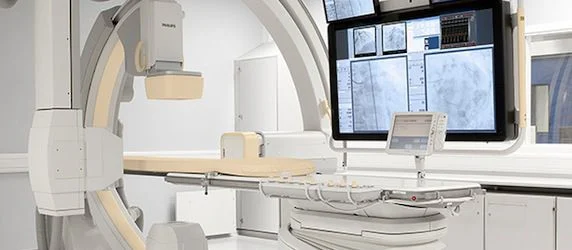
Philips has announced that it has received 510(k) clearance from the US Food and Drug Administration (FDA) to market its AlluraClarity live image guidance system in the US.
Philips’ AlluraClarity system provides high quality imaging for a comprehensive range of clinical procedures, achieving excellent visibility at low X-ray dose levels for patients of all sizes. To reflect the cost pressures that modern hospitals and health systems face, ClarityIQ technology will also be available as an upgrade for the majority of Philips’ installed base of monoplane and biplane interventional X-ray systems.
“All patients treated via X-ray guided interventions benefit from the advantage of low radiation exposure, but it is especially important when you are treating patients who have to undergo lengthy and complex procedures,” said Dr. Marco van Strijen, interventional radiologist at the St. Antonius Hospital Utrecht/Nieuwegein, the Netherlands. “We have been using Philips’ AlluraClarity system for more than a year now and have really grown to appreciate the low dose settings. This technology is making a difference where it really matters."
“The transition from highly invasive surgical procedures to minimally-invasive image-guided therapies, with all their intrinsic patient benefits, is a transformation in the delivery of healthcare that is rapidly accelerating around the globe,” said Gene Saragnese, CEO Imaging Systems at Philips Healthcare. “It is an area where technology innovation and procedure innovation go hand in hand. AlluraClarity is a perfect example of how Philips’ close collaboration with clinical partners has combined these two areas of innovation to facilitate more advanced treatment while at the same time managing radiation dose.”
Latest Articles
X-ray, Philips, Interventions
Philips has announced that it has received 510(k) clearance from the US Food and Drug Administration (FDA) to market its AlluraClarity live image guidance...