Determining the origin of cancer becomes difficult when tumour cells have already spread to other tissues (metastases). An investigational molecular imaging technique — combining positron emission tomography and computed tomography (PET/CT) — could be the key to finding the elusive primary tumour, according to a new study presented at the 2015 Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI).
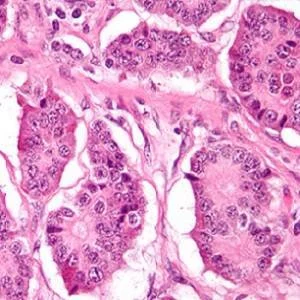
The PET/CT study focused on neuroendocrine tumours (NETs), a malignancy of nerve cells scattered throughout various organ systems that are sensitive to the signalling of neurotransmitters and hormones.
What makes the PET/CT technique novel is the use of an imaging agent known as Ga-68 DOTATOC, which binds to somatostatin receptors in neuroendocrine cells that use the hormone somatostatin to mediate endocrine function, the release of neurotransmitters and cellular proliferation. PET/CT with Ga-68 DOTATOC highlights malignant tumours because the agent binds to these more strongly than normal tissues.
Such an imaging technique could provide critical information that alters cancer patients' treatment. "Our study shows that Ga-68 DOTATOC PET/CT can change the management of patients with unknown primary tumour and neuroendocrine metastases, including potential surgical resection of the primary tumour," said lead author Yusuf Menda, MD, a researcher in the department of radiology and division of nuclear medicine at the University of Iowa. "There is evidence that surgery could lead to improved survival for these patients."
For the study, Ga-68 DOTATOC — short for gallium-68 1,4,7,10 tetraazacyclododecane-NI,NII,NIII,NIIII-tetraacetic acid (D)-Phe1-thy3 octreotide — was administered to 36 patients with metastatic NET and unknown primary tumour site. All patients underwent PET/CT imaging. Based on the results:
Overall, 28 percent of patients underwent a major change in cancer management as a result of Ga-68 DOTATOC PET/CT.
"Ga-68 DOTATOC and similar agents are investigational drugs in the United States and are not yet approved for clinical use," notes Dr. Menda. "They are being investigated in clinical trials across several U.S. institutions. It is hoped that these studies and previous European published experience will ultimately lead to regulatory approval of these agents within the next one to two years."
Source: Society of Nuclear Medicine and Molecular Imaging
Image credit: Wikimedia Commons
The PET/CT study focused on neuroendocrine tumours (NETs), a malignancy of nerve cells scattered throughout various organ systems that are sensitive to the signalling of neurotransmitters and hormones.
What makes the PET/CT technique novel is the use of an imaging agent known as Ga-68 DOTATOC, which binds to somatostatin receptors in neuroendocrine cells that use the hormone somatostatin to mediate endocrine function, the release of neurotransmitters and cellular proliferation. PET/CT with Ga-68 DOTATOC highlights malignant tumours because the agent binds to these more strongly than normal tissues.
Such an imaging technique could provide critical information that alters cancer patients' treatment. "Our study shows that Ga-68 DOTATOC PET/CT can change the management of patients with unknown primary tumour and neuroendocrine metastases, including potential surgical resection of the primary tumour," said lead author Yusuf Menda, MD, a researcher in the department of radiology and division of nuclear medicine at the University of Iowa. "There is evidence that surgery could lead to improved survival for these patients."
For the study, Ga-68 DOTATOC — short for gallium-68 1,4,7,10 tetraazacyclododecane-NI,NII,NIII,NIIII-tetraacetic acid (D)-Phe1-thy3 octreotide — was administered to 36 patients with metastatic NET and unknown primary tumour site. All patients underwent PET/CT imaging. Based on the results:
- 29 patients' scans revealed strong-binding metastases with a high affinity for the imaging agent, with 19 scans indicating the location of primary tumour.
- Confirmation of primary tumour site was achieved in three patients by further imaging and in eight patients by histological examination.
- Five suspected primary tumours remained undefined, and no primary tumour site was found for seven patients.
- False positives were noted for three patients.
Overall, 28 percent of patients underwent a major change in cancer management as a result of Ga-68 DOTATOC PET/CT.
"Ga-68 DOTATOC and similar agents are investigational drugs in the United States and are not yet approved for clinical use," notes Dr. Menda. "They are being investigated in clinical trials across several U.S. institutions. It is hoped that these studies and previous European published experience will ultimately lead to regulatory approval of these agents within the next one to two years."
Source: Society of Nuclear Medicine and Molecular Imaging
Image credit: Wikimedia Commons
Latest Articles
healthmanagement, cancer, neuroendocrine cancer, PET-CT, metastases, primary tumour
An investigational molecular imaging technique — combining positron emission tomography and computed tomography (PET/CT) — could be the key to finding the elusive primary tumour, according to a new study.