Company’s Latest Imaging Software Offers IHE Dose Reporting Capabilities, Image Quality and Productivity Enhancements for CR, DR Systems
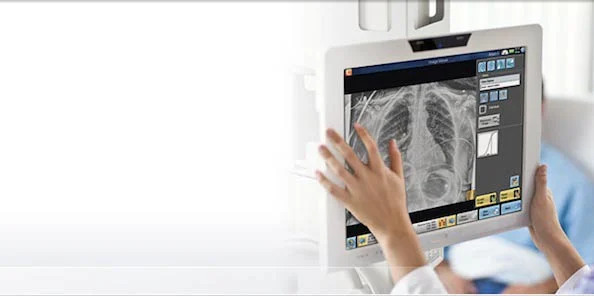
Carestream’s latest image acquisition software that includes innovative features for its portable and room-based CR and DR systems is now commercially available worldwide.
The software’s optional bone suppression feature, which recently received FDA clearance, creates a companion image (from the original exposure) that can improve visibility of lung nodules and other pathology by suppressing the appearance of posterior ribs and clavicles. Carestream’s current software already offers companion images that can enhance visualisation of tubes, PICC lines and pneumothorax (an abnormal collection of air or gas in the pleural space that separates the lung from the chest wall).
The software’s new IHE Dose Reporting capability will collect radiation dose information via the IHE (Integrating the Healthcare Enterprise) Radiation Dose Monitoring Profile from all Carestream DR and CR systems and distribute it to a healthcare provider’s PACS. Carestream demonstrated its ability to collect and share radiation dose information during the IHE North America Connectathon held earlier this year.
“Our new software collects radiation dose details from our CR and DR systems and sends this data to PACS systems that support the IHE dose monitoring profile,” said Helen Titus, Carestream’s Marketing Director of X-ray Solutions. “We are one of the first companies to provide radiation exposure data for CR and DR systems using the IHE profile. This information can then be linked to management systems that enable dose monitoring and reporting for each patient.”
Software Designed to Support Transition from CR to DR
The new software will allow a single console to support one CR and one DR system—or two CR systems—from Carestream to help boost workflow, save valuable space and lower costs. Users will be able to identify CR cassettes or DR detectors, view patient demographics and review images from one console for both imaging systems. The software will also offer other workflow improvements.
“This new software will support both CR and DR systems to smooth the transition and expansion to digital radiography for hospitals and imaging facilities of all sizes,” Titus explained.
The new software will be available for Carestream DRX systems and Carestream Directview Classic, Elite and Max CR systems. It will be offered as an upgrade to these CR and DRX platforms already installed at customer sites worldwide.
Source: Carestream
4 April 2014
Latest Articles
Imaging, Carestream, DR, Workflow, IT solutions, CR, imaging software, radiation dose, bone suppression
Company’s Latest Imaging Software Offers IHE Dose Reporting Capabilities, Image Quality and Productivity Enhancements for CR, DR Systems Carestream’...