ECR delegates will be advised on how best to cooperate with nuclear medicine physicians and radiation oncologists in order to improve imaging and radiotherapy outcomes during today’s Special Focus Session, ‘Imaging and radiotherapy: all you need to know’.
Prof. Regina Beets-Tan (pictured above), oncologic and abdominal radiologist from Maastricht University Hospital and Oncology Center, the Netherlands, will explore the future of oncologic imaging by addressing how new imaging biomarkers that highlight tumour heterogeneity can help radiation oncologists plan therapy. Radiologists have increasing value in radiation oncology as a result of rapid advances in the approach to dose distribution in radiotherapy, which requires a combined professional approach.
“They need us to guide their treatment. You will hear from my co-lecturers in radiation oncology, Prof. Vincenzo Valentini and Prof. Karin Haustermans, about intensity-modulated radiation therapy and dose painting. These are new ways to irradiate patients and shape dose distribution to match the differences in radiobiology across tumours. Higher doses are given to certain tumour areas that are more radio-resistant, the so-called ‘target within a target’, while sparing as much normal tissue as possible from damage,” she said.
“After Roentgen discovered x-rays in 1895 and Marie Curie discovered radium, radiologists used radiation to diagnose and treat cancer. After the introduction of megavolt radiation treatment, radiotherapy became more complex and required specific skills, so both specialties continued as separate disciplines,” Beets-Tan explained during the ECR Opening Press Conference on Thursday.
With the spectacular advances in imaging technology made over the last 40 years, the way radiologists visualise cancer has changed, and so has the way radiotherapists treat their patients. “Thirty years ago, radiotherapy fields were standardised, including large target volumes, in order not to miss tumours. These protocols presented a high risk of harming healthy surrounding tissue. Thanks to modern imaging such as CT, MRI, PET-CT and, soon, MR-PET, radiation treatment has moved away from standard therapy to personalised treatment, which increases the therapeutic index and reduces the costs,” she said.
Cooperation between both disciplines in cancer management has reached unprecedented levels of efficiency with the new tools at their disposal. Radiologists have morphological and functional imaging while radiotherapists can use intensity-modulated radiation treatment (IMRT) and dose painting, a technique that uses high radiation doses to kill tumours without compromising normal tissue.
But it is also the result of increased collaboration at the political level, Beets-Tan believes. “The European Society of Radiology (ESR) and the European Society for Radiotherapy and Oncology (ESTRO) recognise their synergy by working together and collaborating in multidisciplinary education,” she said.
“Now that therapy and diagnosis have been separated, it’s time to get together again. Two technology-oriented disciplines have become true patient-oriented disciplines. And through collaboration and synergy they both have a chance to become leaders in these new paradigms,” Beets-Tan concluded.
With dose shaping, radiation oncologists increasingly rely on imaging to identify a tumour’s heterogeneity and evaluate response both during and after radiotherapy. They want radiologists to show them which regions in the tumour are more resistant to their treatment and how these regions respond during the radiation treatment so that the dose can be adapted.
Functional and metabolic imaging is a more accurate tool than morphological assessment of radiotherapy response evaluation, and PET and MRI can offer improved imaging in this respect, according to Beets-Tan. MRI has an advantage as it can display morphology in high resolution, as well as functional images of tumour biology and behaviour. Imaging biomarkers, in particular, can provide an objective measure of pathophysiological processes. “This is why I think the future of MR imaging will involve diffusion, perfusion, proliferation, and further into the future still, hypoxia and automated image segmentation. This is where we radiologists will have a significant role in our collaboration with radiation oncologists,” she said. However, validation of these techniques, as well as protocol standardisation and implementation, is required before they start benefiting the patient.
Perfusion MRI of a rectal tumour compared with histology
(All images provided by Prof. Regina Beets-Tan, Maastricht University Hospital and Oncology Center, the Netherlands)
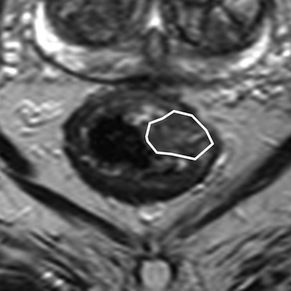
T2-weighted MR image of a patient with rectal tumour (encircled) before chemoradiotherapy.
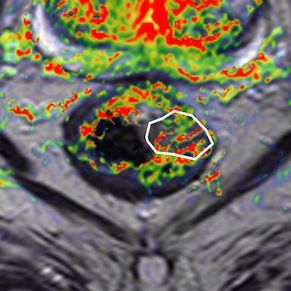
Corresponding perfusion MR image (K-trans map). There is a heterogeneity in tumour angiogenic activity with areas of higher (red) and lower K-trans values.
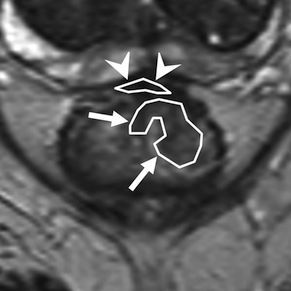
T2-weighted MR image of the same patient after irradiation of the tumour
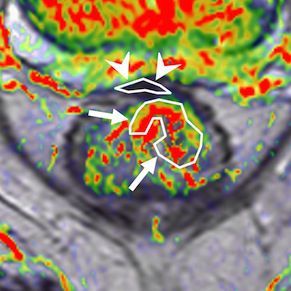
Corresponding perfusion MR image (K-trans map). Residual tumour (white arrows) is visualised, showing persistent heterogeneous angiogenic activity with areas of high (red) and low K-trans values (arrowheads pointing at fibrosis in the anterior rectal wall)
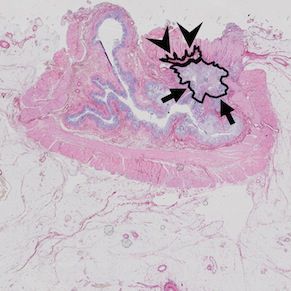
Histopathology confirmed the areas of residual tumour (arrows) and fibrosis (arrowheads)
Hybrid imaging, and in particular PET-CT, will also play a role in radiotherapy planning in the future, and it will require a more multidisciplinary approach, according to Dr. Annika Loft, chief physician from the department of clinical physiology, nuclear medicine and PET, Rigshospitalet, Copenhagen University Hospital, Denmark, who will speak during the session.
Ideally, a nuclear physician together with a radiologist should identify malignancies on the PET-CT scan, while the oncologist is in charge of treating the patient and devising an individual treatment plan, she said.
PET-CT provides the functional information of PET with the anatomical imaging of CT, and the combination helps with planning the volume for radiotherapy. “You might find lymph nodes that are too small to be defined as malignant on CT, but are definitely malignant on PET because of FDG uptake, so planning volume would increase. Conversely, planning volume would decrease if nodes are suspicious on CT but definitely look non-malignant on PET,” said Loft, who will also focus on the misreading of scans and reducing false positives by showing some clinical examples.
Latest Articles
#ECR2013, Radiology, Radiotherapy
ECR delegates will be advised on how best to cooperate with nuclear medicine physicians and radiation oncologists in order to improve imaging and radiother...