem Cell-Elicited Cardioprotection in Polymicrobial Sepsis. Sci Rep. 5:13721.
Ward PA (2010) Role of C5 activation products in sepsis. ScientificWorldJournal. 10:2395-402.
Watanabe E, Nishida O, Kakihana Y et al. (2020) Pharmacokinetics, Pharmacodynamics, and Safety of Nivolumab in Patients With Sepsis-Induced Immunosuppression: A Multicenter, Open-Label Phase 1/2 Study. Shock. 53(6):686-94.
Yamakawa K, Aihara M, Ogura H et al. (2015) Recombinant human soluble thrombomodulin in severe sepsis: a systematic review and meta-analysis. J ThrombHaemost. 13(4):508-19.
Ye Z, Ting JP (2008) NLR, the nucleotide-binding domain leucine-rich repeat containing gene family. CurrOpin Immunol. 20(1):3-9.
Yoshihiro S, Sakuraya M, Hayakawa M et al. (2019) Recombinant Human-Soluble Thrombomodulin Contributes to Reduced Mortality in Sepsis Patients With Severe Respiratory Failure: A Retrospective Observational Study Using a Multicenter Dataset. Shock. 51(2):174-9.
Zhu YG, Feng XM, Abbott J et al. (2014) Human mesenchymal stem cell microvesicles for treatment of Escherichia coli endotoxin-induced acute lung injury in mice. Stem Cells. 32(1):116-25.














